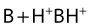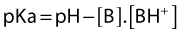CHAPTER 3 Local anesthetics
The peripheral nerve
Applied anatomy
The typical nerve cell has been traditionally described in terms of having a cell body (perikaryon), multiple dendrites, and a single axon (Fig. 3.1). Sensory neurons are classified as unipolar; that is, they have an axon that divides to extend a branch to both the spinal cord and the periphery. Motor neurons are classified as multipolar because, in addition to an axon, they possess many dendrites. Impulses arriving via the dendrites and cell body are integrated at the axon hillock, a specialized area of the cell body. Summation of excitatory and inhibitory impulses occurs at the axon hillock and determines whether impulses are generated or not.
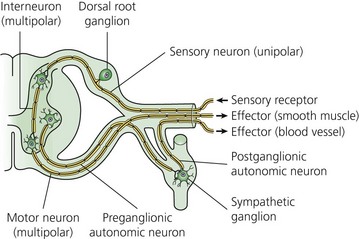
(From Ref. 1, Strichatz GR. Neural Physiology and Local Anesthetic Action. In: Cousins MJ, Bridenbaugh PO (eds). Neural blockade in clinical anesthesia and management of pain, 3rd edn. Philadelphia: © Lippincott-Raven; 1998.)
The axon is always enclosed within a nutriprotective Schwann cell envelope. Most are further invested in a myelin sheath formed by a single Schwann cell wrapped many times around the axon and interrupted periodically at the nodes of Ranvier. Many unmyelinated nerves, on the other hand, may have their axons enclosed within the folds of a single Schwann cell (Fig. 3.2).
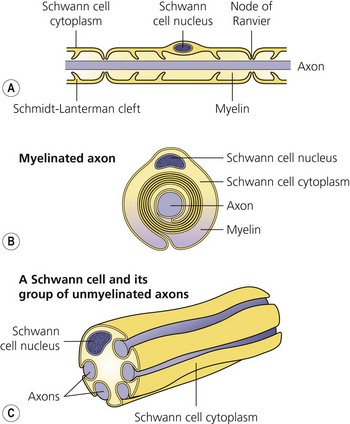
(From Ref. 1, Strichatz GR. Neural physiology and local anesthetic action. In: Cousins MJ, Bridenbaugh PO (eds). Neural blockade in clinical anesthesia and management of pain, 3rd edn. Philadelphia: © Lippincott-Raven; 1998.)
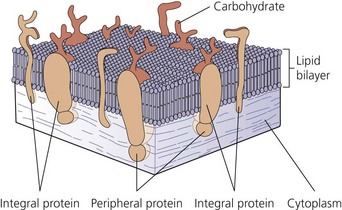
The nerve cell membrane, in common with all cells of the body, comprises a phospholipid bilayer traversed by proteins that selectively regulate the influx and efflux of ions and molecules, act as hormone and transmitter receptors, are involved in cell-to-cell interactions, and enhance the structural integrity of the membrane (Fig. 3.3). It is the specialized nature of some of these proteins that is responsible for the unique character of nerve cells.2
Ionic basis of conduction
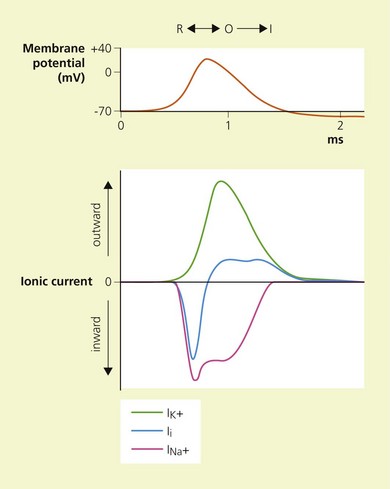
A special membrane protein, the Na+–K+ ATPase pump, is responsible for the transmembrane concentration gradient of these ions peculiar to nerve cells. It transports sodium out of the cell and potassium into it.3 At rest, the membranse is selectively permeable to K+, resulting in an efflux of positive charge. Thus, the interior of the cell is negatively charged relative to the exterior; this resting membrane potential is in the order of 70–80 mV. Because of its chemical and electrical gradient, there is a tendency for Na+ to enter the cell.
Temporal and spatial summation of excitatory and inhibitory potentials occurs at the axon hillock. Small net depolarizations of 15–20 mV will raise the membrane potential to −55 mV, resulting in a voltage-dependent opening of Na+ channels and a rapid change in transmembrane potential to +40 mV.4–6 This is shortly followed by the opening of K+ channels, and the subsequent outward flow of K+ returns the membrane potential to normal and beyond (the refractory period where it is more difficult to stimulate the nerve).3 The Na+−K+ pump then serves to restore the chemical gradient to its initial state. These changes in transmembrane potential account for the familiar action potential (Fig. 3.4). The electrical changes occurring during the action potential serve to open adjacent voltage-dependent Na+ channels, and so the action potential is propagated along the axon. Because the area immediately preceding the action potential is in the refractory period, the action potential is propagated in one direction only.
Structure and function of local anesthetics
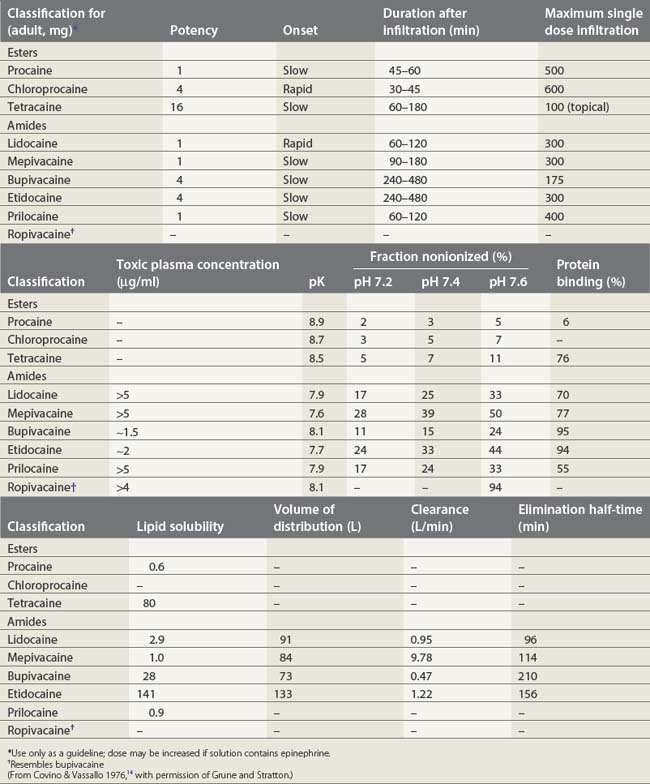
Local anesthetics consist of a lipophilic aromatic ring connected by a hydrocarbon chain to a hydrophilic tertiary amine (Fig. 3.5). The lipophilic moiety is responsible for the anesthetic activity of the molecule. The drugs are classified as amide or ester local anesthetics based on the nature of the bond linking the hydrocarbon chain and the aromatic ring. The ester drugs are rapidly hydrolyzed by plasma and other esterases,8–12 and have been associated with allergic and hypersensitivity reactions linked to their breakdown product para-aminobenzoic acid.13 In contrast, amides are relatively stable compounds, are metabolized in the liver, and allergic reactions to them are exceedingly rare. The comparative pharmacology of local anesthetics is shown in Table 3.1.
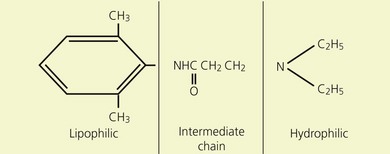
(From Ref. 7, Stoelting RK. Pharmacology and physiology in anesthetic practice. 2nd edn. Philadelphia: © JB Lippincott; 1991.)
Local anesthetics produce conduction blockade through reversible inhibition of Na+ channel function.15,16 Physiological studies have demonstrated that local anesthetics inhibit stimulated channels more readily than resting channels; this is known as phasic block and tonic block, respectively.17 The modulated receptor hypothesis has been proposed to explain these features.18,19 It is based on the fact that Na+ channels pass through various states during membrane depolarization. They begin in the resting state (R), pass through an intermediate closed form (C), to reach an open form (O), and then close to reach an inactivated state (I). According to the modulated receptor hypothesis, local anesthetics have greater affinity for Na+ channels in the O and I configurations than in the C and R configurations. Thus, local anesthetics will more readily bind Na+ channels of stimulated or active nerves.
Two possible binding sites for local anesthetics have been identified on the Na+ channel.15,18 The first site is thought to be responsible for phasic block and is situated near the channel pore. Binding and unbinding from this site is relatively slow. The second site is on the inner aspect of the channel in the hydrophobic center of the membrane. Binding and dissociation at this site is rapid.
Pharmacodynamics
Their ratio is given by the Henderson–Hasselbach equation:
Both the ionized and non-ionized forms can inhibit Na+ channels.20–23 The observations that tertiary amine local anesthetics are more potent when applied externally at an alkaline pH, or applied directly internally, suggest that the neutral form of the local anesthetic traverses the membrane, where it assumes its ionized form once again to become active at the internal aspect of the Na+ channel.24 Following injection, the alkaline pH of the tissues releases the base:
Physiochemical properties of local anesthetics
Ionization
The degree of ionization depends on the pKa of the agent and the ambient pH. The pKa is defined as the negative logarithm of the dissociation constant (Ka) of the conjugate acid. It is equal to the pH at which the local anesthetic is 50% ionized. The greater the pKa of the base, the smaller the proportion existing in its non-ionized form at any pH, and so the slower the speed of onset.25,26

Full access? Get Clinical Tree