153 Venous Thromboembolism in Medical-Surgical Critically Ill Patients
Venous thromboembolism (VTE) is a common complication of serious illness, conferring considerable morbidity and mortality in hospitalized patients. Patients with deep vein thrombosis (DVT) are at risk of subsequently developing pulmonary embolism, which may be fatal if untreated. Approximately 90% of cases of pulmonary embolism are believed to arise in the lower limbs,1 so DVT can be viewed as an important precursor to more serious disease. Most clinical research on VTE in the intensive care unit (ICU) is focused on DVT, and it will be the major focus of this chapter.
In the ICU, patients with DVT are significantly more likely to have pulmonary embolism2 and a longer duration of mechanical ventilation (P = .02), ICU stay (P = .005), and hospitalization (P < .001) than patients without DVT.3 Clinically unsuspected DVT and pulmonary embolism are found frequently at autopsy in critically ill patients.4–6
Prophylaxis against VTE was rated the number-one patient safety initiative for hospitalized patients in the U.S. Agency for Health Care Policy Research Evidence Report and Technology Assessment document.12 Juxtaposed against the foregoing is the invisibility of medical-surgical critically ill patients in publications such as the National Institutes of Health Consensus Conference on Prevention of Venous Thrombosis and Pulmonary Embolism,13 the European Consensus Statement on Prevention of Venous Thromboembolism,14 the Thromboembolic Risk Factors Consensus Conference,15 the Fifth American College of Chest Physicians Antithrombotic Consensus Conference,16 and the American Thoracic Society Clinical Practice Guideline on Diagnosis of Venous Thromboembolism.17 An editorial in 1998 stated that the medical-surgical ICU was “the last frontier for prophylaxis.”18
 Risk Factors for Venous Thromboembolism in Medical-Surgical ICU Patients
Risk Factors for Venous Thromboembolism in Medical-Surgical ICU Patients
Clinical Risk Factors
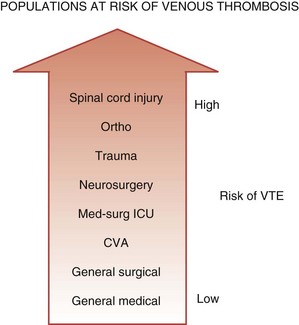
One conceptualization of risk factors for VTE in the ICU is to consider ICU admitting diagnosis as a risk factor. Medical-surgical ICU patients are at higher risk of VTE than general medical or surgical patients cared for on the ward, but at lower risk than other subgroups of critically ill patients such as trauma victims or neurosurgical patients (Figure 153-1). In the largest prospective cohort study using venographic diagnosis, of 716 trauma patients who did not receive prophylaxis, 201 (58%) had DVT between days 14 and 21, one-third of which were in the proximal venous circulation (and likely of clinical significance).19 Of these 201 patients, only 3 patients had symptoms of DVT. Among neurosurgical patients in three cohort studies using radioactive iodine leg scanning, the DVT rate was 35% without prophylaxis; in 7 randomized clinical trials that included a nonprophylaxis arm, the pooled incidence of DVT was 22%.7 Patients with acute spinal cord injury have been evaluated in 4 randomized trials and 6 cohort studies, 5 of which did not use prophylaxis.7 Four studies using either radioactive iodine fibrinogen or impedance plethysmography identified DVT in 39% to 90% of patients. In the single study using the reference standard for the diagnosis of DVT, which is ascending venography, 81% of the subgroup of trauma patients with spinal cord injury had DVT.19
Another conceptualization of risk factors for VTE in the ICU is to consider patient characteristics, events, and exposures that increase the risk of VTE. Critically ill patients have an increased risk of VTE due acute and chronic illnesses, immobility propagated by sedatives and paralytic drugs, and thrombin-generating invasive procedures. Observational studies in medical-surgical ICU patients have identified VTE risk factors11,20 including patient demographics (e.g., female sex), prior VTE events (i.e., personal history of VTE), morbidity (e.g., malignancy), ICU procedures (e.g., central venous catheters), treatments (e.g., mechanical ventilation), and VTE prophylaxis (i.e., decreasing risk). Inferences about many of these risk factors are limited by small sample sizes and infrequent use of multiple logistic regression to rigorously evaluate baseline and time-dependent risk factors.
Studies large enough to perform multivariate analysis are most helpful.2,3,21,22 In a prospective cohort study of patients ventilated for at least 1 week, the only independent risk factor for VTE was central venous catheterization; each day the catheter was in place was associated with a relative risk (RR) increase of 1.04.2 In another prospective cohort study,3 we enrolled consecutive medical-surgical patients 18 years of age or older expected to be in the ICU for 72 hours or more. Exclusion criteria were an admitting diagnosis of trauma, orthopedic surgery, pregnancy, and life-support withdrawal. We performed bilateral lower extremity compression ultrasound within 48 hours of ICU admission, then twice weekly thereafter or if VTE was clinically suspected. Thromboprophylaxis was protocol-directed and universal using unfractionated heparin. We recorded DVT risk factors at baseline and daily, using multivariate regression analysis to determine independent predictors. Patients were followed to hospital discharge. Among 261 patients with a mean Acute Physiology, Age, and Chronic Health Evaluation (APACHE) II score of 26, we identified four independent risk factors for ICU-acquired DVT: personal or family history of VTE (hazard ratio, 3.9; 95% confidence interval [CI], 1.5-10), end-stage renal failure (hazard ratio, 3.7; 95% CI, 1.3-11.2), platelet transfusion (hazard ratio, 3.2; 95% CI 1.2-8.5), and vasopressor use (hazard ratio, 2.8; 95% CI 1.1-7.2).
Congenital Hypercoagulable States
A growing number of epidemiologic studies have highlighted how inherited and acquired abnormalities in the coagulation system predispose to VTE. Activated protein C resistance due to factor V Leiden (found in 5% of the population) is the most common hereditary biochemical defect that predisposes to venous thrombosis, followed by the prothrombin 20210A regulatory sequence mutation, found in 2%.23–25 Although the impact of these prothrombotic states on the risk of VTE is confounded by the use of prophylactic anticoagulants, there is some evidence that these states increase the risk of first DVT in patients in high-risk clinical situations. Lowe and colleagues,26 in a large prospective cohort study of patients undergoing elective hip replacement, found in a univariate analysis that patients with the factor V Leiden mutation had an increased risk of postoperative venous thrombosis. No large-scale studies have yet reported on the incremental risk of DVT in high-risk situations for patients with factor V Leiden, but it is known that the prothrombin gene mutation predicts DVT in otherwise healthy outpatients (odds ratio [OR], 2.8).27 Additional but less common inherited hypercoagulable states include deficiencies of antithrombin, protein C, and protein S, each of which is a naturally occurring anticoagulant protein. The ORs for venous thrombosis are 8.1 to 13.7 for antithrombin deficiency, 7.3 to 11.9 for protein C deficiency, and 8.5 to 10 for protein S deficiency.28–30 Antiphospholipid antibodies including the lupus anticoagulant and anticardiolipin antibody are strong predictors of first and recurrent venous thrombosis. Elevations in the levels of homocysteine and coagulation factors VIII, IX, and XI also predispose to VTE in other settings.31–35
In the observational study described earlier, we evaluated the frequency and clinical importance of thrombophilia markers at the time of ICU admission and during the ICU stay.36 To examine whether baseline markers of activation of the coagulation system and known thrombophilic risk factors predicted the development of DVT, a comprehensive battery of tests was done at the time of enrollment, including activated protein C ratio (with confirmation of factor V Leiden where appropriate), protein C level, protein S level, antithrombin level, anticardiolipin antibody titer, and screening and confirmatory assays for the lupus anticoagulant. The receiver operating curves for four baseline coagulation tests at the time of ICU admission showed areas under the curve for each of the activated protein C ratio, antithrombin, protein C, and protein S tests that were not significantly different than 50%; that is, the presence of these abnormalities did predict the presence of DVT at the time of ICU admission. Tests with areas under the curve of 0.75 to 0.80 represent moderate diagnostic power. Baseline coagulation tests also were not useful predictors of DVT developing during the ICU stay.
Acquired Hypercoagulable States
Coagulation abnormalities acquired in the ICU have received considerable attention. Acquired thrombophilic markers associated with thrombosis include lupus anticoagulant, anticardiolipin antibody, and increased levels of homocysteine. In critically ill patients, acquired reductions in the levels of antithrombin, protein C, and protein S due to consumption may be common, and it is possible these deficiencies are associated with a high risk of VTE and other complications of ICU stay including death. The relationship between the inflammatory and coagulation cascades has been the focus of intense discussion in the sepsis literature.37 Longitudinal studies have shown that protein C levels in sepsis are inversely correlated with mortality.38 A randomized trial of recombinant activated protein C in 1690 patients with systematic inflammation and organ dysfunction showed a decrease in 28-day mortality from 30.8% to 24.7% (number needed to treat, 16).39 Approximately 80% of patients had protein C deficiency on entry into the trial, highlighting the prevalence of this acquired thrombophilic marker. The efficacy of recombinant activated protein C was the same, however, in patients with and without protein C deficiency. In another large randomized trial of antithrombin administration in patients with sepsis, antithrombin levels were less than 60% of normal functional levels in more than 50% of patients, but antithrombin administration did not decrease mortality.40
In the study of DVT incidence described earlier,3 we also evaluated whether quantitative D-dimer tests at the time of ICU admission and during ICU stay41 were associated with DVT. At the time of enrollment, twice weekly during the ICU stay, and at the time of any suspected venous thromboembolic events, patients had a battery of D-dimer tests, including whole-blood SimpliRed D-dimer tests, and five D-dimer assays performed using D-dimer Plus, IL test DD, MDA-DD, Sigma DD, and Biopool. For the five quantitative baseline D-dimer tests in relation to DVT detected at the time of ICU admission, the areas under the curve for each of D-dimer Plus (P = .01), MDA-DD (P = .002), and Sigma DD (P = .054) were significantly different from .50. The receiver operating curves for time-dependent quantitative D-dimer tests and DVT developing during the ICU stay did not differ from 50%, indicating that D-dimer tests are not useful for predicting the development of VTE in the ICU.
Summary
Venous thromboembolism is a multicausal disease.42 In considering clinical risk factors, it is useful to classify them into risk factors that are fixed, such as admitting diagnoses, and risk factors that are modifiable, such as invasive procedures. Modifiable risk factors can form the basis of VTE prevention strategies. Studies to analyze the relative contributions of congenital and acquired thrombophilia markers suggest that these markers are not useful for screening or diagnostic purposes in the ICU.
 Prevalence and Incidence
Prevalence and Incidence
The incidence of VTE in the ICU depends on whether the events are clinically diagnosed or detected by screening methods. Venous thromboembolism rates observed in usual clinical practice are much lower than rates observed during systematic screening, because the former primarily represent diagnoses prompted by signs or symptoms. For example, 10%43 to 100%11,44 of proximal DVTs found by ultrasound screening were clinically unsuspected. In this section, we report the incidence of VTE in critically ill patients based on studies using systematic screening methods for case identification.
Understanding DVT rates requires distinguishing events diagnosed at the time of ICU admission (prevalence at a point in time) from the events that develop over the course of critical illness (incidence over the ICU stay). Cross-sectional studies at the time of admission to a medical ICU45 and surgical ICU44 suggest a 10% prevalence of DVT diagnosed by screening compression ultrasonography. As mentioned earlier in the section on risk factors, however, the prevalence of DVT on admission to any ICU is influenced heavily by the case mix of patients.
The risk of DVT developing over the ICU stay was established in three longitudinal studies using systematic screening.11,43,46 Among ICU patients not receiving prophylaxis, 76% of whom were mechanically ventilated, radioactive iodine fibrinogen scanning for 3 to 6 days identified DVT in 3 of 34 (9%) patients.46 Using Doppler ultrasound twice weekly then at 1 week after ICU discharge in 100 medical patients expected to stay more than 48 hours—70% of whom were ventilated—DVT was diagnosed in 32% of 100 patients receiving no prophylaxis, in 40% of patients receiving unfractionated heparin, and in 33% of patients who received mechanical prophylaxis.11 In a third study of 102 medical-surgical ICU patients undergoing duplex ultrasound during days 4 to 7 and as clinically indicated,43 DVT rates were 25%, 19%, and 7% in patients receiving no prophylaxis, mechanical prevention, and unfractionated heparin.
Earlier studies suggest that the prevalence of proximal DVT on admission to a medical-surgical ICU is estimated to be 10%, and the incidence of DVT developing over the ICU stay based on systematic screening ranges from 9% to 40%. Two of these studies performed surveillance for approximately 1 week,43,46 however, and one study used radioactive iodine fibrinogen scanning for detection,46 which likely underestimated the risk of ICU-acquired DVT. No studies used systematic screening for pulmonary embolism, and the true incidence of pulmonary embolism is not known.
More recent studies suggest a lower rate of VTE in medical-surgical ICU studies, partly due to the administration of thromboprophylaxis. In a single-center cohort of 239 medical ICU patients who did not undergo systematic screening ultrasound, 44 (18.4%) patients had lower-extremity DVT.47 Ibrahim et al.,2 in a cohort study involving twice-weekly upper- and lower-extremity ultrasound screening, found a 26.6% incidence of DVT. Among 261 patients with a mean APACHE II score of 25.5 (±8.4), the prevalence of DVT was 2.7% (95% CI, 1.1-5.5) on ICU admission, and the incidence was 9.8% (95% CI, 6.5-14.2) over the ICU stay.3
< div class='tao-gold-member'>

Full access? Get Clinical Tree