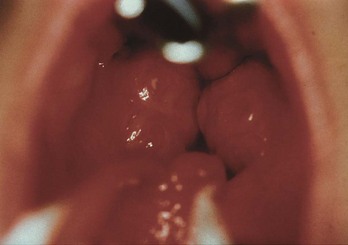
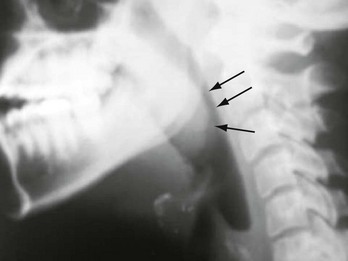
Chapter 75 Viruses are responsible for most cases of pharyngitis in both children and adults. Group A beta-hemolytic Streptococcus (GABHS) is the most common bacterial cause of pharyngitis in children, with a peak incidence of 30%. As in adults, non–group A streptococci, Mycoplasma pneumoniae, and Chlamydia pneumoniae infections are on the rise in pediatric populations.1–4 In adults, sexually transmitted diseases should be considered, as well.1–8 Whereas immunization has led to a decline in diphtheria as a cause of pharyngitis, it can result in serious complications and needs to remain in any differential diagnosis. Mixed aerobic and anaerobic bacteria often cause chronic or recurrent pharyngitis, especially those that produce β-lactamase. Epstein-Barr virus (EBV) and Actinomyces are also implicated in chronic or recurrent pharyngitis. Rare causes of bacterial pharyngitis include Francisella tularensis, Yersinia pestis, and Yersinia enterocolitica.1,2,5 The most common symptom is pharyngeal pain that is aggravated by swallowing and may radiate to the ears. Examination usually reveals fever, pharyngeal erythema, pharyngeal or tonsillar exudate, and tonsillar enlargement (Fig. 75-1). The infection tends to localize to lymphatic tissue and produces suppuration and swelling of the tonsils, along with tender cervical adenopathy. Occlusion of the eustachian tubes may result in secondary otitis media. Clinical differentiation of the causative organisms is virtually impossible.1,2,5–7 Viral pharyngitis usually occurs in conjunction with cough, rhinorrhea, myalgia, headache, stomatitis, conjunctivitis, exanthem, and odynophagia. Low-grade fever and pharyngeal exudates may be present. Cervical lymphadenopathy is generally absent.1,2 Mild pharyngeal edema and erythema associated with a “scratchy” throat are present in 50% of patients with the common cold. Systemic viral infections, including measles, cytomegalovirus (CMV), rubella, and human immunodeficiency virus (HIV), may initially manifest as mild pharyngitis.1,2,5 HIV and CMV pharyngitis may be clinically indistinguishable from infectious mononucleosis.1,2 Influenza occurs in epidemics and is associated with high fever, myalgia, and headache. Although 50 to 80% of patients with influenza experience pharyngeal discomfort, pharyngeal exudate and cervical lymphadenopathy are rare. Adenovirus may cause severe exudative pharyngitis with cervical adenitis similar to that in streptococcal pharyngitis. Thirty percent to 50% of cases of adenoviral pharyngitis are associated with a follicular, usually unilateral, conjunctivitis, and preauricular lymphadenopathy. Coxsackieviruses are the most frequent causes of hand-foot-and-mouth disease and herpangina.1,2 Pharyngitis is a common manifestation of infectious mononucleosis (caused by EBV) in young adults.1,2,5 Symptoms develop after an incubation period of 4 to 7 weeks. Fever and a tonsillar exudate or a membrane (that is cheesy or creamy white) is often present. Cervical as well as generalized lymphadenopathy (90-100%) and splenomegaly (50%) are usually noted, and palatal petechiae may be present. Hepatomegaly is present in 10 to 15% of cases. Periorbital edema and rash are rare findings. In up to 90% of patients with mononucleosis who are given ampicillin or amoxicillin, a diffuse macular rash develops that may be misdiagnosed as an allergic reaction.1,2 Patients with early (days to weeks) HIV infection can develop an acute retroviral syndrome. This is manifested by fever, sore throat, generalized nontender lymphadenopathy, a diffuse maculopapular rash, arthralgias, mucocutaneous ulcerations, and, commonly, diarrhea. Nonexudative pharyngitis is present in 50 to 70% of patients. Oral thrush and ulcers may be present. Acute HIV infection can be differentiated from infectious mononucleosis by a more acute presentation, the absence of tonsillar hypertrophy or exudates, the frequent occurrence of rash, and the presence of oral ulcerations.1 Herpes simplex infections, which typically affect young adults, also cause pharyngitis and are signaled by the presence of painful vesicles with erythematous bases. Ulcers may be present on the pharynx, lips, tongue, gums, and buccal mucosa. Pharyngeal erythema and exudate, fever, and tender lymphadenopathy are common for 1 to 2 weeks. In an immunocompromised host, large painful ulcers may be present. Herpes pharyngitis can be caused by primary infection or reactivation. Concomitant bacterial superinfection may occur.1,2 GABHS pharyngitis is primarily a disease of children 5 to 15 years old and, in temperate climates, occurs in winter and early spring.1,2 It is responsible for less than 15% of cases of pharyngitis in patients older than 15 years and is rare in patients younger than 3 years. In epidemics, the incidence may double.1,2 GABHS pharyngitis is associated with sudden-onset sore throat, temperature over 38.3° C (101° F), tonsillar erythema and exudates, palatal and uvular petechiae, uvular edema and erythema, and tender anterior cervical lymphadenopathy. Headache, nausea, vomiting, and abdominal pain may be present, especially in children. Cough, rhinorrhea, coryza, and other viral symptoms are usually absent. GABHS pharyngitis associated with a fine sandpaper erythematous rash that subsequently desquamates is termed scarlet fever. These findings, however, cannot be used to reliably diagnose or exclude streptococcal pharyngitis. Patients with recent exposure to others at risk for GABHS pharyngitis or in whom it has been diagnosed are more likely to become infected.1,2,5–7 Non-GABHS species can cause pharyngitis indistinguishable from GABHS.1 Diphtheria is a potentially lethal cause of pharyngitis that is uncommon where adequate vaccinations are administered. U.S. serologic surveys indicate that a large percentage of adults and adolescents lack immunity to diphtheria toxin.1 After a 2- to 4-day incubation period, patients develop malaise, sore throat, fever, and dysphagia. Examination early in the disease process may reveal pharyngeal erythema and isolated spots of gray or white exudate that later coalesce to form a pseudomembrane. This gray-green pseudomembrane is usually well demarcated and covers the nares, tonsils, soft palate, pharyngeal mucosa, and occasionally the uvula. The membrane may extend to involve the larynx and tracheobronchial tree, leading to hoarseness, cough, stridor, and airway obstruction. Painful cervical lymphadenopathy may be found. Severe inflammation and edema can produce dysphonia and a characteristic “bull neck” appearance. Some strains of Corynebacterium diphtheriae produce a systemic toxin that may cause myocarditis, polyneuritis (at first autonomic and then peripheral), vascular collapse, diffuse focal organ necrosis, and death. Asymptomatic carriers may transmit the disease.1,2 Corynebacterium ulcerans is an animal pathogen passed on by consumption of raw milk that can produce infection indistinguishable from that caused by C. diphtheriae. Arcanobacterium haemolyticum typically affects the 10- to 30-year-old age group and can be indistinguishable from streptococcal pharyngitis. Most patients have an associated rash that may be scarlatiniform, urticarial, or erythema multiforme (occasionally, skin manifestations may be the only complaint). Patients report a moderately severe sore throat and are usually nontoxic and afebrile. A. haemolyticum may cause a membranous pharyngitis that strongly mimics diphtheria; it is also associated with chronic tonsillitis.1,2,5 Anaerobic pharyngitis, or Vincent’s angina, is characterized by superficial ulceration and necrosis that often results in the formation of a pseudomembrane. Foul-smelling breath, odynophagia, submandibular lymphadenopathy, and exudate are often present. Patients typically have poor oral hygiene.5 Gonococcal pharyngitis is a sexually transmitted disease that may occur independently of genital infection. Those at highest risk are persons who practice receptive oral sex, especially men who have sex with men (in whom the incidence is reported to be as high as 15%). Its severity is variable, and it may result in an exudative or nonexudative pharyngitis. These differing manifestations occur after a lack of symptoms during the latent period of infection. Asymptomatic carriers are described, as is chronic and recurrent pharyngitis. Gonococcal pharyngitis is an important source of gonococcemia.1,2,5 Syphilitic pharyngitis is a manifestation of primary or late (tertiary) syphilis and manifests with painless mucosal lesions. Chlamydia trachomatis pharyngitis is a sexually transmitted disease that manifests similarly to gonococcal pharyngitis and is associated with orogenital sex. Urogenital culturing is necessary, along with treatment of sexual contacts. Patients are usually asymptomatic or may have only mild symptoms.1,2 M. pneumoniae infection usually causes a mild pharyngitis. Mycoplasma infection occurs in epidemics and in crowded conditions and can be responsible for approximately 10% of cases of adult pharyngitis. Pharyngeal and tonsillar exudates, cervical lymphadenopathy, and hoarseness are common. Lower respiratory tract infection may also be present.1,2,5 C. pneumoniae pharyngitis resembles M. pneumoniae pharyngitis. It also occurs in epidemics or crowded conditions. Severe pharyngitis with laryngitis is suggestive of C. pneumoniae infection. Swelling and pain in the deep cervical lymph nodes may be prominent. Lower respiratory tract and concomitant sinusitis occur. The hallmarks of chlamydial pharyngitis are recurrence and persistence.2,5 Monospot test results may be negative in up to 10% of patients with infectious mononucleosis, especially in the early stages of the illness. Immunoglobulin M (IgM) antibodies to EBV capsid antigen develop in 100% of cases. EBV nuclear antigens develop within 3 to 6 weeks and are useful if an initially negative test result becomes positive at a later date. Peripheral blood smears demonstrate atypical mononuclear cells in 75% of patients, with the peak incidence occurring in the second to third week of illness.1,2 Herpes pharyngitis may be diagnosed by culture, cytopathologic tests on scrapings of lesions, and serologic tests. Enzyme-linked immunosorbent assay testing for HIV can be falsely negative during the first 3 to 4 weeks of illness. During this period of time, quantitative assays for plasma RNA should be performed.1 Diagnosis of GABHS infection is important to prevent complications, particularly rheumatic fever. Even the most experienced practitioner has difficulty clinically diagnosing streptococcal pharyngitis.1,2,5–7 Several authors have proposed scoring systems based on clinical findings,2,5–7 but the only valid method of determining acute GABHS infection is by use of acute and convalescent antistreptolysin O titers, which is not practical in the emergency department (ED). A single throat culture has a sensitivity of 90 to 95% in detecting Streptococcus pyogenes in the pharynx. Variables that affect the accuracy of throat cultures include collection and culturing technique, as well as the recent use of antibiotics.1,5–7 Rapid diagnostic tests for GABHS detect streptococcal antigens. Rapid streptococcal tests (RSTs) have a reported specificity and sensitivity of up to 95%. Sensitivity and specificity in actual practice are lower than in controlled trials.1 Patients with positive cultures or RSTs may actually be carriers who may not need treatment and are at low risk for transmission and complications. The use of RSTs in patients without clinical findings consistent with GABHS increases false-positive results. A positive RST result seems to reliably indicate the presence of S. pyogenes in the pharynx. In contrast, RST results are often negative in the setting of pharyngitis with a low bacterial count (these patients are still at risk for complications, including rheumatic fever). It is recommended that a negative RST result in a child be followed by a confirmatory culture.1,5–7 Diagnosis and treatment of GABHS in adults is controversial and the subject of two expert panel recommendations.5–7 Use of clinical criteria in conjunction with RSTs improves the accuracy of RSTs, adults with negative RST results do not require confirmatory cultures (because of the lower incidence of GABHS infection and the extremely low risk for complications), and neither testing nor antibiotic treatment should be used in patients who are clinically at low risk for GABHS infection. Both panels agree that the most useful clinical criteria for determining GABHS pharyngitis are the Centor criteria (Box 75-1).5–7 The position of the Infectious Diseases Society of America is that a positive throat culture or RST result, in addition to clinical symptoms and signs, is needed to confirm the diagnosis of GABHS pharyngitis. The society stresses that clinical criteria alone are appropriate to determine which patients do not need testing but are insufficient, without bacterial confirmation, to diagnose GABHS pharyngitis.1 We recommend that any patient with none or only one Centor criterion not be tested or treated; patients with all four criteria should be treated without testing. Patients with two or three criteria should undergo RSTs and should be treated only if they have positive results (Box 75-2).6,7 These recommendations apply only to immunocompetent patients with no underlying comorbid conditions or a history of rheumatic fever. They do not apply in settings of outbreaks of GABHS infection or rheumatic fever, nor are they appropriate in situations in which the endemic rate of rheumatic fever is higher than that in the United States. It is important to consider local epidemics and be prepared to revise the approach to treatment if evidence of GABHS infection or complications exists.5–7 Non–group A streptococcal pharyngitis should also be treated, because the same suppurative complications occur as with group A streptococcal pharyngitis. Pharyngitis caused by other treatable organisms should also be considered and is associated with serious complications.1,2,6 Confirmation of diphtheria requires culturing on the proper media and immunologic testing (polymerase chain reaction). Toxigenicity testing should also be performed.1,2 The diagnosis of A. haemolyticum infection should be considered if rash, including erythema multiforme, accompanies pharyngitis. The diagnosis of Vincent’s angina is based on clinical findings and Gram’s stain. In cases of possible gonococcal infection, a sample should be plated on Thayer-Martin agar. Tuberculous pharyngitis is diagnosed by acid-fast staining. Syphilitic pharyngitis is diagnosed with darkfield microscopy, direct immunofluorescence, and serologic testing. Candidal pharyngitis is diagnosed by noting yeast on potassium hydroxide preparations of throat swabs or Sabouraud’s agar.2 The diagnosis of mycoplasmal pharyngitis can be confirmed serologically or by culture. Rapid antigen tests for Mycoplasma are available. Chlamydial pharyngitis can be diagnosed by serologic testing, by culture, or by antigen detection tests. Studies of patients with chronic pharyngitis find that surface cultures do not correlate well with the causal pathogens, which are often concealed within the tonsillar crypts. The differential diagnosis of adult pharyngitis includes deep space infections, tumors, foreign bodies, pemphigus, Stevens-Johnson syndrome, drug reactions, allergic reactions, uvulitis, angioneuropathic edema, chemical and thermal burns, esophagitis, gastroesophageal reflux disease, cricoarytenoid arthritis, thyroiditis, and epiglottitis.7 Patients with pharyngitis should be treated symptomatically with topical anesthetic rinses or lozenges and with acetaminophen or ibuprofen. Oral hydration and saltwater gargles are helpful. Most cases of pharyngitis are self-limited and follow a benign course.1,5–7 Antibiotics are overused in the treatment of pharyngitis and are not indicated in the vast majority of cases of pharyngitis diagnosed in the United States. Treatment of infectious mononucleosis is supportive (see Chapter 130). Patients should avoid contact sports for 6 to 8 weeks to minimize the small risk of splenic rupture. Corticosteroids are indicated for patients with tonsillar hypertrophy that threatens airway patency, severe thrombocytopenia, or hemolytic anemia.1,2 Acyclovir, valacyclovir, or famciclovir is indicated in immunocompromised patients with herpetic pharyngitis and may be beneficial in the treatment of acute herpetic pharyngitis.1,7 The use of antiretrovirals is indicated in acute HIV infection.1 GABHS pharyngitis in children and adolescents should be treated adequately (within 9 days) to prevent rheumatic fever. The incidence of rheumatic fever parallels that of GABHS and has markedly diminished with the use of antibiotics. Patients with mild cases of GABHS pharyngitis may develop rheumatic fever. Rheumatic fever complicates 0.3% of cases of GABHS pharyngitis, but in epidemics the incidence increases to 3%. More troubling is an increase in sporadic outbreaks of rheumatic fever.5–7 The incidence and course of poststreptococcal glomerulonephritis caused by nephritogenic strains are unaffected by antibiotic therapy.1,5–7 Antibiotic therapy is extremely effective in eradicating GABHS and its other complications. Untreated, GABHS pharyngitis is a self-limited illness that lasts 3 to 4 days. Early antibiotic treatment of streptococcal pharyngitis leads to a 13% earlier resolution of symptoms and shortens the course of illness by about 1 day. Antibiotic therapy also decreases transmission, and patients are no longer infectious after 24 hours of antibiotic treatment.1,5–8 The antibiotic regimen of choice for adults with GABHS pharyngitis is either a single intramuscular injection of 1.2 million units of benzathine penicillin or a 10-day course of penicillin V, 500 mg orally twice a day. Less frequent administration is less effective in preventing rheumatic fever.1,5–7 Intramuscular penicillin may be more effective than oral penicillin and ensures compliance, but allergic reactions are more severe as a result of procaine allergy, and treatment is more expensive. Penicillin failure usually reflects noncompliance, reinfection, or the presence of β-lactamase–producing organisms. Erythromycin or azithromycin is recommended for patients who are allergic to penicillin. A 1-g total daily dose of erythromycin is given for 10 days, but administration intervals of two, three, and four times a day are equally effective in preventing rheumatic fever. A 5-day course of azithromycin is recommended.1,5–7 Use of cephalosporins or clindamycin is also acceptable for penicillin-allergic patients. Once-daily amoxicillin therapy may be effective in children.1,9 Oral cephalosporins or clindamycin should be reserved for patients not responding to penicillin or unable to tolerate either penicillin or erythromycin.1,5,8,9 Patients whose symptoms return within a few weeks of treatment may have been noncompliant with oral therapy or may have acquired a new infection (at times from asymptomatic close contacts). Evaluation and treatment should be similar to that of the first episode, with consideration given to treatment with intramuscular penicillin. Further recurrences require more extensive evaluation. Pharyngeal cultures should be obtained and consideration given to evaluating and treating close contacts for GABHS infection.5 Although many studies focus on GABHS pharyngitis, proper treatment of nonstreptococcal pharyngitis can also prevent serious complications. Because clinical judgment is insufficient and rapid diagnostic tests are not always accurate and diagnose only GABHS, this disease process is often treated empirically. The choice of antibiotic for the empirical treatment of adult bacterial pharyngitis is not fully elucidated. It is unclear how effective antibiotics are in uncomplicated cases of non-GABHS pharyngitis in adults. Antibiotics may modestly shorten the course of the disease process, but they are also associated with increased recurrence, increased bacterial drug resistance, decreased immune response, and patient expectations for antibiotics with subsequent episodes of pharyngitis.1,5–8 Successful treatment of diphtheria is inversely related to the duration of disease. When diphtheria is strongly suspected on the basis of clinical findings, treatment is begun empirically. Airway collapse may occur suddenly and without warning. The mainstay of therapy is antitoxin (a horse serum product), which should be administered immediately on clinical suggestion of diphtheria. The dose of antitoxin varies widely and depends on the site of infection and the duration of symptoms. Antibiotics have little effect on the resolution of systemic toxicity, but they are useful in eradicating C. diphtheriae infection and preventing transmission. Infected patients should remain in strict isolation to prevent transmission. The antibiotic of choice is penicillin G for 5 days, followed by penicillin VK for 5 days, or erythromycin 500 mg four times a day for 10 days. A small percentage of patients require an additional 10-day course of erythromycin for persistent infection. Rifampin 600 mg/day for 10 days is also effective in eradicating the carrier state of C. diphtheriae and treating erythromycin-resistant diphtheria. Diphtheria toxoid should be administered during convalescence and to unvaccinated close contacts.1,2,10 A. haemolyticum may be resistant to penicillin. Erythromycin 250 mg orally four times a day for 10 days is the treatment of choice.1,2 Vincent’s angina is treated with penicillin or clindamycin and rinses with an oral oxidizing agent (hydrogen peroxide).10 Gonococcal pharyngitis is often more difficult to eradicate than genital infections. Treatment is similar to that for gonococcal urethritis and consists of ceftriaxone (125 mg intramuscularly) with concomitant treatment of chlamydial infection with a single oral dose of 1 g of azithromycin or a course of doxycycline 100 mg orally twice a day for 7 days.2,10 Oral fluoroquinolones are no longer recommended owing to the high prevalence of gonococcal resistance. Tuberculous pharyngitis is seen with disseminated disease. Patients should be isolated and treated with a multidrug regimen. Pharyngitis caused by primary syphilis is treated with 2.4 million units of benzathine penicillin (long acting), with 14 days of tetracycline or doxycycline used as an alternative. Candidal pharyngitis is treated with systemic fluconazole or itraconazole. Alternative therapy includes nystatin (suspension or tablets) or oral clotrimazole for 14 days. Chronic suppression therapy with ketoconazole, clotrimazole, or fluconazole is usually required for HIV pharyngitis.10 M. pneumoniae is treated with erythromycin, tetracycline, or doxycycline for 7 to 14 days.2,10 Chlamydial pharyngitis is treated with doxycycline, trimethoprim-sulfamethoxazole, or a macrolide antibiotic. C. pneumoniae pharyngitis should be treated for 7 to 10 days to prevent treatment failure and recurrence. C. trachomatis pharyngitis may require prolonged or repeated courses of antibiotics.2,10 Treatment of recurrent or chronic tonsillitis should include β-lactamase–resistant antibiotics active against aerobic and anaerobic organisms. Choices include oral cephalosporins, amoxicillin-clavulanic acid, penicillin with rifampin or metronidazole, and clindamycin.2 Steroids given in conjunction with oral antibiotics in adults with acute pharyngitis may significantly shorten the duration of symptoms and provide a greater degree of pain relief without increasing complications. Oral (40-60 mg of prednisone per day for 1-5 days) or intramuscular (a single dose of 10 mg of dexamethasone) administration is equally effective.11,12 Although most cases of pharyngitis follow a benign course, life-threatening complications can rarely occur. Airway compromise from tonsillar enlargement, local and distant spread of infection, deep neck abscesses, necrotizing fasciitis, sleep apnea, bacteremia, sepsis, and death have been reported but are very rare, given the prevalence of these diseases.1,5 Infectious mononucleosis may lead to hepatic dysfunction, splenic injury, neurologic disorders, pneumonitis, pericarditis, and hematologic disorders, including thrombocytopenia and hemolytic anemia.1,2 Complications of GABHS pharyngitis are both suppurative and nonsuppurative. Suppurative complications include peritonsillar abscess, deep space abscesses, suppurative cervical lymphadenitis, otitis media, sinusitis, mastoiditis, bacteremia, sepsis, osteomyelitis, empyema, meningitis, and soft tissue infections. Nonsuppurative complications include scarlet fever, rheumatic fever, poststreptococcal glomerulonephritis, nonrheumatic perimyocarditis, erythema nodosum, and streptococcal toxic shock syndrome. In contrast to rheumatic fever, other complications of GABHS pharyngitis are increasing in incidence and severity. A chronic carrier state of streptococcal infection exists and can persist for several months despite treatment. Affected patients are asymptomatic, at low risk for rheumatic fever, and not considered highly contagious. Non–group A streptococcal pharyngitis may be complicated by the same suppurative complications as group A infections. Scarlet fever and acute glomerulonephritis, but not rheumatic fever, are linked to group C and G pharyngitis.1,2,5–7 Lingual tonsillitis is a rarely diagnosed cause of pharyngitis that predominantly occurs in patients who have had palatine tonsils removed. The lingual tonsils are most commonly (size and location are highly variable) located symmetrically on either side of the midline just below the inferior pole of the palatine tonsils and anterior to the vallecula at the base of the tongue. This lymphoid tissue may enlarge after puberty, repeated infection, and tonsillectomy.13 Patients with lingual tonsillitis have a sore throat that worsens with movement of the tongue (including tongue depression) and phonation. The patient may have a classic “hot potato” voice (the muffled voice one has when eating very hot food) and report feeling a swelling in the throat. Dysphagia, fever, respiratory distress, and stridor may be present. Chronic or recurrent lingual tonsillitis may also cause a chronic cough or sleep apnea. Physical findings often include a normal-appearing pharynx with mild hyperemia. Direct or indirect laryngoscopy reveals an edematous lingual tonsil covered with a purulent exudate. Lateral soft tissue neck films aid in the diagnosis. These films demonstrate a normal-appearing epiglottis and arytenoepiglottic folds, with a scalloped appearance on the anterior surface of the vallecula caused by an enlarged lingual tonsil (Fig. 75-2).13 Management includes maintenance of airway patency, antibiotics, and supportive therapy. Rarely, acute lingual tonsillitis may be a life-threatening condition. Airway management includes warmed humidified oxygen, hydration, and corticosteroids. Nebulized epinephrine can relieve the acute respiratory distress and stridor. Antibiotics of choice are similar to those used for the treatment of pharyngitis.13 Laryngitis is manifested as hoarseness and aphonia. It is usually caused by viral upper respiratory tract infections. In up to 10% of cases, bacterial infections (including streptococcal infection and diphtheria) may be responsible. Rare causative entities are tuberculosis, syphilis, leprosy, actinomycosis, and fungi. Noninfectious causes include tumors, caustic or thermal injuries, trauma, and esophageal reflux disease.14,15 Laryngitis generally is a benign viral illness with peak symptoms lasting 3 to 4 days. “Voice rest” often is recommended, but there is no evidence that this is of any benefit in terms of duration or severity of symptoms. Antibiotics are not indicated unless signs of bacterial infection are present.16 Steroids may hasten resolution of symptoms.17
Upper Respiratory Tract Infections
Pharyngitis (Tonsillopharyngitis)
Etiology
Clinical Features
Diagnostic Strategies
Differential Diagnosis Considerations
Management
Disposition
Lingual Tonsillitis
Laryngitis
Upper Respiratory Tract Infections
Only gold members can continue reading. Log In or Register to continue

Full access? Get Clinical Tree