Package
PRBCs
Plasma
Platelets
Cryoprecipitate
Initiation
6 units (UD/TS)
6 units (UD)
1 (0.5 h)
6 units (UD/TS)
6 units (UD)
1 apheresisa
2 (1 h)
6 units (UD/TS)
6 units (TS)
20 units
3 (1. 5 h)b
6 units (UD/TS)
6 units (TS)
1 apheresisa
4 (2 h)
6 units (UD/TS)
6 units (TS)
10 units
5 (2.5 h)
6 units (UD/TS)
6 units (TS)
1 apheresisa
6 (3 h)c
6 units (UD/TS)
6 units (TS)
10 units
Table 16.2
Open abdomen coverage techniques
Skin only | Polypropylene mesh |
|---|---|
Towel clip | Polyglycolic/polyglactic acid mesh |
Silastic sheet | Polytetrafluoroethylene mesh |
Bogota bag | Parachute silk |
3-liter genitourinary bag | Hydrogel/Aquacel |
Steri-drape/x-ray cassette | Ioban |
Zippers | Vacuum pack |
Slide fasteners | Abdominal wound VAC |
Velcro analogue/Wittmann | Bioprosthetics |
Vascular Damage Control Surgery (DCS) Indications
The maturation of DCS has led to fundamental tenants that include (1) arresting surgical hemorrhage, (2) containment of gastrointestinal spillage, (3) surgical sponge insertion, and (4) temporary abdominal closure. This sequence is followed by immediate transfer to the intensive care unit with subsequent rewarming, correction of coagulopathy, and hemodynamic stabilization. Return to the operating theater is then pursued 6–48 h later for a planned re-exploration that includes definitive repair and primary fascial closure if possible. It is clear that the DCS approach leads to improved survival for both blunt and penetrating injures in patients who are approaching physiologic exhaustion [12].
Despite the clear utility of DCS, its widespread propagation throughout the trauma community has led to a clear overutilization of this technique. More specifically, multiple injured patients who are not approaching physiologic exhaustion are often exposed to the potential risks associated with open abdomens. As a result, the pertinent question remains: who needs DCS? The succinct response is “patients who are more likely to die from uncorrected shock states than from failure to complete organ repairs.” In essence, these are metabolic cripples who continue to suffer the sequelae of tissue shock that is manifested as persistent hypothermia, persistent metabolic acidosis, and nonmechanical (i.e., nonsurgical) bleeding. More specifically, DCS triggers include core temperature <35 °C, pH < 7.2, base deficit > −15, and/or significant coagulopathies [13–16]. It must be emphasized however that not even all patients with initial physiologic deficits as significant as these values mandate DCS[17–33]. With rapid arrest of hemorrhage, as well as ongoing resuscitation, some patients will improve dramatically in all parameters on repeated intraoperative blood gases. These patients stabilize and begin to recover. It should also be stated that patients with multiple intra-abdominal injuries are not always in metabolic failure.
Vascular Damage Control Techniques
Although it is clear that arresting ongoing hemorrhage is the most crucial of damage control tenants, vascular damage control has been traditionally limited to vessel ligation . More recently, however, balloon catheter tamponade and temporary intravascular shunts (TIVS) have increased in popularity. The impressive utility of balloon catheters for tamponade of exsanguinating hemorrhage has a long history dating back more than 50 years [34]. Although this technique was originally described for esophageal varices [35], it was quickly extended to patients with traumatic vascular and solid organ injuries [36]. Since the initial treatment of an iliac arteriovenous lesion in 1960 [3], balloon catheters have also been used for cardiac [37], aortic [38], pelvic vascular [39], neck (carotid, vertebral, and jugular vessels) [40, 41], abdominal vascular [42], hepatic vascular [43], subclavian [44], vertebral [34], and facial vascular trauma [45]. While this technique was originally intended as an intraoperative endovascular tool [34], it has since been employed as an emergency room maneuver with the balloon being placed outside of the lumen of the injured vessel [46, 47].
Balloon Catheter Tamponade
Modern indications for this damage control technique are limited. This is primarily because routine methods for controlling hemorrhage , such as direct pressure, are typically successful. As a result, indications for catheter tamponade include (1) inaccessible (or difficult to access) major vascular injuries, (2) large cardiac injuries, and (3) deep solid organ parenchymal hemorrhage (liver and lung) [34, 37]. The specific type of balloon catheter (Foley, Fogarty, Blakemore, or Penrose with Red Rubber Robinson) (Figs. 16.1 and 16.2), as well as the duration of indwelling, can vary significantly. The take-home message is to ensure a selection of various catheters is available in a central kit that is easily accessible within the operating theater and emergency department . In its purest essence, balloon catheter tamponade is a valuable tool for damage control of exsanguinating hemorrhage when direct pressure fails or tourniquets are not applicable. It can be employed in multiple anatomic regions and for variable patterns of injury.
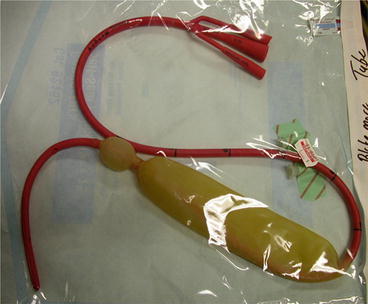
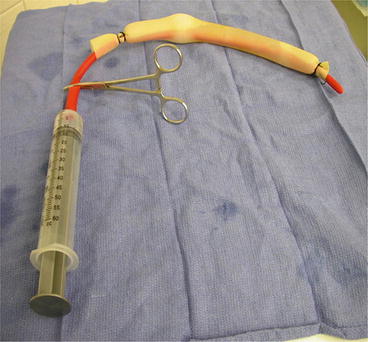

Fig. 16.1
Blakemore occlusion balloon

Fig. 16.2
Red rubber/Penrose occlusion balloon
The technical nuances and skill required to successfully insert a balloon catheter into a wound or organ with ongoing hemorrhage are relatively minimal. Think of the balloon and the wound as a geometric puzzle. Select the type of balloon that you think will best fit within the space. This may range from a Foley (penetrating neck wound) to a Blakemore (central hepatic gunshot wound) to a Fogarty (insertion into the internal carotid artery when it is sheared off of the mastoid via a penetrating wound) catheter (Figs. 16.3, 16.4, 16.5, 16.6, and 16.7). The important point is to make the decision to insert the balloon early after an initial one or maximum of two other techniques have failed. An experienced clinician will usually recognize wound dynamics and geometry and select the appropriate balloon as a primary hemostatic choice. Once the catheter is inserted into the wound, it should be gently inflated with water. If the hemorrhage stops, then the catheter should be either tied off with a knot or clamped to prevent both movement and blood flow through some balloon devices (i.e., Foley). If the catheter is left in place for any significant length of time, it should be secured with copious amounts of tape and warning labels begging all caregivers not to touch the catheter itself. If the ongoing hemorrhage is not stopped by the initial insertion of the catheter, there is one of two potential problems: (1) you’ve used the wrong balloon for the geometry of the wound, or (2) the balloon needs to be repositioned. In the second scenario, desufflate the balloon and either advance or retract it and then reinflate it again. This nuanced cycle may be required more than once. Don’t be discouraged!! If the skin wound is too large to keep the catheter contained and it continues to pop out of the wound (e.g., neck), then close the skin around the tube itself (i.e., similar to a chest tube suture) to lock it into place. As previously mentioned, a successfully placed balloon catheter can remain in place for an extended duration (i.e., prolonged interval for central hepatic gunshot wounds) .
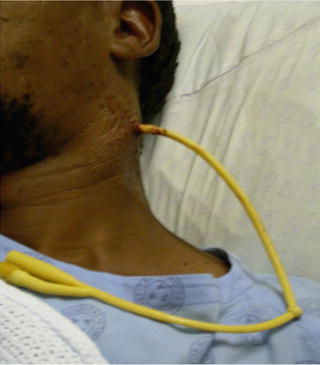
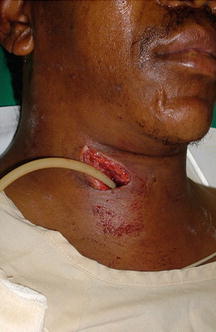
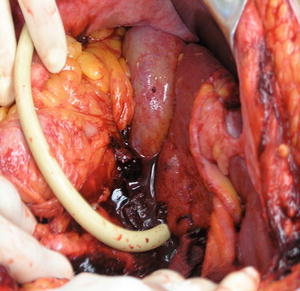
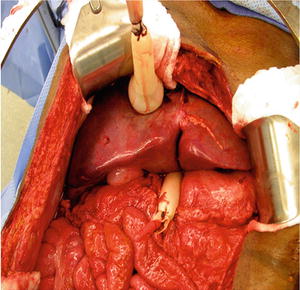
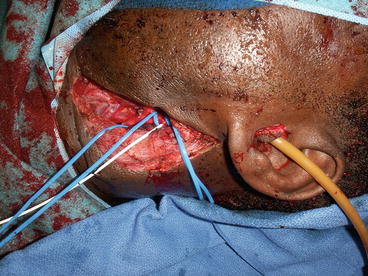

Fig. 16.3
Cervical Foley catheter balloon occlusion

Fig. 16.4
Cervical Foley catheter balloon occlusion

Fig. 16.5
Balloon occlusion for central hepatic gunshot wound

Fig. 16.6
Deflation of balloon occlusion for central hepatic gunshot wound

Fig. 16.7
Foley catheter balloon occlusion of the severed internal carotid artery at the skull base
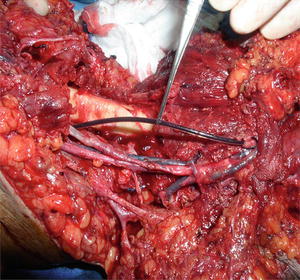
Temporary Intravascular Shunts
Temporary intravascular shunts (TIVS ) are intraluminal synthetic conduits that offer nonpermanent maintenance of arterial inflow and/or venous outflow [48] (Figs. 16.8 and 16.9). As a result, they are frequently life- and limb-saving when patient physiology is hostile. By bridging a damaged vessel and maintaining blood flow, they address both acute hemorrhage and critical warm ischemia of distal organs and limbs. Although Eger and colleagues are commonly credited for pioneering the use of TIVS in modern vascular trauma [49], this technique was initially employed by Carrel in animal experiments [50]. The first documented use in humans occurred in 1915 when Tuffier employed paraffin-coated silver tubes to bridge injured arteries [51]. This technique evolved from glass to plastic conduits in World War II [52] and continues to vary both in structure and material among today’s surgeons [53].
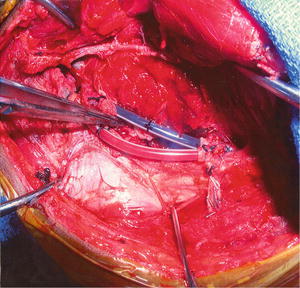
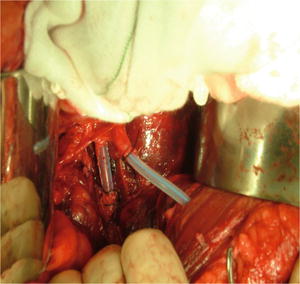

Fig. 16.8
Vascular shunt of the iliac artery and vein

Fig. 16.9
Vascular shunt of the iliac artery and ureterostomy intubation
Modern indications for TIVS include (1) replantation, (2) open extremity fractures with concurrent extensive soft tissue loss and arterial injury (Gustilo IIIC) (Fig. 16.10), (3) peripheral vascular damage control, (4) truncal vascular damage control (Fig. 16.11), and (5) temporary stabilization prior to transport [48, 54]. While the understanding of TIVS use for military and civilian settings is increasing [53], the optimal shunt material, dwell time, and anticoagulation requirements remain poorly studied. It can be noted however that TIVS are remarkably durable and rarely clot off unless they (1) are too small (diameter), (2) kink because of inappropriate length, and/or (3) are placed in an extremity without appropriate (or shunted) venous outflow (venous hypertension leads to arterial thrombosis) [54].