1. The musculoskeletal system is derived from the paraxial mesoderm which begins to differentiate into somites during the 4th week of development.
2. Many common pediatric syndromes and associations such as VACTERL Association (Vertebral anomalies, Anal atresia, Cardiac defects, Tracheoesophageal fistula and/or Esophageal atresia, Renal & Radial anomalies, and Limb defects) have associated vertebral and long-bone congenital abnormalities.
3. Scoliosis surgery may be done in young children as well as in adolescents. Up to 10% of children treated for congenital heart disease in infancy will go on to develop scoliosis as adolescents.
4. Patients undergoing orthopedic surgery may be at higher risk for developing venous air embolism (scoliosis surgery) and fat embolism (long-bone repairs) compared to nonorthopedic surgery.
5. Postoperative neurovascular checks are extremely important. Many procedures put the patients at risk for late neurologic sequelae due to poor circulation or edema around nerves or in closed spaces (compartment syndromes).
6. Positioning of patients especially in the prone position is critical to avoid skin breakdowns and postoperative nerve damage including ocular blindness.
DEVELOPMENT OF THE MUSCULOSKELETAL SYSTEM
MUSCLE, CARTILAGE, AND BONE
Embryonic mesoderm differentiates into three distinct regions of paraxial, intermediate, and lateral mesoderm (see Chapter 15). The paraxial mesoderm begins to segment into somites at approximately the 4th week of development. With subsequent differentiation into sclerotomes and dermomyotomes, the stage is set for the dermomyotome to now further differentiate into myotomes and dermatomes. Mesenchymal cells within the myotome then differentiate into myoblasts; with amazing concurrence, myoblasts are also formed from mesenchyme in the anterolateral body walls and in the limb buds. Fetal muscle forms from additional myoblasts as well as hypertrophy of existing myoblasts. These myoblasts organize into muscle fibers and become arranged in groups surrounded by connective tissue, also derived from local mesenchyme (Table 26.1).
Cartilage begins its formation by about the 5th week of development from local mesenchyme. These cells enlarge locally, take on the shape of the future cartilage, become rounded, and secrete a granular intercellular matrix which envelops the developing cartilage cells. They also become covered by a fibrous perichondrium, growing by interstitial (interior of the cartilage) as well as appositional growth (interior of the perichondrium, chondroblasts) within this cartilage “sandwich.”
Bone is formed by intramembranous and enchondral ossification. Intramembranous ossification proceeds from mesenchymal cells in close approximation, lying parallel to each other, with collagenous fibers elaborated such that the area resembles a fibrous membrane. Some of these mesenchymal cells differentiate into osteoblasts, continuing to produce collagenous material which fills the intercellular spaces (osteoid) which is, as yet, devoid of calcium salts, and therefore still membranous in appearance. With the secretion of alkaline phosphatase, calcium salts become deposited, forming bone matrix. Ongoing matrix formation promotes the development of some osteoblasts into osteocytes, and eventually bone spicules with a center of ossification are formed. Vascular mesenchymal ingrowth results in the formation of a periosteum external to the developing bone. The inner cellular layer of the periosteum differentiates into osteoblasts, which lay down additional parallel plates of compact bone (lamellae) locally. Bone continues to remodel itself during growth and development, and another category of cell, the osteoclast, is responsible for resorption. Osteoclasts may be formed either from osteoblasts or from more primitive mesenchymal cells. The process of bone creation and bone resorption therefore takes place concurrently.
TABLE 26.1 Embryology of regional muscle development
Region | Embryology | Comments |
Head | Mesenchyme of the pharyngeal arches |
|
Tongue | Occipital myotomes | Hypoglossal nerve grows to meet these developing myotomes |
Extrinsic ocular muscles | Preotic myotomes | Innervated by the third, fourth, and sixth cranial nerves |
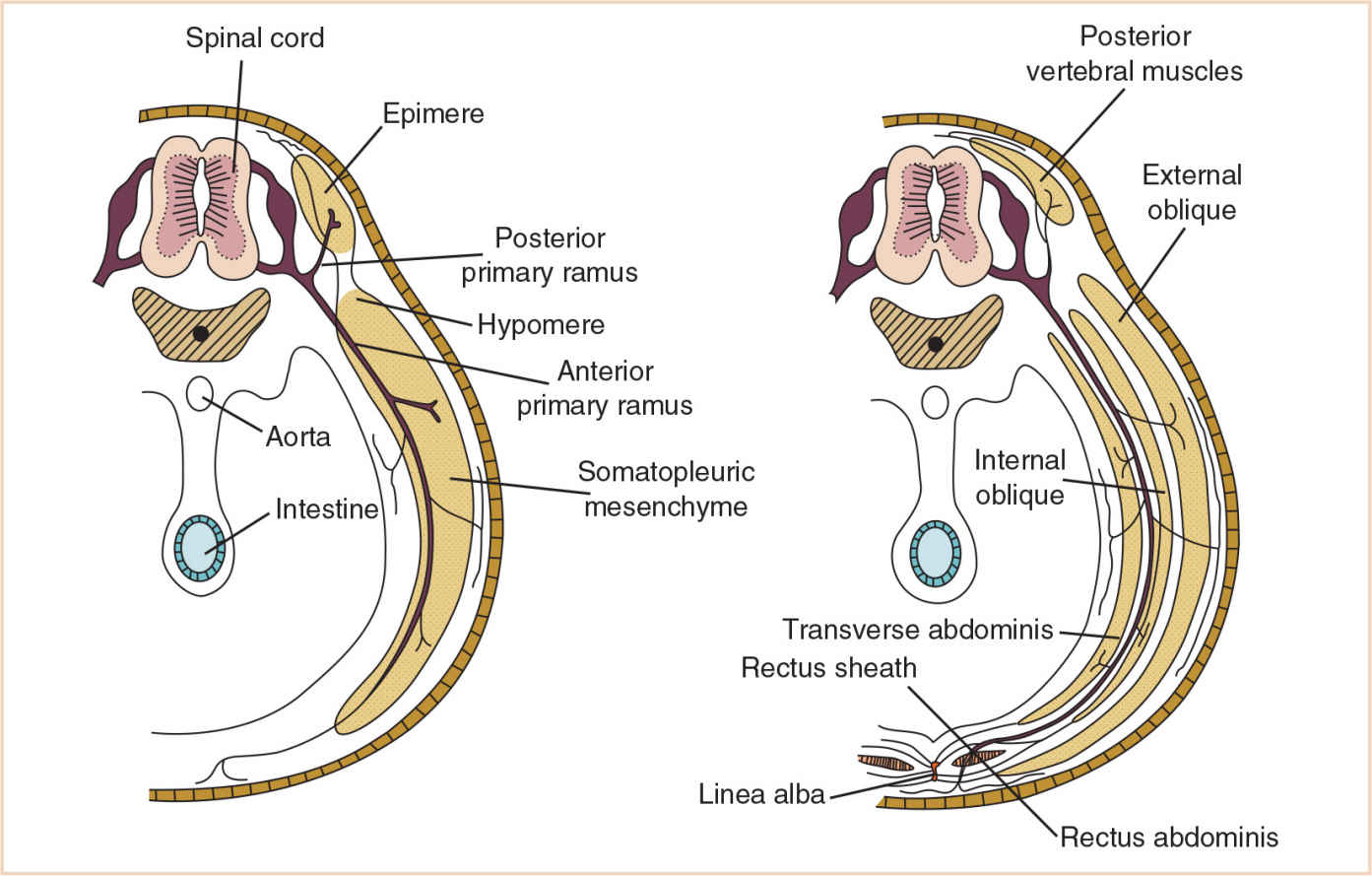
Neck and trunk | Division of segmental myotomes into dorsal epimere and ventral hypomere | Each segmental spinal nerve divides into a posterior primary ramus (innervating the epimere) and an anterior primary ramus (innervating the hypomere). The epimeres develop into the vertebral column extensors, while the hypomeres form the prevertebral flexors. The scalene and prevertebral muscles in the neck form from hypomeres. |
Thorax | Anterolateral somatopleuric mesenchyme | Splits into three layers; in the thorax this forms the external, internal, and transverse intercostal muscles |
Abdomen | Anterolateral somatopleuric mesenchyme | Splits into three layers; in the abdomen this forms the external oblique, internal oblique, and transversus abdominis muscles |
Diaphragm | Third, fourth, and fifth cervical myotomes | These myotomes fuse and migrate caudally into the thorax as the septum transversum |
Pelvis | Sacral and coccygeal hypomeres and local mesenchyme | Levator ani, coccygeus muscles, and the external anal sphincter and urethral sphincter |
Muscles of the limbs (appendicular skeleton) | Hypomeres and local body wall mesenchyme of the regions from which the limb buds develop |
|
Enchondral ossification is responsible for long-bone formation in developing limbs. Initial modeling with mesenchyme is replaced by hyaline cartilage. Ossification begins in the middle of this cartilage by the 8th week of development, and calcium salts are once again deposited within the intercellular matrix. Cartilage cells subsequently involute, and empty spaces remain. At the same time, the perichondrium develops into periosteum, with its inner layer of cells differentiating into osteoblasts, once again depositing bone locally around the perimeter of the hollowing shaft, strengthening the developing bone. Spongy bone eventually develops in the center of the hollowing shaft as a result of bone resorption by osteoclasts and bone deposition by osteoblasts. This marrow cavity will eventually be occupied by bone marrow. At birth, the limbs have a shaft (diaphysis) with two cartilaginous ends (epiphyses). Following birth, secondary centers of ossification appear in each epiphysis; for now, new bone formation takes place in all three areas. Longitudinal limb growth eventually ceases when the epiphyseal cartilage chondrocytes no longer divide, and the epiphyseal plates at this point will fuse with the diaphysis, for the most part, by 20 years of age.
Skull: See Chapter 14.
APPENDICULAR SKELETON
Limb buds appear during the 6th week of development as a result of localized proliferation of somatopleuric mesenchyme (Fig. 26.1). Overlying ectoderm bulges from the two pairs of trunk locations and appears as flattened paddles of tissue. These ectodermal cells differentiate into the apical ectodermal ridge (AER) which signals the underlying mesoderm. A zone of polarizing activity (ZPA) lies beneath the AER and determines the pattern of digit formation. Development proceeds in a craniocaudal manner and therefore the arm buds appear first, emerging from the lower cervical and upper thoracic segments; leg buds emerge from the lower four lumbar and upper three sacral segments. These buds have a cephalic preaxial border and a caudal postaxial border. The lower five cervical nerves innervate the preaxial border of the upper buds while the postaxial border is innervated by the eighth cervical and first thoracic nerves. The lower limb buds’ preaxial border is innervated by the second lumbar to first sacral nerves and the postaxial border by the first to third sacral nerves. Again, because of differential growth (cranial being more rapid than caudal), the limbs “appear” to move caudally. The area between bones is known as the interzone and mesenchyme surrounds the articular surfaces of bones which then differentiate to become the synovial cavity of the joint (Fig. 26.2).
The limb buds of the lower extremity arise a few days after the upper limb bud arises. The lower buds develop from somites from the lumbar and sacral regions and consist of a mesenchymal core with an outer covering of ectodermal cells. These ectodermal cells differentiate into the AER which signals the underlying mesoderm. An ZPA lies beneath the AER and determines the pattern of digit formation. As the limb bud grows, the mesenchymal layer differentiates into deep, intermediate, and superficial layers and the deep layer develops into the skeleton. The area between bones is known as the interzone, and mesenchyme surrounds the articulating surfaces of bones which then differentiate to become the synovial cavity of the joint. Limb plates develop at the distal end of the limb buds which differentiate into the foot. Five bands of mesenchyme condense and become the digital rays. These chondrify and become the long bones of the feet. Apoptosis of loose mesenchyme at the distal end of the limb plates leaves interdigital clefts and defines the toes (Fig. 26.3).
The lower limb muscles are derived from mesenchyme from somites which migrate into the limb bud along with nerve and blood supply. Muscles tend to differentiate into flexor and extensor muscles which are innervated by nerves which are derived from nerve fibers from the ventral rami of L2–S3 spinal nerves. These nerve fibers form a plexus with the anterior division of the plexus innervating the developing flexor muscle mass and the posterior division innervating the developing extensor muscle mass. The dermis is derived from the superficial layer of the mesenchyme and the epidermis is derived from the ectoderm of the limb bud.
The limb buds develop by lateral extension with the flexor mesenchyme located anteriorly and the big toe facing superiorly. The limb then changes from a lateral position opposite the lumbar and sacral vertebrae to a nearly sagittal position. The limb then rotates medially 90 degrees so that the flexor surface of the knee joint is positioned posteriorly and the muscle extensors are positioned anteriorly.
Limb plates develop at the distal end of the limb buds which differentiate into the hands and feet. Five bands of mesenchyme condense and become the digital rays, which chondrify and become long bones. Apoptosis of loose mesenchyme at the distal end of the limb plates leaves interdigital clefts and defines the fingers and toes (Fig. 26.3).
VERTEBRAL COLUMN
Each body somite differentiates into a ventromedial sclerotome and a dorsolateral dermomyotome (Fig. 26.4). The sclerotome consists of rapidly dividing, loosely arranged mesenchymal cells that migrate medially during the fourth week of fetal life and surround the notochord, encasing it in a continuous cylinder of mesenchyme. The caudal half of each sclerotome fuses with the cephalic half of the succeeding sclerotome to form the mesenchymal vertebral body, making each vertebral body an intersegmental structure. Although the notochord degenerates in the region of the vertebral body, in the intervertebral region it enlarges to form the nucleus pulposus of the intervertebral disc. The annulus fibrosus, the surrounding fibrous cartilage, is derived from the remaining intervertebral mesenchyme (Fig. 26.5).
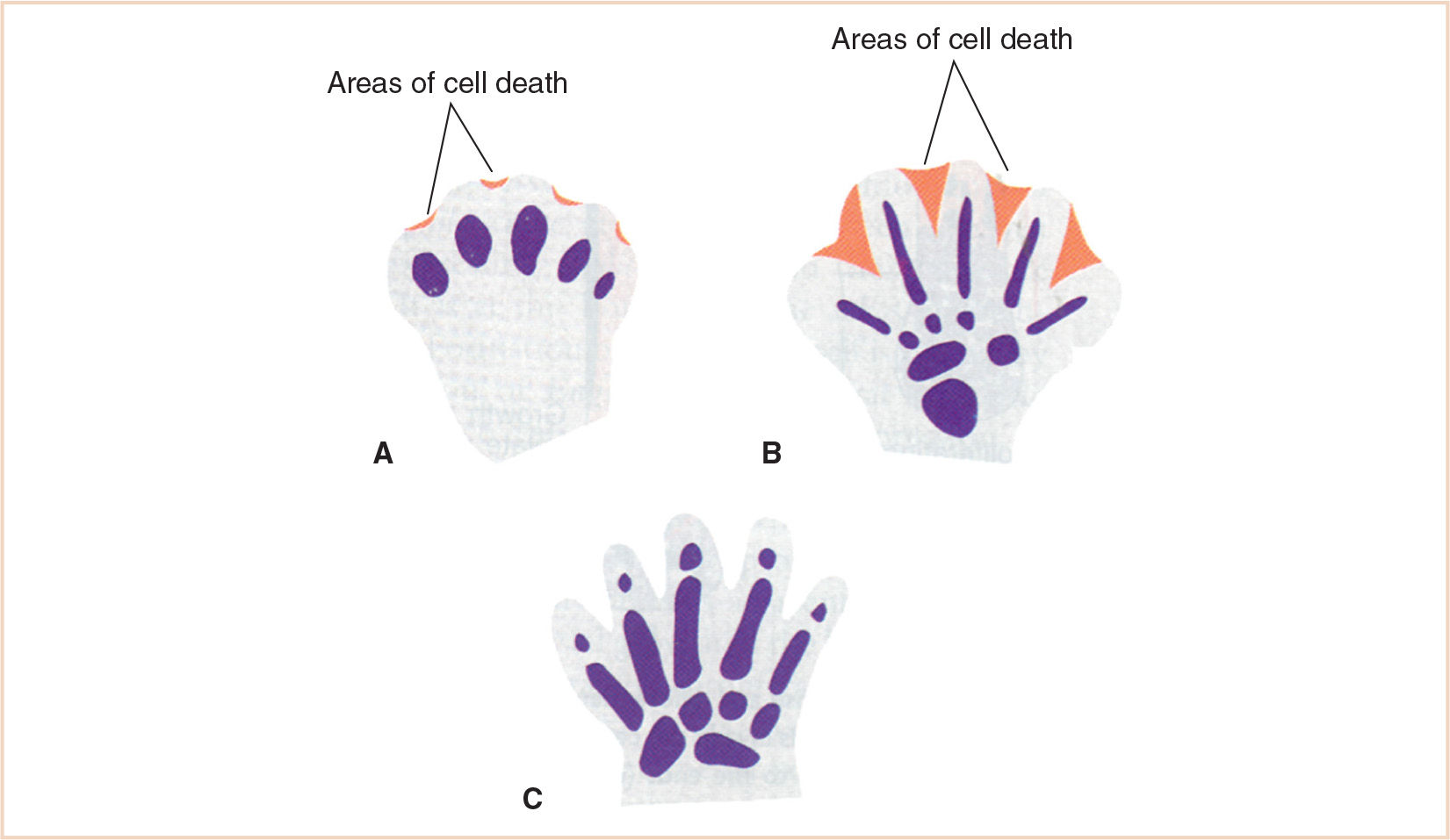
FIGURE 26.3 Development of digital separation. A: Cell death occurring at the apical ectodermal ridge beginning to define the digit. B: Cell death further defining the interdigital space. C: Completion of digital separation.
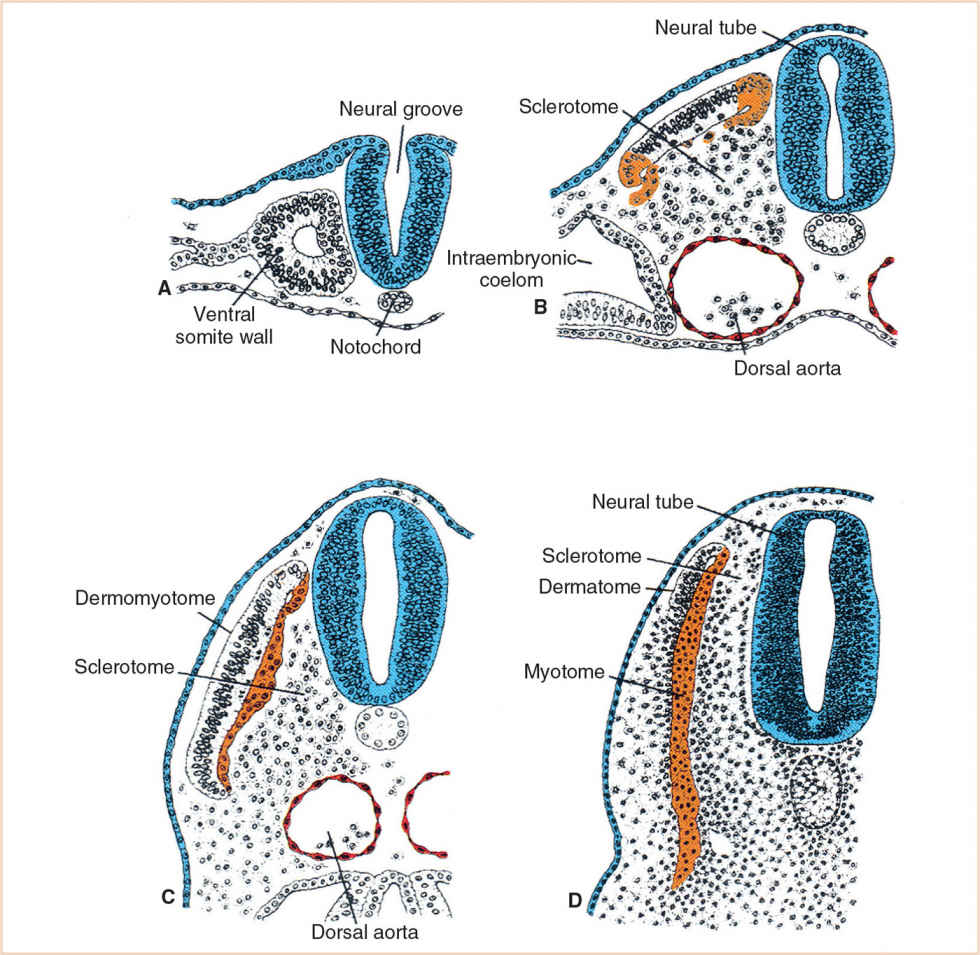
FIGURE 26.4 Each body somite (A) differentiates into a ventromedial sclerotome (B) and a dorsolateral dermomyotome (C) which then further developes into the myotome and dermatome (D).
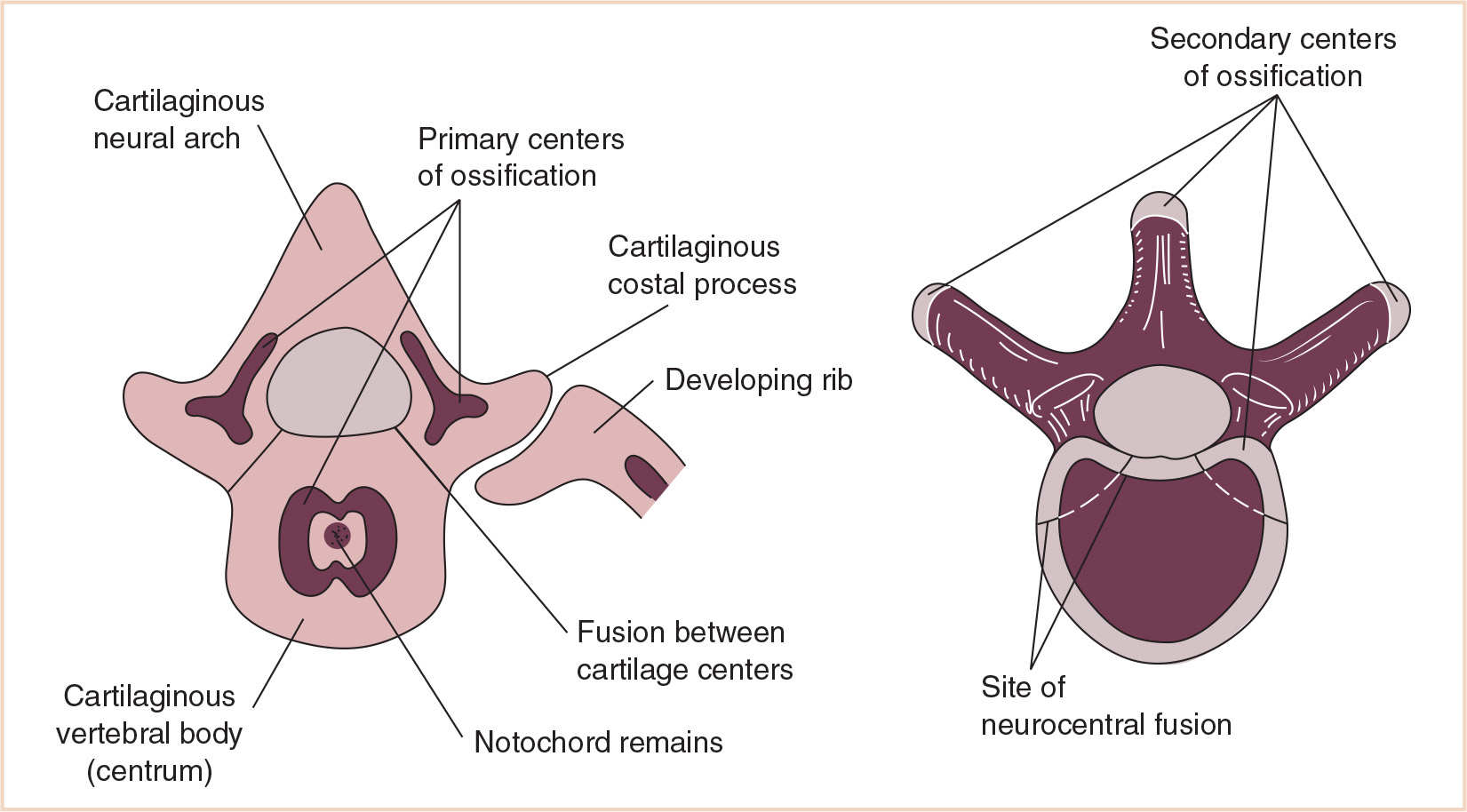
This mesenchymal vertebral body gives rise to dorsal and lateral outgrowths. The dorsal outgrowths grow around the neural tube and fuse to form the neural arch. The lateral outgrowths grow between myotomes to form the costal processes that will eventually form the ribs (Table 26.2). Chondrification centers appear in the middle of the mesenchymal vertebral bodies, forming a cartilaginous centrum. Chondrification centers form in each half of the neural arch and spread dorsally to fuse behind the neural tube. They also extend forward to fuse with the centrum and laterally into the costal processes, forming a cartilaginous vertebra. The fate of the costal processes varies with its location in the vertebral column.
Developmentally, at approximately the ninth week, primary ossification centers appear, two for each centrum and one for each half of the neural arch. The two centers for the centrum usually unite quickly, but complete union of all the primary centers does not occur until several years of age. During adolescence, secondary centers appear in the cartilage covering the superior and inferior ends of the vertebral body and the epiphyseal plates are formed. A secondary center also appears at the tip of each transverse process and at the tip of the spinous process, completing their eventual fusion with the rest of the vertebrae at approximately 25 years of age (Fig. 26.6).
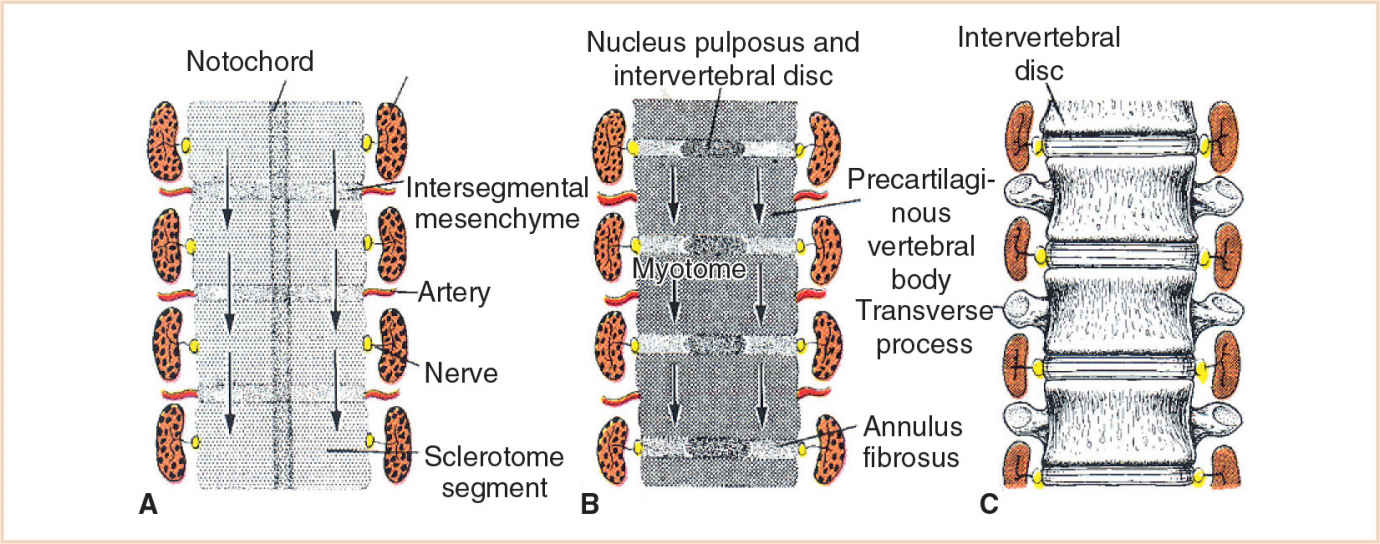
FIGURE 26.5 Development of the spine. A: At week 4, sclerotomes are separated by lower-density intersegmental tissue. B: The caudal half of each sclerotome fuses with the cephalic half of the succeeding sclerotome. C: Each vertebral body is therefore an intersegmental structure.
It is ironic that the wonderfully strong spinal column is actually a composite of many centers of migration, chondrification, and ossification, and not surprising that abnormalities of curve, slippage, and herniation may result, particularly in relation to unbalanced growth episodes during development.
DISORDERS OF THE UPPER EXTREMITIES
Disorders of the upper extremity are varied in morphology and severity. In 1976, the International Federation for Societies for Surgery of the Hand introduced a classification system based on known etiopathogenic pathways (Table 26.3). In 2002, the system was modified based on practical considerations and an improved understanding of the embryology and genetics. Even with these improvements, a recent study still found classification difficult in 6.6% of the cases and impossible 7.8% of the time (1). Of particular interest to anesthesiologists is that 26% of individuals with congenital anomalies of the hand had other associated anomalies (1,2).
DISORDER: Polydactyly
CLINICAL PEARL Many children with congenital bony abnormalities will have abnormal airways. Since it is often difficult to do an adequate airway assessment on uncooperative children, it is important to have several methods available to secure the airway safely. Likewise, many of these children need several procedures; so it is important to document carefully maneuvers used to secure their airways.
TABLE 26.2 Fate of the costal processes
Cervical | Processes remain short, forming the lateral and anterior boundaries of the foramen transversarium |
Thoracic | Each process forms a cartilaginous rib |
Lumbar | Forms part of the transverse process |
Sacral | Costal processes fuse, forming the lateral mass of the sacrum |
BACKGROUND: Polydactyly is the presence of additional fingers or toes. It can range from a simple soft tissue pedicle to a multiphalangeal digit, complete with an extra metacarpal (Fig. 26.7). Although generally not associated with other anomalies, polydactyly may occur as part of a syndrome. These patients need to be carefully evaluated for associated anomalies (Table 26.4).
EMBRYOLOGY/ANATOMY
The extra digit may consist of soft tissue only or have extensive bony involvement. Polydactyly can be pre– or postaxial. Determined by the fifth week of gestational age, the proximo-distal axis runs from the origin of the limb at the body wall to its tip. The cranial border of the limb is preaxial and the caudal border postaxial. Preaxial polydactyly is a result of a defect in sonic hedgehog (Shh) expression. Normal expression occurs specifically in the ZPA arising from mesenchyme located postaxially in the limb bud. With transplantation of a ZPA onto a preaxial border, duplication of distal elements with reversed orientation occurs.
TABLE 26.3 Hand anomaly classification
| Category | Comment |
I | Failure of formation A—Transverse B—Longitudinal | This group should include radial and ulnar deficiencies limited to the hand, without forearm deficiencies. |
II | Failure of differentiation | This is the largest group, with 513 anomalies. Congenital trigger finger and thumb is included in this group. |
III | Polydactyly | Triphalangeal thumbs are generally included in this group if it is considered a duplication in length. |
IV | Overgrowth |
|
V | Undergrowth |
|
VI | Amniotic band syndrome |
|
VII | Generalized skeletal syndromes | Failure of finger ray induction. |
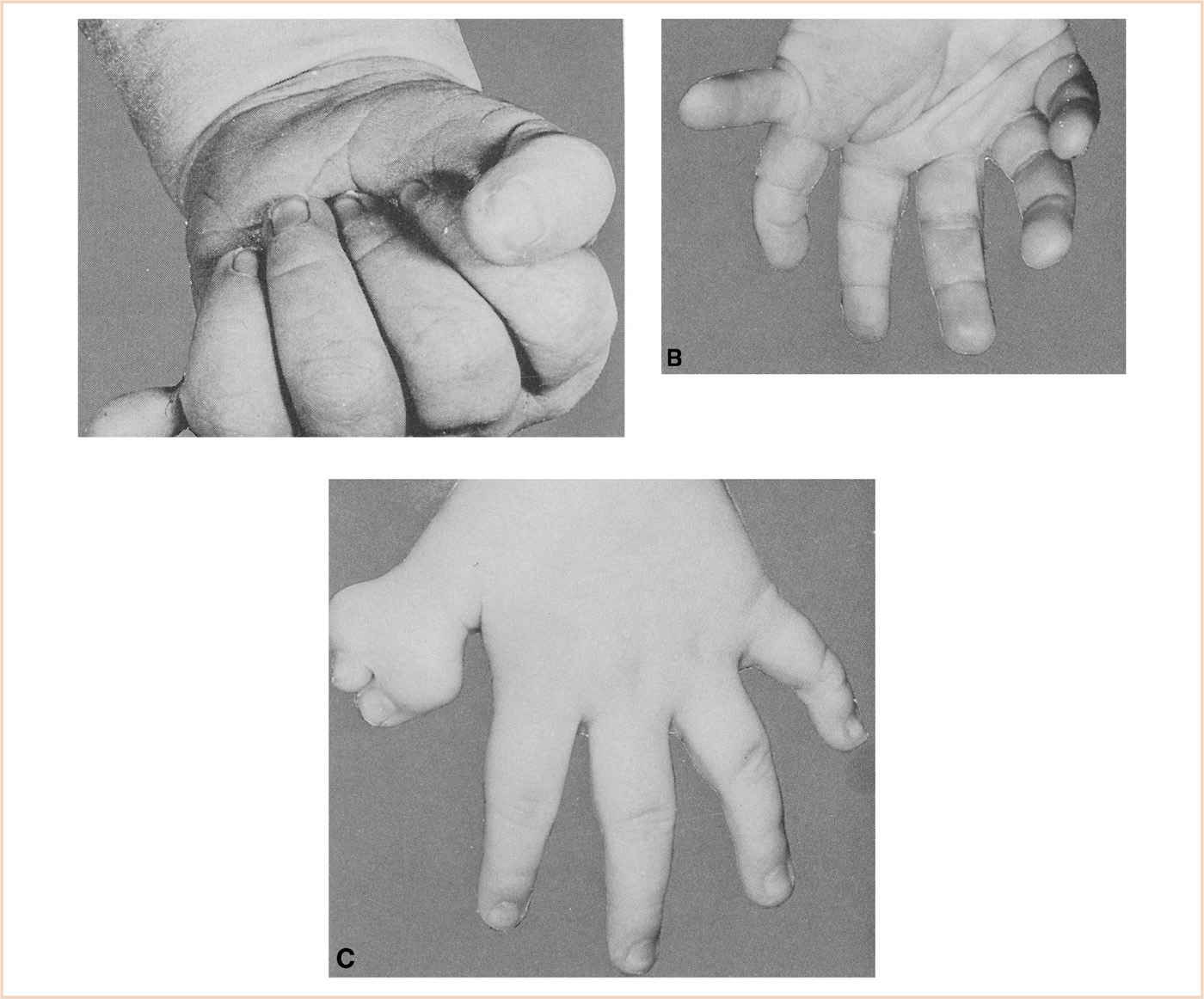
FIGURE 26.7 A: A supernumerary digit amenable to treatment by simple ligation. B: A supernumerary thumb and metacarpal. C: A bifid distal phalanx of the thumb.
TABLE 26.4 Polydactyly syndromes
Syndrome | Inheritance | Nonorthopedic features |
Ellis–van Creveld syndrome | Autosomal recessive | Dwarfism, cardiac anomalies, dental abnormalities, short trachea, restrictive lung disease |
Carpenter syndrome | Autosomal recessive | Craniosynostosis, acrocephaly, mandibular hypoplasia, short neck, cardiac anomalies, elevated intracranial pressure |
Trisomy 13 (Patau syndrome) |
| Multiple craniofacial, cardiac, neurologic and renal anomalies, apneic episodes, short neck, small mouth, micrognathia |
Rubinstein–Taybi syndrome | Autosomal dominant | Mental retardation, craniofacial abnormalities, choanal atresia, obstructive sleep apnea, recurrent respiratory infections, cardiac anomalies |
Smith–Lemli–Opitz syndrome | Autosomal recessive | Enzymatic defect of 7-dehydrocholesterol reductase, micrognathia, feeding problems, GERD, pulmonary hypoplasia, single-lobed lungs, and cardiac anomalies |
Bardet–Biedl syndrome | Autosomal recessive | Mental retardation, obesity, renal insufficiency |
Jeune syndrome (asphyxiating thoracic dystrophy or thoracic-pelvic-phalangeal dystrophy) | Autosomal recessive | Severe thoracic hypoplasia with restrictive lung disease and/or chronic hypoxemia, renal failure, cardiac failure, cirrhosis |
PHYSIOLOGIC CONSIDERATIONS
1. Major body system physiology is not typically affected by isolated limb deformities that are not associated with a syndrome.
2. Tourniquets may be used for more extensive corrections. Increases in blood pressure and heart rate should be treated with short-acting agents. The duration of tourniquet inflation should be carefully monitored. In the absence of associated syndromes, there is infrequently significant pathophysiology.
SURGICAL REPAIR
1. Small incomplete soft tissue pedicles are ligated in the newborn period.
2. When the excision involves an entire digit or a portion of a digit the surgical decision needs to be based on function as well as aesthetics. For this reason, surgery may be delayed for several years until this determination can be made.
DISORDER: Syndactyly
CLINICAL PEARL Intraoperative tourniquets can decrease blood loss but can also lead to permanent limb damage secondary to ischemia. It is important to limit the duration of tourniquet time.
BACKGROUND
Syndactyly or webbing of the fingers occurs in approximately 1 in 2,500 births with a male predominance; most often the space between the middle and ring finger is affected (3). However, it can range from a simple web of skin connecting the two otherwise normal fingers to complex fusion of multiple digits involving not only skin but the soft tissue, bones, and joints. Syndactyly may be an isolated anomaly or part of a syndrome (Fig. 26.8 and Table 26.5). Nonsyndromic syndactyly can be associated with prolonged Q and T waves (QT) syndrome. These patients need to be carefully evaluated for associated anomalies and should have a preoperative electrocardiogram (ECG) prior to their initial surgery.
EMBRYOLOGY/ANATOMY
The presence of webbed or fused digits is due to incomplete apoptosis (Fig. 26.3). Ideally, this process of programmed cell death results in the destruction of unnecessary developmental tissue and the sculpting of individual digits. The mechanism of this process and its failure has not yet been clearly identified; however it may be related to impaired lysosomal enzyme release. This could be why steroids, which stabilize lysosomal enzyme membranes, might produce syndactyly.
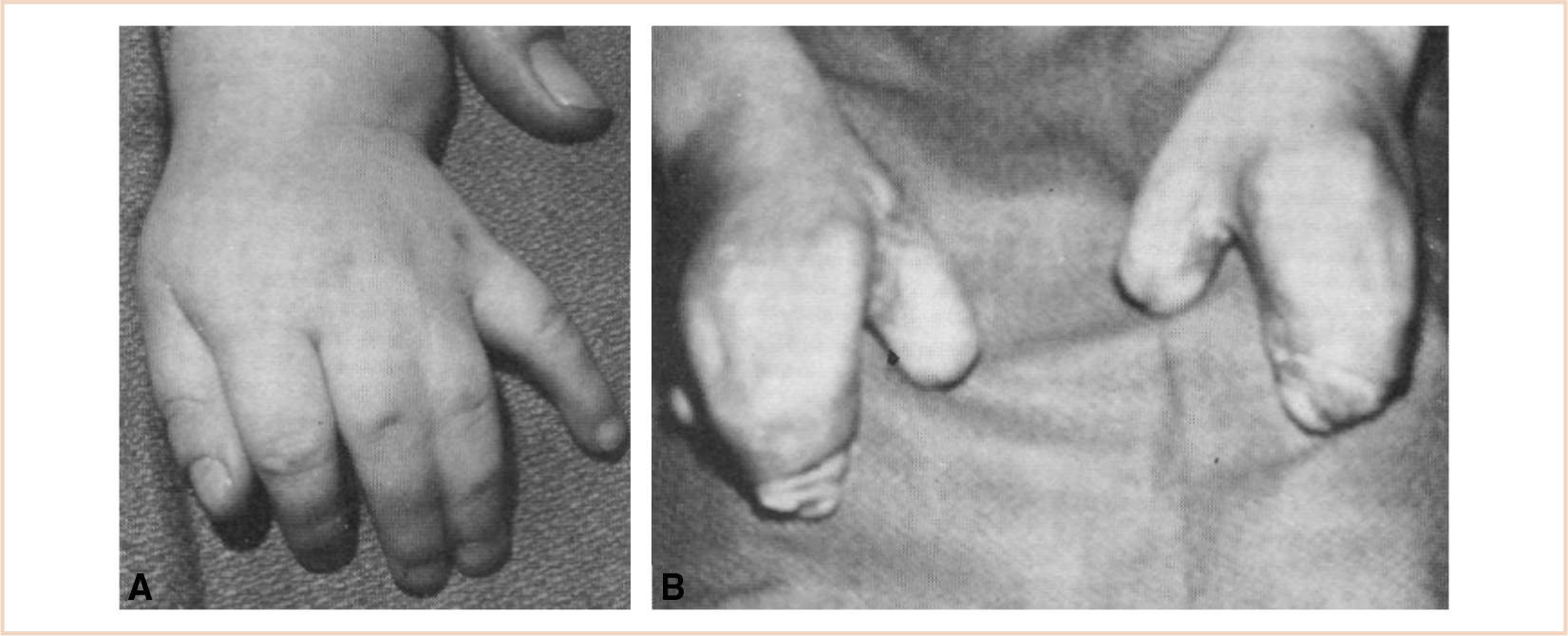
FIGURE 26.8 Syndactyly can range from simple webbing of two adjacent otherwise normal fingers (A), as seen on the left, or extensive webbing of all the digits sometimes known as mitten hand (B).
TABLE 26.5 Syndactyly syndromes
Syndrome | Inheritance | Nonorthopedic features |
Apert syndrome | Autosomal dominant | Craniosynostosis, acrocephaly, midface hypoplasia, small nasopharynx, choanal stenosis or atresia, tracheal stenosis with or without fusion of cervical vertebrae. Ten percent of patients have cardiac anomalies. Cranial involvement may lead to increased intracranial pressure. Vascular access may be difficult. Patients have mutation in the fibroblast growth factor receptor-2 gene (FGFR2). |
Fetal hydantoin syndrome (aka fetal dilantin syndrome) |
| Microcephaly, glaucoma, cleft lip and palate, short neck, webbed neck, midface hypoplasia, congenital heart disease, pyloric stenosis, duodenal atresia |
Carpenter syndrome | Autosomal recessive | Craniosynostosis, acrocephaly, flat midface, hypoplastic mandible, short neck, abnormal dentition, congenital heart defects in 50%.Variable mental retardation w/wo elevated intracranial pressure |
Pfeiffer syndrome | Autosomal dominant | Brachycephaly with coronal and +/− sagittal craniosynostosis, maxillary hypoplasia, rare choanal atresia, laryngomalacia, tracheomalacia, bronchomalacia, obstructive sleep apnea, congenital cardiac malformations, vertebral fusion usually upper cervical, may have seizures and /or increased intracranial pressure. Most cases have mutations in the fibroblast growth factor receptor-1 or -2 gene (FGFR1 or FGFR2) |
Cornelia de Lange syndrome | Autosomal dominant? | Microbrachycephaly, occasional choanal atresia, cleft palate, micrognathia, short neck, GE reflux, repeated aspiration, ventricular septal defect (VSD), severe mental/motor retardation, seizures, sometimes exhibiting autistic self-destructive antisocial behavior. |
PHYSIOLOGIC CONSIDERATIONS: See preceding text under polydactyly.
SURGICAL REPAIR
Simple webbing involving the skin is only usually corrected in the newborn period. More complex reconstructions are usually performed when the children are between 6 months and 2 years of age. When multiple digits are involved, only one side of the digit may be separated at a time in order to preserve vascular integrity.
DISORDER: Severe thumb hypoplasia/finger pollicization
BACKGROUND
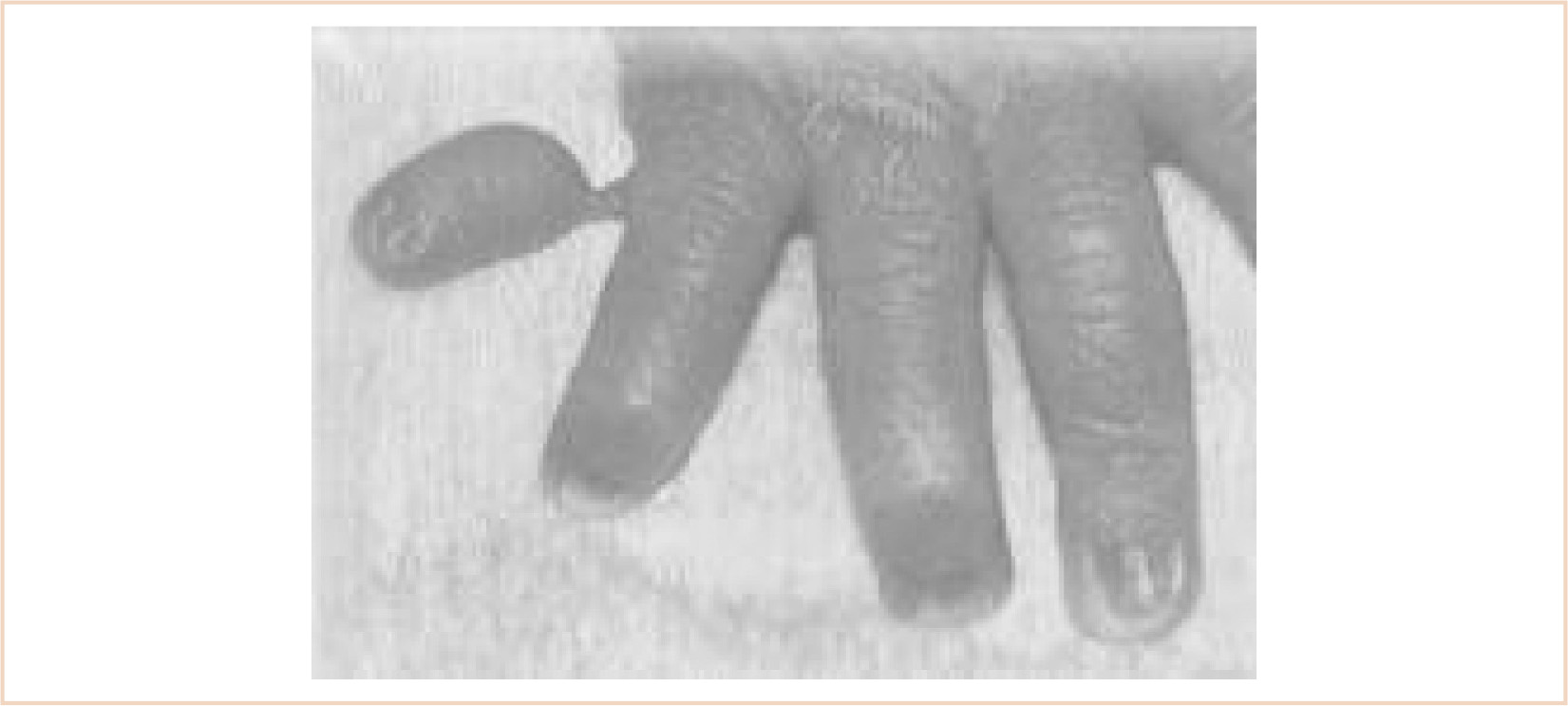
Finger pollicization actually refers to the surgical procedure where the index finger is transferred to the position of the thumb. This type of procedure is necessary in instances of severe thumb hypoplasia. Thumb hypoplasia can vary from a thumb that is slightly smaller than normal to one that may be completely absent (Fig. 26.9). It has been reported to occur in 1 out of every 100,000 births, can affect either or both hands, and may be associated with other conditions where the radial side of the forearm does not develop (4). It may be an isolated anomaly or part of a syndrome (see radial club hand for a list of associated syndromes). These patients need to be carefully evaluated for associated anomalies.
EMBRYOLOGY/ANATOMY: The cause of thumb hypoplasia is unknown.
PHYSIOLOGIC CONSIDERATIONS
1. Tourniquets may be used for more extensive corrections.
a. Increases in blood pressure and heart rate should be treated with short-acting agents.
b. The duration of tourniquet inflation should be carefully monitored and should not exceed 145 minutes before reperfusion, unless determined that deflation or removal would be harmful or endanger the patient’s safety. Following deflation, a minimum of 5 minutes of reperfusion for every 30 minutes of tourniquet inflation is provided.
1. In the absence of associated syndromes, there is infrequently significant pathophysiology.
2. The need to maintain microvascular circulation makes the use of hemodilution techniques, dextran infusions, and regional anesthesia important considerations.
SURGICAL REPAIR
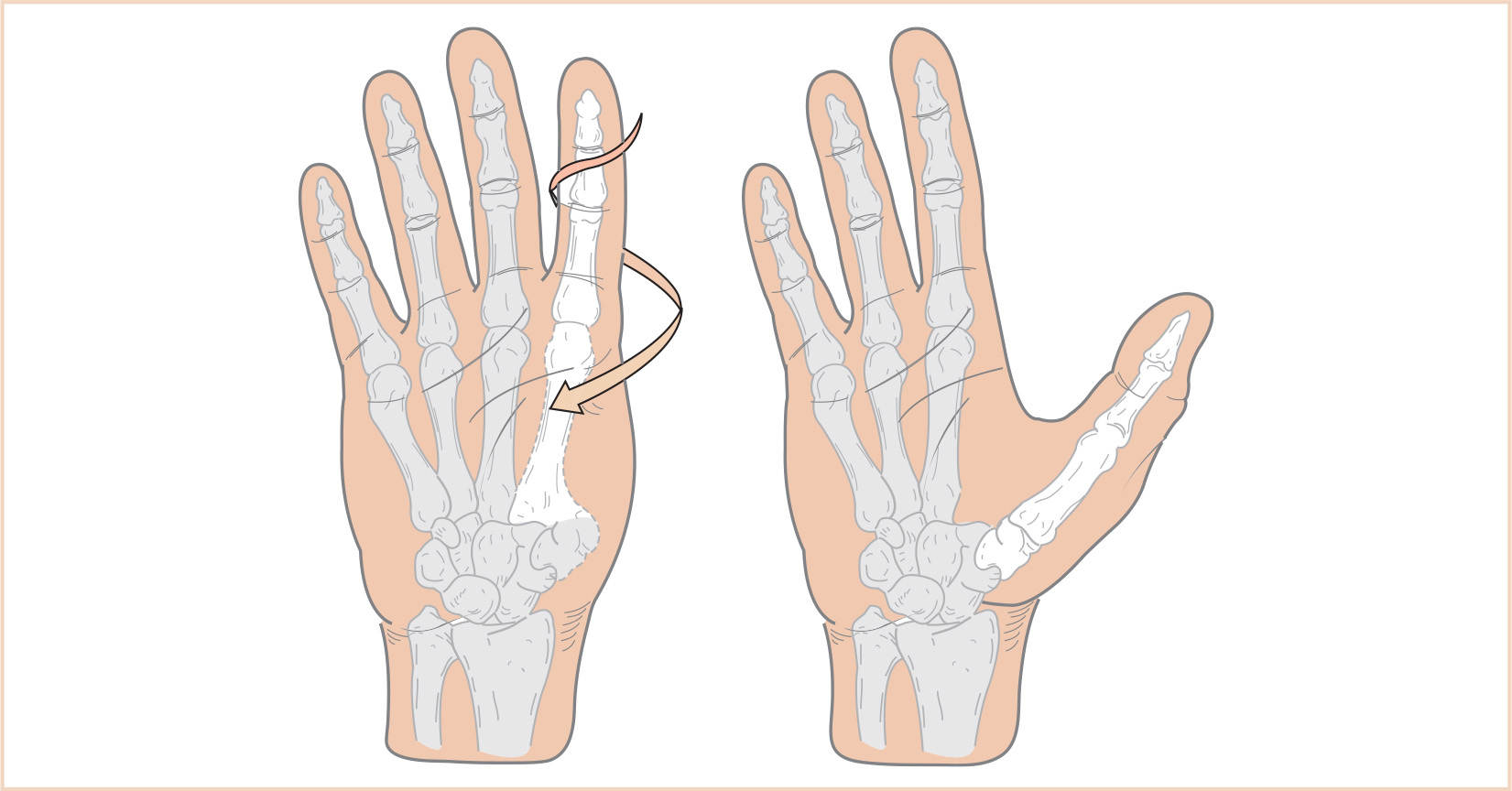
1. Pollicization entails creating a functional thumb by moving a normal index finger to the thumb position (Fig. 26.10).
2. Success of the procedure depends on a normally functioning index finger because the entire finger including blood vessels and nerves are moved to the new position (Fig. 26.11).
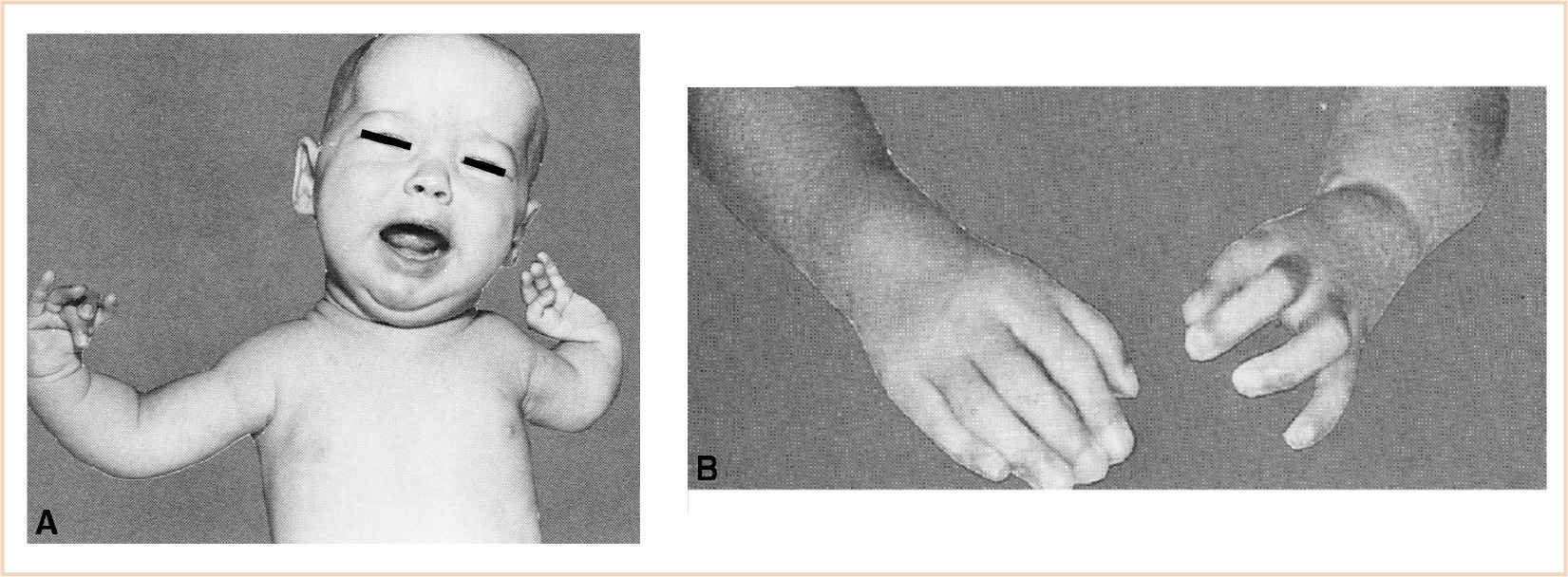
FIGURE 26.11 A: The picture of a girl with bilateral congenital hypoplasia of the radius that is more extensive on the left than right. Note that she has complete aplasia of the left thumb. She also had congenital heart disease that required correction. B: Picture after pollicization of the left index finger when she was 6 years old.
DISORDER: Upper extremity—radial club hand
BACKGROUND
Radial club hand occurs spontaneously, with no known etiology or genetic link. The incidence is between 1:30,000 and 1:100,000 births, and it is classified into one of four types according to severity (see Table 26.6) (5). Although generally not associated with other anomalies, radial club hand may be part of a syndrome (Table 26.7). In these patients, the genetics are those of the related syndrome. These patients need to be carefully evaluated for associated anomalies.
EMBRYOLOGY/ANATOMY
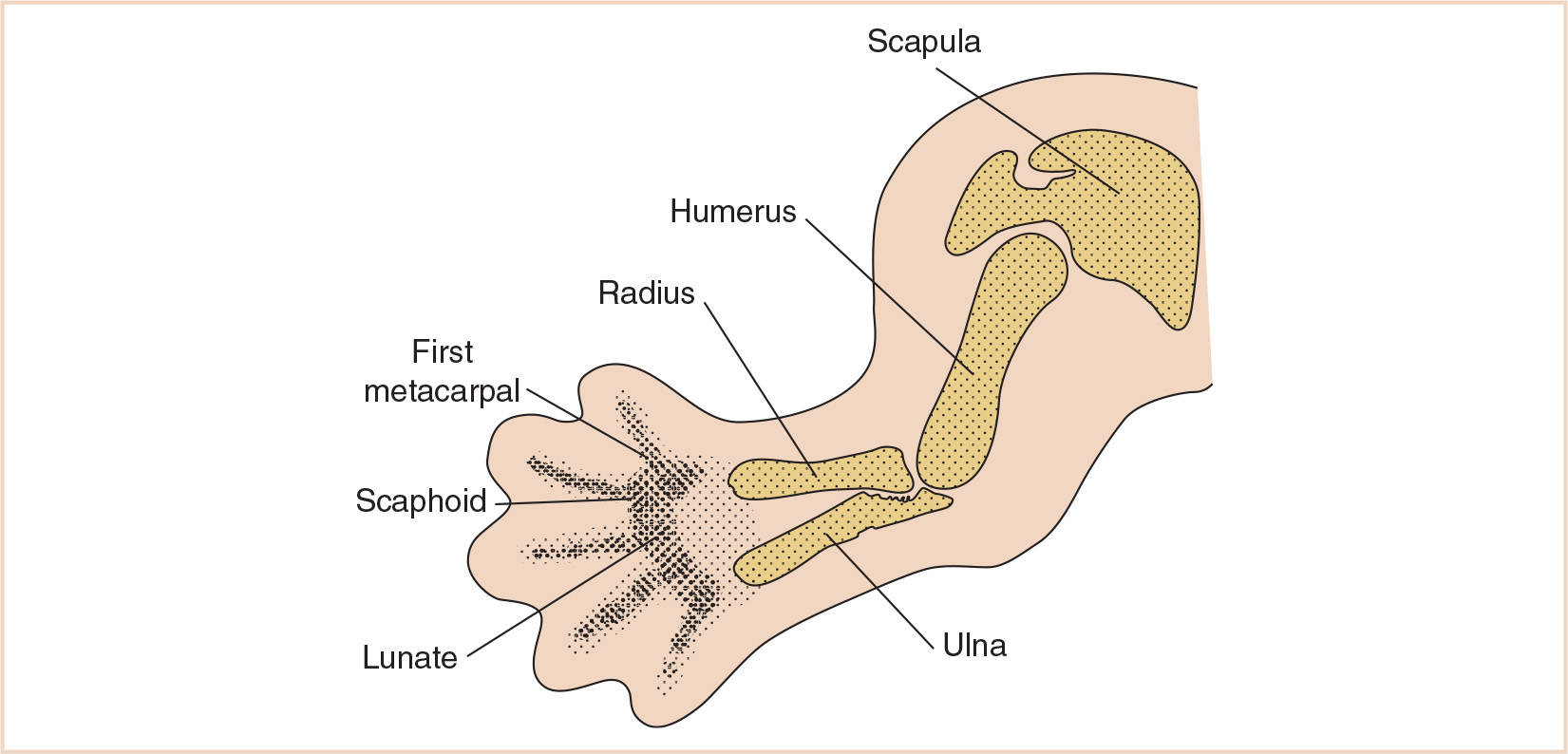
Varying degrees of hypoplasia, all the way to aplasia, of the radial ray of the upper extremity including the radius, scaphoid, trapezium, first metacarpal, and thumb are illustrated (Fig. 26.12). The associated blood vessels, muscles, and nerves are also affected. When the radius does develop, it often grows in a curve. Radial club hand develops early in pregnancy, between the 28th and 56th days.
PHYSIOLOGIC CONSIDERATIONS
Tourniquets may be used for more extensive corrections. Increases in blood pressure and heart rate should be treated with short-acting agents. The duration of tourniquet inflation should be carefully monitored. In the absence of associated syndromes, there is infrequently significant pathophysiology.
TABLE 26.6 Classification of radial club hand
Type | Description |
1 | Mild deviation of wrist with a defect of the growth plate of the distal end of the radius. Usually does not require surgical intervention unless associated with a hypoplastic thumb. |
2 | Limited growth of radius on both distal and proximal ends. Wrist deviation is more prominent, associated with ulna bowing. Hypoplastic thumb and bony deficits are more prominent. |
3 | Absence of 2/3 of radius on distal side. Severe wrist deviation with limited mechanical support of the hand. Hypertrophied and bowed ulna, multiple issues with the thumb and digits. |
4 | Complete absence of radius usually accompanied with complete or near complete agenesis of the thumb. Severe ulna bowing, with limited range of motion. Unfortunately, aside from being the most severe type it is also the most common. |
TABLE 26.7 Radial club hand: commonly associated syndromes
Syndrome | Inheritance | Nonorthopedic features |
Trisomy 13 (Patau syndrome) | Sporadic | Short neck, small mouth, micrognathia, apneic episodes common, severe mental retardation, +/− seizures, renal insufficiency, over 80% have cardiac anomalies. Patients may be mosaics with variable expression of clinical manifestations. |
Trisomy 18 (Edwards syndrome) | Sporadic | Small mouth, micrognathia, apneic episodes and hypertonia as neonate, severe mental retardation, poor thermal regulation, multiple congenital lesions. Only mosaics or patients with a partial trisomy survive past newborn period. |
Trisomy 21 (Down syndrome) | Sporadic | Atlantoaxial instability after age 4 years, smaller than average trachea, macroglossia, pharyngeal muscle hypotonia, congenital heart disease, pulmonary hypertension. |
VATER and VACTERL | Variable | Vertebral anomalies, anal atresia, tracheoesophageal atresia, esophageal atresia, radial and limb anomalies. May be associated with hydrocephalus (autosomal and x-linked inheritance variety). |
SURGICAL REPAIR
1. Mild cases require casting and splinting.
2. Correction of the wrist may involve any or all of the following procedures.
a. Lengthening: This is performed before surgery to stretch the hand into a better orientation. Usually initiated in infancy, but may be used in adolescence for recurrent deformities.
b. Centralization: This procedure removes bones from the wrist so the hand sits straight on the ulna.
c. Radialization: Repositioning the hand further toward the ulnar border of the forearm. The muscles of the wrist are also reimplanted to improve their balance.
d. Splinting: A pin is placed through the wrist and ulna following centralization and radialization procedures to stabilize the correction. This pin may remain in place for 3 months to a year or more.
3. Osteotomies to correct the bowing of the bones of the forearm.
4. Pollicization or tendon transfer to correct the hypoplastic or missing thumb.
5. Tissue releases to improve the range of motion of the elbow.
6. Initial correction of hand deformities occurs before the age of one. Correction of the forearm occurs between age 6 and 8 years and then revisional surgery, if needed, occurs after growth at 12 to 14 years of age.
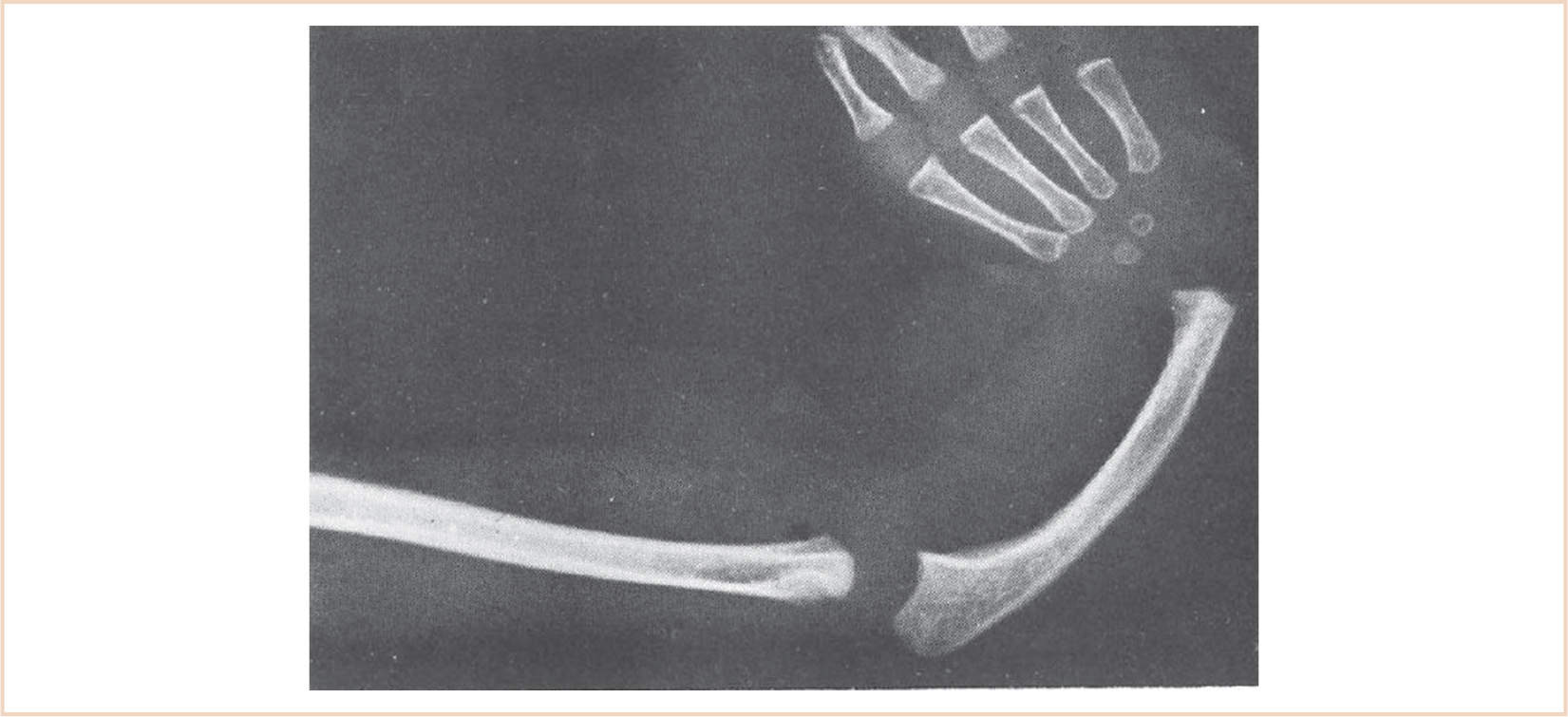
FIGURE 26.12 The radiograph demonstrates the absence of the radius, a hypoplastic curved ulna, radial deviation of the hand, and absence of the thumb and first metacarpal.
ANESTHESIA FOR HAND SURGERY
READINESS FOR SURGERY
1. Patients with multiple anomalies must have a careful preoperative assessment to appropriately address associated conditions.
2. Patients with cardiac and respiratory concerns need to have appropriate preoperative consultation and perioperative intensive care unit (ICU) arrangements for monitoring and care.
3. Specialized equipment may need to be available for difficult airways, external cardiac pacing, and so on.
ANESTHESIA GOALS
1. Secure airway (may need difficult airway protocol for syndromic patients).
2. Maintain stable vital signs, normothermia.
3. Control pain perioperatively.
4. Minimize postoperative nausea and vomiting.
5. Minimal sedation postoperatively to ensure adequate neurologic testing (more complex surgical repairs).
GENERAL ANESTHESIA
Position: Supine
Typical Surgical Time: One to multiple hours depending on skeletal involvement and the number of digits involved.
Induction: Inhalation mask induction with airway secured by mask or laryngeal mask airway (LMA) for shorter procedures, endotracheal intubation with a short-acting or no muscle relaxation for longer more involved procedures. Aspiration precautions are necessary for those syndromic patients with reflux.
Equipment: Routine; single intravenous (IV) cannula; convection warmer
Monitoring: Standard monitors, peripheral nerve monitoring per surgeon
Maintenance: Balanced general anesthesia with inhalational agents and narcotics
Emergence: Extubation at the conclusion of the case. Patient should be alert enough to participate in neurologic examination of the arm and hand at the conclusion of the case.
Perioperative: Postoperative monitoring of digits for continued brisk capillary filling especially when circumferential casts are applied.
REGIONAL ANESTHESIA
1. Not routinely used for these cases because it may interfere with the neurologic assessment on the more extensive excisions.
2. Local infiltration may be utilized adjunctively.
3. Interphalangeal (metacarpal, metatarsal) block may be utilized adjunctively.
DISORDER: Completely severed digits
BACKGROUND: Usually a result of childhood accidents, often in rural areas or around power tools.
EMBRYOLOGY/ANATOMY: Usually normal before injury
PHYSIOLOGIC CONSIDERATIONS
1. Tourniquets will be used to limit blood loss and provide a clear field.
a. Increases in blood pressure and heart rate should be treated with short-acting agents.
b. The duration of tourniquet inflation should be carefully monitored as the tourniquet will need to be deflated and reinflated for longer cases.
SURGICAL REPAIR
1. Careful debridement of recovered digits.
2. Severed digits should be kept cold at 4°C while awaiting reimplantation.
3. Nerves, digital arteries, and veins will need to be anastomosed, ideally two digital arteries and two to four veins per digit.
4. All flexor and extensor tendons will require a primary repair.
5. Bone fragments require oblique or cross fixation with K-wires to maintain the integrity of the joints.
ANESTHESIA ISSUES
READINESS FOR SURGERY
1. These cases need to proceed as quickly as possible to minimize ischemic time.
2. The need to proceed rapidly should not preclude a thorough evaluation of a trauma victim, as other systems may require surgical intervention as well.
ANESTHESIA GOALS
1. Secure airway, avoid aspiration.
2. Maintain stable vital signs, normothermia.
3. Maximize microvascular circulation.
4. Minimize intraoperative and postoperative vasoconstriction.
5. Minimize postoperative nausea and vomiting.
GENERAL ANESTHESIA
Position: Supine
Typical Surgical Time: Multiple hours with the duration dependent on the extent of the injury
Induction: IV rapid-sequence induction with endotracheal intubation with a short-acting muscle relaxant.
Equipment: Routine, multiple IV cannulas, Foley catheter, and convection warmer; depending on the extent of injuries, an arterial line may be useful.
Monitoring: Standard monitors, peripheral nerve monitoring per surgeon
Maintenance: Balanced general anesthesia with inhalational agents and narcotics
Emergence: Although extubation at the conclusion of the case would be preferable, extensive surgeries with fluid shifts may make this impossible.
Perioperative
1. Postoperative monitoring of digits for continued brisk capillary filling and viability of anastomosed tissue.
2. When patients are adequately awake, more extensive neurologic assessment is conducted.
REGIONAL ANESTHESIA
1. Routinely used for these cases because vasoconstriction may be mitigated by long-acting local anesthetics that block sympathetic pathways.
2. Local infiltration may be utilized adjunctively.
3. Continuous bupivacaine infusion improves microcirculation of revascularized tissues.
DISORDERS OF THE UPPER EXTREMITIES: SHOULDER
DISORDER: The Sprengel deformity
BACKGROUND
The Sprengel deformity is the most common congenital malformation of the shoulder girdle with a male to female ratio of 3:1. The condition is usually sporadic although it can be familial with an autosomal dominant pattern of inheritance. The scapula is dysplastic and located higher than normal. There may be some limitation of motion due to an omovertebral connection between the medial border of the scapula and the lower cervical spinous process. This rhomboid- or trapezoid-shaped cartilage or bone can be found in approximately one third of these patients. Additionally, the prescapular muscles may be contracted and fibrotic. These deformities can vary in severity and have been classified by Cavendish (Table 26.8). The Sprengel deformity (Fig. 26.13) is usually associated with other malformations including, but not limited to, fused ribs, chest wall asymmetry, cervical ribs, congenital scoliosis, and cervical spina bifida. It can also be associated with a number of syndromes. These patients must be carefully evaluated for other anomalies.
TABLE 26.8 Cavendish grades
Grade 1 | Very mild | Shoulders almost level. Clothing mask deformity. |
Grade 2 | Mild | Shoulders almost level, but superomedial portion of high scapula is visible. |
Grade 3 | Moderate | Shoulder is elevated 2–5 cm higher on the affected side. |
Grade 4 | Severe | Scapula is very high with superomedial angle at occiput with neck webbing and brevicollis. |
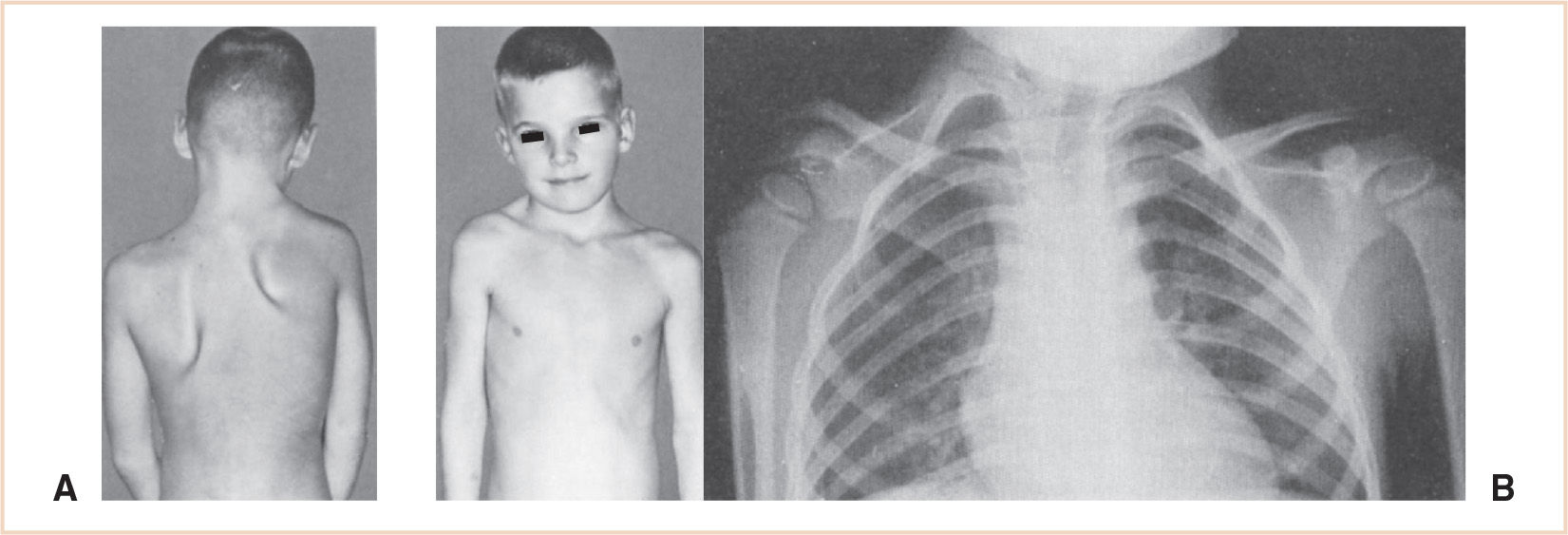
FIGURE 26.13 Congenital high scapula in a 6-year-old boy. (A) The right scapula is smaller and higher than the left scapula; it is also adducted. Treatment consists of surgical resection of the upper one third of the scapula and surgically lowering the bone. The photograph on the right (B) demonstrates the omovertebral bone (connection between the scapula and the cervical vertebra) between the superomedial corner of the small right scapula and the lower cervical spinous processes.
EMBRYOLOGY/ANATOMY
The scapula begins as a cervical appendage that differentiates opposite the fourth, fifth, and sixth cervical vertebral bodies at approximately 5 weeks of gestation. Normally it descends into the appropriate position approximately 90 days later. Any obstruction to this migration will result in the hypoplastic, elevated scapula associated with the Sprengel deformity.
A disturbance of the development of the subclavian artery, vertebral artery, or one of their branches around the sixth week of development can result in one of the subclavian artery supply disruption syndromes. Klippel–Feil sequence, Moebius sequence, and Poland sequence all share common chest wall and limb anomalies. The Klippel–Feil sequence is the most common of the sequences associated with the Sprengel deformity and is also the syndrome with the greatest impact on anesthetic management (Table 26.9).
PHYSIOLOGIC CONSIDERATIONS
Because the scapula is not only high and small but is also rotated downward these patients will often present with a limited ability to abduct the shoulder. Other unusual physiologic considerations are dependent upon the particular comorbidity.
SURGICAL REPAIR
1. Resection of the upper third of the scapula and omovertebral bone
2. Surgically lowering the scapula
DISORDER: Shoulder dislocation
CLINICAL PEARL A multidisciplinary approach is often needed for children with concurrent multisystem disease requiring orthopedic surgery.
TABLE 26.9 Syndromes associated with Sprengel’s deformity
Syndrome | Inheritance | Nonorthopedic features |
Klippel–Feil syndrome | Sporadic | Micrognathia, webbed neck, torticollis, limited mobility of cervical spine, cervical spine may be unstable, syncope may be associated with sudden neck rotation congenital cardiac anomaly |
Greig syndrome | Aut dom | None that might affect anesthetic management |
VATER or VACTERL | Variable | Vertebral anomalies, Anal atresia, Tracheoesophageal atresia, Esophageal atresia, Radial and Limb anomalies. May be associated with hydrocephalus (autosomal and x-linked inheritance variety |
Velocardiofacial syndrome | Aut dom | Microcephaly, micrognathia, mental retardation, congenital heart disease |
Floating Harbor syndrome | Sporadic | None that might affect anesthetic management |
Goldenhar syndrome | Sporadic | Craniofacial vertebral anomalies making airway management difficult. Difficulties increase with age. Arnold–Chiari malformation or hydrocephalus, congenital heart defects, renal anomalies |
BACKGROUND: The shoulder has the greatest range of motion of all joints. Dislocations most commonly occur when the humeral head rolls out the front of the glenoid fossa when the arm is struck with the elbow held out, the classic position for a football linebacker. Less commonly, the humeral head can also be pushed out posteriorly from the glenoid fossa. This can occur from breaking a fall with an outstretched hand or from a direct football tackle to the front of the shoulder. Dislocations, although rare in young children, are not uncommon in adolescents.
EMBRYOLOGY/ANATOMY
The humeral head sits in the glenoid fossa, a shallow cup-shaped socket. This socket is made functionally deeper with a rim of fibrocartilage called the labrum. There are three main ligaments that attach to the humeral head and the glenoid. These are integrated into a loose capsule that forms around the joint. Different ligaments tighten at different arm positions to hold the shoulder together. Rotator cuff muscles and tendons wrap around the humeral head and pull deeply into the glenoid fossa to further improve stability. A dislocation occurs when the ligaments, rotator cuff, or bone of the shoulder are torn, stretched, or otherwise damaged.
PHYSIOLOGIC CONSIDERATIONS
There are no special physiologic considerations.
SURGICAL REPAIR
1. The humeral head can be relocated back into the glenoid fossa. This can occur spontaneously following rest, ice compression, and elevation (RICE). However, because of the ensuing muscle spasm, traction with some sedation will most likely be needed to relocate the shoulder. The arm is then placed in a sling to minimize stress to the injured capsule.
2. Surgery focuses on tightening the stretched capsular ligaments and/or repairing the torn labrum. Arthroscopic techniques may be used, although open repair is currently the favored technique. The resulting stabilization is successful 90% to 95% of the time.
ANESTHESIA ISSUES
READINESS FOR SURGERY
1. Because this can be emergency surgery, many patients may be considered as having a full stomach before their anesthetic.
2. One needs to be cautious when asked for “a little” sedation in patients who have not been properly fasted.
ANESTHESIA GOALS
1. Good relaxation for relocation of shoulder.
2. Secure airway for anything more than minimal sedation.
3. Maintain stable vital signs, normothermia.
4. Control pain/spasm perioperatively.
5. Minimize postoperative nausea and vomiting.
GENERAL ANESTHESIA
Position: Supine; occasionally, beach chair. The beach-chair position is associated with decreased cerebral perfusion, so avoidance of hypotension is mandatory.
Typical Surgical Time: Minutes for a reduction to 60 to 90 plus minutes for a surgical repair.
Induction: IV sedation or inhalation mask induction with airway secured by mask or LMA for younger patients. Endotracheal intubation with a short-acting muscle relaxant for patients with full-stomach considerations. Intraoperative muscle relaxation may or may not be required to facilitate the surgical repair; this should be discussed with the surgeon.
Equipment: Routine; single IV cannula; convection warmer
Monitoring: Standard monitors, peripheral nerve monitoring per surgeon
Maintenance: Balanced general anesthesia with inhalational agents and narcotics
Emergence: Extubation at the conclusion of the case
Perioperative: Special attention for positioning patients
REGIONAL ANESTHESIA
1. Not routinely used for reductions.
2. Local infiltration or interscalene nerve block may be helpful for surgical repairs. Regional anesthesia for surgery in the beach-chair position is not associated with decreased cerebral perfusion as measured by near infrared spectroscopy even when the patient is hypotensive.
DISORDER: Fractures and dislocations (fingers, wrist, elbow, closed vs. percutaneous pinning vs. open)
CLINICAL PEARL Adjunctive regional and peripheral nerve blocks are an essential part of delivering anesthesia for orthopedic procedures.
BACKGROUND
1. Because bone growth is not yet completed, greenstick fractures and epiphyseal injuries are unique to children. Although most fractures can wait until an appropriate NPO period has passed, fractures around the elbow and any fracture resulting in the loss of a peripheral pulse require immediate attention.
2. Children who have traumatic injuries need a complete workup to assure that all the injuries are being appropriately addressed.
3. Children should also be assessed for signs of abuse. X-ray evidence of multiple fractures at various stages of healing, multiple soft tissue injuries, especially on the abdomen, or cigarette burns are all signs that additional social situation evaluation is warranted.
EMBRYOLOGY/ANATOMY: The embryology and anatomy were usually normal before the injury.
PHYSIOLOGIC CONSIDERATIONS
Volkmann’s ischemic contracture, first described in 1881, is most commonly associated with supracondylar fractures, but may also be associated with soft tissue injuries around the elbow or may occur following fractures of both bones of the forearm. Arterial occlusion is caused by brachial artery entrapment by fracture fragments, arterial spasm, or compression due to swelling. This in turn causes muscle ischemia which increases capillary permeability, leading to more intramuscular edema that further increases tissue pressure. The vicious cycle continues, leading to necrosis of the muscle with secondary fibrosis and calcification. Treatment requires immediate attention to improve circulation. This usually consists of removing constricting bandages, reduction of the fracture, and extension of the elbow. Fasciotomy and exploration of the brachial artery may be necessary.
Tourniquets may be used for more extensive corrections. Increases in blood pressure and heart rate should be treated appropriately with short-acting agents. The duration of tourniquet inflation should be carefully monitored.
Pediatric trauma cases performed in the early hours of the morning tend to require smaller doses of analgesics to produce the same degree of somnolence in patients. Although this has not been formally studied it is a consistent clinical observation. It is speculated that this is related to the child’s natural diurnal rhythms as well as accumulated fatigue and exhaustion at the end of a long day of multiple examinations.
SURGICAL REPAIR
1. Simple fractures may be reduced with a closed reduction. Pinning may be necessary to maintain the reduction.
2. Open reduction with internal fixation is necessary if a closed reduction is impossible, if the fracture crosses the epiphyseal plate, or if there is soft tissue trapped within the bony fragments. Other indications include displaced avulsion fractures, intra-articular fractures, and fractures with coexisting vascular injury that require exploration and/or repair.
ANESTHESIA ISSUES
READINESS FOR SURGERY
1. Preoperative assessment should carefully review the time of last food intake before injury and the time of the injury; the NPO status is suspect from the time of the injury.
2. An IV may need to be placed with nitrous oxide sedation, or a skin wheal of local anesthetic.
3. The loss of a peripheral pulse makes this a truly urgent surgical emergency.
4. Be certain to review medications given in the emergency department.
ANESTHESIA GOALS
1. Secure the airway. A rapid-sequence induction should be the default technique until additional information is gathered.
2. Maintain stable vital signs, normothermia.
3. Control pain perioperatively.
4. Minimize postoperative nausea and vomiting.
a. Antiemetics
b. Oral gastric suctioning
GENERAL ANESTHESIA
Position: Generally supine
Typical Surgical Time: Thirty minutes to hours depending on the injury and repair required
Induction: Inhalation mask induction with the airway secured by mask or LMA (if appropriate) for shorter, more elective procedures. Neuromuscular blockade may be required for longer, more involved procedures. Patients with full stomachs undergoing emergency surgery should be induced with a rapid-sequence technique.
Equipment: Routine; single IV cannula; convection warmer
Monitoring: Standard monitors, peripheral nerve monitoring per surgeon
Maintenance: Balanced general anesthesia with inhalational agents and narcotics. Short-acting muscle relaxation may be necessary for closed reductions. Muscle spasm in the forearm especially in athletes may make reducing the fracture impossible without blocking the neuromuscular junction.
Emergence
1. Extubation at the conclusion of the case.
2. Patient should open eyes spontaneously before extubation as risk of vomiting and extubation is still present.
Perioperative: Postoperative monitoring of digits for continued brisk capillary filling even if circumferential casts have been bivalved.
REGIONAL ANESTHESIA
1. Brachial plexus, regional IV in older more cooperative patients, or as a combined general/regional technique.
2. Stellate ganglion block for fractures with arterial spasm.
3. Local infiltration may be utilized adjunctively.
4. Interphalangeal (metacarpal) block may be utilized adjunctively.
DISORDER: Brachial plexus injury
BACKGROUND
Neonatal brachial plexus injuries occur at a rate of 1 to 3 cases per 1,000 live births (1,6). Risk factors include maternal gestational diabetes, forceps delivery, and shoulder dystocia. Approximately 73% of cases involve the upper cervical roots C5 and C6. Clinically, this presentation is known as Erb’s palsy. Patients with an Erb palsy hold their shoulder in an adducted, internally rotated position with the elbow in extension, the forearm in pronation, and the wrist and fingers in flexion because of variable weakness in the wrist and finger extensors (flail arm) (Fig. 26.14). Approximately 25% of patients have total plexus injuries which are characterized by complete arm paralysis, decreased sensation, and a pale extremity and only 2% of patients exhibit an isolated lower or Klumpke palsy which is manifested by a paralysis confined to the hand (2,7).
EMBRYOLOGY/ANATOMY
Formation of the brachial plexus begins in early development in the fourth week of gestation. Because the sclerotome directs axonal growth, nerve formation follows the dorsal rotation of the upper limb bud. Axons from the ventral column motor cells start to grow toward the sclerotome cell mass, forming the ventral root. The dorsal root similarly forms by axons growing in the opposite direction from the dorsal root ganglion cells. These roots join to form three trunks, the superior trunk which is made up of the C5 and C6 roots, middle trunk which is made up of the C7 root, and inferior trunk which is formed by the roots of C8 and T1. These trunks divide into anterior and posterior divisions, with the anterior divisions of the brachial plexus destined to innervate the ventral muscles, and the posterior divisions destined to innervate the dorsal or extensor muscles (Fig. 26.15).

Full access? Get Clinical Tree