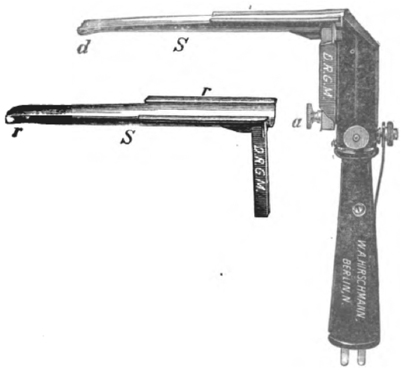
Fig. 54.1
Trendelenburg apparatus. (Courtesy of American Society of Anesthesiologists, ASA Newsletter Vol 72, Sep 2008, p. 9)
In 1909, Chevalier Jackson advanced the performance of tracheostomy by describing what are now modern techniques and principles (optimizing airway control before surgery; employing local anesthesia; using a well-designed tube; and applying meticulous surgical technique and postoperative care). These principles decreased mortality to 3% and increased the acceptance of the procedure [10,14,15].
Cricothyrotomy
In 1805, French surgeon and anatomist Vicq d’Azyr described cricothyrotomy [16]. Initially ignored, these “high tracheostomies” increased in popularity in parallel with tracheostomies. However, concerned about the associated risk of subglottic stenosis, Chevalier Jackson decried cricothyrotomy. His objections hindered the performance of cricothyrotomy for over half a century [13,14,17].
In 1976, Colorado cardiothoracic surgeons Charles Brantigan and John Grow, Sr. published results of cricothyrotomies in 655 patients, with no incidence of chronic subglottic stenosis. Published complication rates of traditional tracheostomy exceeded the overall complication rate of 6.1%. Brantigan and Grow concluded that cricothyrotomy was faster, easier, and less likely to bleed [16]. Today, although the use of either tracheostomy or cricothyrotomy has decreased, they remain the preferred techniques when all other approaches have failed [7–9].
Anesthesia Facemasks and Upper Airway Obstruction
For more than a century, the facemask provided the primary interface to the anesthetic system for spontaneously breathing patients, and for most of those receiving positive pressure ventilation. After the 1950s, tracheal tubes increasingly displaced the facemask. Ventilation via a facemask remains an essential component of all difficult airway algorithms [7–9]. Modern transparent, high-volume, low pressure disposable plastic facemasks minimize dead space, maximize seal, and allow prompt recognition of regurgitation’as the ideal facemask should [18–20].
The facemasks created soon after the discovery of anesthesia took two forms. One provided a gauze surface (sometimes simply a handkerchief) onto which ether or chloroform was dripped or poured, and through which the patient might breathe, acquiring anesthetic in the process. The gauze might be supported by a wire frame, such as that devised by Thomas Skinner, a Scottish obstetrician [21]. These devices evolved into many variations, the most popular being the Yankauer and Schimmelbusch masks. Facemasks made of gauze had the advantages of simplicity and minimal cost, but the operating room became contaminated with anesthetic vapor (wasting anesthetic in the process) and positive pressure ventilation was not possible.
Skinner’s reason for designing the wire frame mask was truly advanced for his time. Although the antisepsis debate developed eight years later, Skinner felt that facepieces preceding his design were “disgusting”. Typically, a mask would be used and reused, without cleaning, without considering whether it had been applied to a patient with a possibly contagious infection before applying it to the next patient. Skinner’s mask came with multiple flannel cloth coverings that could easily be replaced allowing each patient to have a clean mask [22,23].
The second form of facemask, like that devised by Snow, might be recognized as the forerunner of the modern mask. It was made with harder material’leather or leather edged metal. In the latter part of the nineteenth century, most masks (often in the form of “inhalers”) were made of metal, sometimes with a rubber rim. Masks in the first half of the twentieth century were made of rubber, softer than leather or metal, but far harder than today’s masks.
In the late 1950s, pediatric facemasks were miniature versions of adult masks. Because an infant’s face differs from an adult’s, these masks provided a poor seal and imposed a deadspace that might equal an infant’s tidal exchange. Anesthesiologist Leslie Rendell-Baker and dental student Donald Soucek, (both from Western Reserve University) designed a better facemask from plaster moulds of infants’ faces. They eliminated the cuffed rim and added a partitioned adapter that further minimized deadspace. Their 1961 result produced the still popular RBS facemask for children ≤5 years old [24,25].
Adequate ventilation via facemask requires both a good seal and an unobstructed airway [18,19]. In the late 1800s, Joseph Clover, Jacob Heiberg and Johannes Von Esmarch each recommended jaw thrust to relieve obstruction [20,26,27]. In 1894, surgeon Theodor Kocher advocated jaw thrust and chin lift for all anesthetized patients [28]. Jaw thrust became known as Esmarch’s maneuver although it was first described by Heiberg [26,27]. In 1880, Benjamin Howard found that head tilt and neck extension opened the airway by lifting the epiglottis and tongue [27]. In the 1950s and 1960s, radiologic studies documented that chin lift, head tilt, and jaw thrust opened the airway [18,20].
Position change was used to resolve some forms of upper airway obstruction. Snow argued that the recumbent position permitted surgical debris to pool in the back of the throat where it might pass down the esophagus’never mind that it might also enter the glottis. If the anesthesiologist heard gurgling sounds, the patient’s head should be tilted forward or sideways to empty accumulated debris [4,29,30]. British anesthetist Alexander Wilson tied his patients to a rocking chair for surgeries of the tongue, thereby allowing an array of positions: reclined for induction; seated for surgery; and, body/head tilted forward to clear the pharynx of debris [29,30]. James Murphy described an 1894 Parisian patient position’supine with his head hanging over the end of the operating table, using gravity to protect the airway against aspiration. In 1904, operating tables were fitted with special headrests to simulate Murphy’s position [30]. Trendelenburg’s downward tilt was similarly used to drain debris from the airway [31].
Devices Used to Manage Upper Airway Obstruction
Tongue Forceps
In an 1856 letter describing “never before pointed out impediments of respiration,” Marshall Hall observed that glottic obstruction sometimes occurred in supine patients. He believed that relaxation of the tongue caused the epiglottis to block the glottis [26]. In seeming support of Hall’s suggestion in 1880, Benjamin Howard found that traction on the tongue to displace it from the posterior pharyngeal wall could relieve obstruction (actually implying that the tongue and not the epiglottis was the problem) [27].
Accordingly, tongue forceps were invented (Fig. 54.2). But in 1874, Jacob Heiberg gave a frightful image of forceps-based management of airway obstruction. Their use broke teeth and injured the tongue. Injury could impair speaking and/or swallowing and cause prolonged pain [26]. Despite the English Suspended Animation Committee’s 1879 objection to these devices’recommending instead Esmarch’s maneuver’tongue forceps continued in use into the 1930s [26,32].
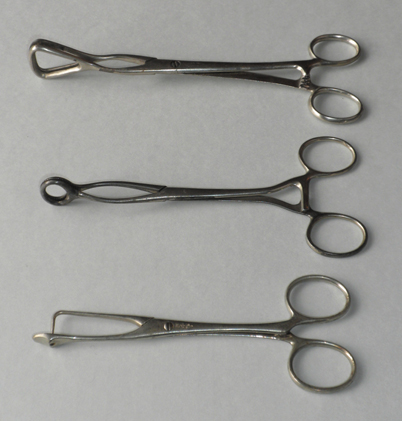
Fig. 54.2
Various tongue forceps were devised to allow the user to pull the subject’s tongue forward, sometimes damaging tongue and teeth in the process. (Courtesy of Rod Westhorpe and the Geoffrey Kaye Museum of Anaesthetic History)
Oropharyngeal and Nasopharyngeal Airways
An oropharyngeal or nasopharyngeal airway may be used to assist in opening the upper airway [18,19]. In 1859, Faure of Paris inserted a long rubber tube through the nares, a primitive nasopharyngeal airway, to create a passage for chloroform delivery [33]. Similarly, Joseph Clover in 1881 connected his chloroform/nitrous oxide bag to an India-rubber nasal tube to deliver anesthesia during jaw surgery [32–34]. In 1913, Karl Connell, a Columbia University surgeon, described anesthetic delivery through either a single or double nasopharyngeal airway [32,35].
Frederic Hewitt’s classic 1890 summary noted the frequency and danger of upper airway obstruction. In 1908, dissatisfied with the use of tongue forceps and acknowledging that Esmarch’s maneuver sometimes failed, he described the first oropharyngeal airway, the Hewitt Airway (Fig. 54.3). The tube was less than 8 cm long, ideally connecting the oral opening to the hypopharynx [27,30,36]. Within a year, a longer curved version replaced the first, straight model. The Hewitt Airway anticipated many subsequent oropharyngeal airway designs [4,27,32].

Fig. 54.3
Hewitt oropharyngeal airway, original straight version on left. (Courtesy of Rod Westhorpe and the Geoffrey Kaye Museum of Anaesthetic History)
Improvements and increased use of the oropharyngeal airway followed World War I [32]. In 1930, Ralph Waters, the first Chairman of an Anesthesiology department in the US (University of Wisconsin), designed an airway made of metal, further differing from Hewitt’s design by being oval in shape and later including a side tube for insufflation [34,37,38].
Although Arthur Guedel and Waters never worked at the same institution, they corresponded and collaborated on research projects [39,40]. Guedel, at the University of Southern California, believed that steel oropharyngeal airways caused oral trauma [38–40], and that it could be better contoured to fit the shape of the pharynx [32,40]. Thus he made his airway of rubber, sufficiently soft to be flexible and perhaps less traumatic, yet rigid enough to maintain a patent airway. It included a metal piece at the level of the incisors as a bite block [39,40]. A wide rubber flange at the oral opening prevented over insertion and allowed easy retrieval [40]. Guedel wrote to Waters “As I use the rubber airway more and more, I am growing to like its action better and even as well as or better than the metal (Waters) airway. I am proud of the idea…” [40]
Resident Robert Berman had an incident with a Guedel Oral Airway which resulted in a new oropharyngeal airway [41]. He had placed a Guedel Airway in a shirt pocket that also contained safety pins (safety pins were often used to puncture the lead tops of containers of ether; they formed a wick that facilitated the controlled delivery of ether). After using the oropharyngeal airway during induction, he performed laryngoscopy and noticed a safety pin in the posterior pharynx. The opacity of the black rubber had hidden the presence of the safety pin, which had been dislodged by the force of ventilation [41]. Berman discussed the problem with his neighbor, Meyer Moch, a plastic fabricator. Excited by the problem and seeing a solution, Moch and Berman retired to Moch’s basement where they softened butyrate tubing, formed it into the shape of an airway, and sanded it smooth. Since Moch lacked pharyngeal reflexes, they immediately tested its fitting and function on him [41]. In 1951, Berman and Moch officially introduced the airway. The final version differed from Guedel’s in having no closed tube that might trap foreign bodies. Instead, it had a central longitudinal strut with upper and lower flanges [41,42]. See Fig. 54.4 for a view of the different types of oropharyngeal airways.

Fig. 54.4
Oropharyngeal airways. From top to bottom, Connell, Guedel, and Berman oral airways. (Courtesy of Rod Westhorpe, and the Geoffrey Kaye Museum of Anaesthetic History)
While these oropharyngeal airways could overcome upper airway obstruction, their use did not preclude aspiration. Without tracheal intubation, packing with gauze, sponges, or repositioning of the patient was used to manage this complication. Such management was imperfect and gauze and sponges could be swallowed or inhaled. An alternative solution was required [43]. Just before retiring in 1935, Francis Shipway, anesthetist to King George V, designed the Shipway Oropharyngeal Airway (Fig. 54.5). The Shipway Airway added an inflatable cuff to the Hewitt Airway. It was specifically designed to prevent aspiration of blood during intranasal surgery [30,43,44]. A special fitting permitted the connection of the Shipway Airway directly to an anesthetic circuit [30,43,44].
In 1937, Canadian Beverly Leech invented the Leech Pharyngeal Bulb Gasway. A solid rubber bulbous plug with a hollow core surrounded the distal end of this metal oropharyngeal airway, the assembly fitting closely to the pharyngeal tissues to provide an airtight seal [32,44–46]. While not an inflatable cuff, the plug served a similar function and is often called a “cuffed device”. Its design was based on studies of pharyngeal anatomy [30,32,45]. Used to deliver closed circuit cyclopropane anesthesia, it remained popular until tracheal intubation rendered it obsolete in the mid-1950s [45].
In 1991, Robert Greenberg, at John Hopkins developed the Cuffed Oropharyngeal Airway, a Guedel oral airway which included an inflatable cuff and a 15 mm connector for attaching to an anesthesia circuit. The cuffed oropharyngeal airway was prone to dislodgement when the cuff was inflated [45]. Removal with its cuff inflated helped prevent aspiration of blood or secretions.
Supralaryngeal Airway Devices
Before the 1980s, ventilation via a facemask supplied the primary alternative for tracheal intubation. However, ventilation via a facemask did not preclude aspiration of gastric contents. Securing an airtight seal could also be difficult [4,47,48]. After 1981, progress in supralaryngeal airway devices increasingly overcame these shortcomings.
Determined to develop an airway minimizing the limitations of facemask-assisted ventilation, British anesthetist Archie Brain invented a device connecting the natural airway to an artificial airway that did not require insertion equipment, and did not demand the use of muscle relaxants [4,34,47,48]. Brain noticed that the shapes of plaster casts of a cadaver’s pharynx resembled the Goldman nasal masks used in dental anesthesia. Brain created his first laryngeal mask airway (LMA) by attaching a Goldman nasal mask to a diagonally cut end of a 10 mm Portex tracheal tube, joining them with acrylic adhesive [47–49]. In the summer of 1981, Brain blindly inserted the LMA prototype in a patient undergoing a routine hernia repair under halothane anesthesia, achieving a clear airway that allowed positive pressure ventilation [48,49]. Success! A pilot study in 23 patients undergoing various operations, including gynecological laparoscopic procedures requiring positive pressure ventilation, showed that the device could supply a patent protected airway without requiring muscle relaxants to facilitate placement. Published in the British Journal of Anaesthesia in 1983, the results received surprisingly little attention from the medical or manufacturing community [47–50]. Undeterred, Brain soon filed a dozen patents [4,47].
Despite the lack of commercial interest, Brain continued to modify his invention, experimenting with diverse materials before settling on silicone for the LMA-Classic (cLMA) [47,48]. So strange were some designs that several early prototypes were mistaken for exotic rubber condoms by members of the cleaning staff at Brain’s laboratory in Newham General Hospital [47]. Over the next seven years, Brain produced hundreds of prototypes testing them in approximately 7,000 patients [47–49].
The large number of prototypes and long development process reflected Brain’s quest to balance ease of use, efficacy and safety [48,49]. While one modification improved one aspect of the LMA, it might sacrifice another [48,49]. For example, increasing surface mucosal contact with the cuff provided greater seal pressure but caused ischemia of the tongue. Also, the use of an insertion device eased placement, but increased the risk of trauma. With clinically acceptable seal pressures of 20 cm H2O, the cLMA became commercially available in 1988 [49,51,52]. Following a demonstration of the new device, Colin Alexander, a British anesthetist and chair at the Royal East Sussex Hospital, acquired the cLMA for his hospital and published his experiences in a letter toAnaesthesia [47,49]. Within 2 years, the LMA was available in every hospital in the UK [34,48]. It was marketed in Australia and New Zealand in 1989. Ronald Katz at UCLA received a prototype LMA in 1985. He quickly wrote of his experiences, sparking interest in the US [49]. The cLMA had been used in more than 2 million cases when approved by the FDA in 1991 [48,49]. Commercial release of the LMA in the US occurred in 1992.
British anesthetist, John Nunn, assisted Brain in marketing the cLMA in the UK [47,48]. US anesthesiologists Jonathan Benumof, Paul White and Andranik Ovasspian paved the way for North American recognition. White and his team demonstrated that the cLMA was a useful airway device for routine anesthetic cases. Ovassapian and Benumof tested its use for “cannot intubate, cannot ventilate” patients. They argued that the cLMA provided a “second-last-ditch” effort for cases of failed intubation when a surgical airway was the only other remaining option [48]. With their influence, the LMA was added to the ASA Difficult Airway Algorithm in 1993, with an expanded role in the 1996 and 2003 algorithms [7–9,51,53]. It is currently listed in difficult airway algorithms throughout the world [9]. The American Heart Association’s 2000 Guidelines for CPR and Emergency Medicine Cardiovascular Care incorporated the disposable LMA-Unique (uLMA) as an essential airway rescue device [52]. Development of the LMA has been touted as one of the greatest advancements in anesthesia since the advent of the tracheal tube. It has changed airway management in emergency, critical care, and ambulatory medicine, as well as anesthesia. For his efforts, Brain received numerous awards and accolades [4,47,48].
Brain revolutionized the use of the supralaryngeal airway, but he was not the first to conceive of the idea [4,48,49]. Oropharyngeal airways (1908) and the Fell-O’Dwyer apparatus (1882) took advantage of placing the tip of an artificial airway into the pharynx rather than the trachea. Some argue that the Leech Gasway (1937) was father to all supralaryngeal airway devices, including the LMA [2,4,34]. Brain’s device differed in a crucial attribute’its approach to sealing the airway. While the LMA used a soft inflatable “mask over the larynx” whose tip is positioned in the esophagus, at least one previous device surrounded the tube at the glottic opening with a solid plug [2,4,34].
While some supralaryngeal airway devices were similar to Brain’s LMA design, others resembled a tracheal tube that enabled pulmonary ventilation without passing through the vocal cords. In 1968, Don Michael developed the first of such devices, the Esophageal Obturator Airway. It had a distal balloon blocking the esophagus and sixteen holes for ventilation between this and a proximal balloon. Difficulties arose in using the Esophageal Obturator Airway and comparative studies with tracheal tubes revealed no advantages, thus the device disappeared [34,54,55].
Austrian internist, Michael Frass, with Reainhard Frenzer and Jonas Zahler envisioned a need for an airway device that could ventilate the lungs regardless of whether the device entered the glottis or esophagus. In 1987, they invented the Esophageal Tracheal Combitube (Fig. 54.6), a device with two lumens separated at the proximal end, allowing attachment of the airway circuit to either [52,54–56]. The esophageal lumen is closed at the tip and has eight perforations between a proximal and a distal cuff. The alternative lumen, the tracheal lumen, is open at the end and when placed in the glottis, functions essentially as a tracheal tube. Thus, regardless of where the device is positioned, the Esophageal Tracheal Combitube provides a patent airway. No time is lost to repositioning, making it particularly useful in prehospital and emergency care settings, where time may be crucial [52,54–56].
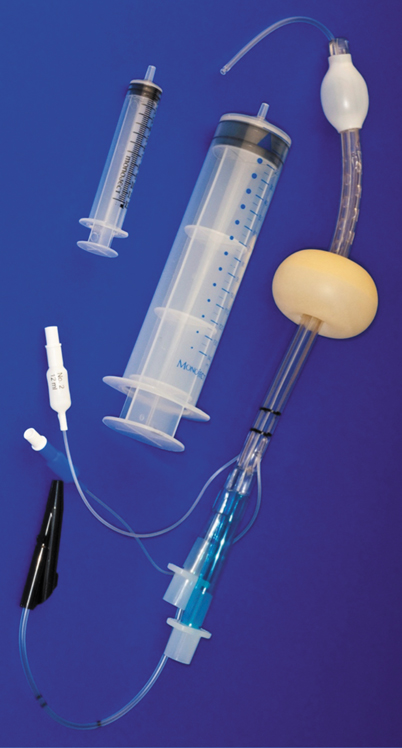
Fig. 54.6
Esophageal-Tracheal Combitube (Frass M et al. [54]) esophageal/endotracheal double-lumen airway: the combitube and the easytube. (From Hagberg C (ed.)Benumof’s and Hagberg’ s Airway Management: Principles and Practice 3rd edition. Mosby, Inc., St. Louis, 2012)
Developed in 2003, the EasyTube was functionally the same as the Esophageal Tracheal Combitube but had a smaller tip and the pharyngeal cuff had a lower volume. The redesigned pharyngeal aperture allowed for easier passage of a flexible fiberoptic bronchoscope, gum elastic bougie or tracheal suction catheter and uses polyvinyl chloride in the large oropharyngeal cuff [34,52,56] Both devices are included in guidelines for difficult airway management and emergency resuscitation throughout the world [9].
Similar in appearance to the Esophageal Tracheal Combitube, the King Laryngeal Tube (King LT) is a supralaryngeal airway device introduced in Europe in 1999 by VBM Medizintechnik. Redesigned by King Systems, it received FDA approval in 2003 [56]. It differs slightly from other supralaryngeal airways in that it consists of a reusable, dual-cuffed single-lumen tube with a blind distal tip. Two anterior ventilation outlets between the low-pressure distal (esophageal) and proximal (oropharyngeal) cuffs allow the device to function as a supralaryngeal airway. Unlike the Esophageal Tracheal Combitube or EasyTube, both cuffs are inflated through a single valve [52,55,56]. The King LT can be used during both positive pressure ventilation and spontaneous ventilation. The King LTS (reusable) and King LTS-D (disposable) devices released in 2003 added an opening in their distal tip for esophageal access. Like the pLMA, the King LTS and LTS-D allow for passage of a gastric tube. The King LT family of devices has gained popularity in emergency medicine and prehospital care settings [52,55,56].
Laryngoscopy, Laryngoscopes, and Tracheal Tubes
Initial visualization of the larynx occurred outside the operating room, and was prompted by curiosity concerning voice production. In 1829, Benjamin Babington, a British physician, used sunlight reflected off a hand mirror and a small mirror attached to the end of a spatula to view the vocal cords in action. A colleague called Babington’s device a “laryngoscope” while Babington called it a glottiscope. He reported his findings to a disbelieving Hunterian Society, his peers certain that the vocal cords could not be seen in a living person [57,58].
A quarter century later, Manuel Garcia, a famous singing teacher at the Royal Academy of Music in London, wanted to see his own vocal cords. Benefiting from naturally obtunded gag reflexes, Garcia performed laryngoscopy of his active vocal cords using a dental mirror positioned in his oropharynx and a hand mirror to simultaneously reflect light and visualize the image in the dental mirror. He meticulously documented observations of his vocalization and that of others, and in 1855, presented his results to the Royal Society. This time, indirect laryngoscopy was met with interest [33,58–60].
After witnessing Garcia’s demonstration, Austrian neurologist, Ludwig Turck, began using laryngeal mirrors in his hospital practice. Attaining indirect laryngoscopy was cumbersome, often requiring cooperative patients to assist with a self-applied tongue depressor and/or forceps, while Turck positioned the mirrors. Turck claimed to see the mucosal surface of the posterior tracheal wall, the bifurcation of the trachea and even the first six circular cartilages of the bronchi in optimal cases. His colleagues considered these assertions exaggerated [33,58–60]. Hungarian physiologist and physician, Johann Czermak, expanded the studies of indirect laryngoscopy, popularizing the technique. His work introduced laryngoscopy as a new branch of surgery [58–60].
Tracheal Intubation
Indirect laryngoscopy allowed a view of the glottis but did not immediately lead to tracheal intubation. After Avicenna, tracheal intubation was mostly performed to aid in the resuscitation of drowning victims and to treat patients with diphtheria. In 1796, Herholdt and Rafn described blind digitally assisted tracheal intubation to resuscitate drowning victims, their work receiving little notice until the mid-1980s [61]. In 1788, the British physician, Charles Kite, described a metal orotracheal tube bent like a male urethral catheter, passed blindly through the glottis, and then connected to a bellows [4,62,63]. Encouragement by European and British medical societies in the late 18th century prompted development of new airway devices for artificial ventilation. These were widely distributed, primarily for use in victims of drowning. However, experiments performed by Jean Leroy d’Etoilles in the late 1820s, revealed the danger of developing a tension pneumothorax from forced inflation of the lungs caused by the use of the bellows in the resuscitation of drowned victims. Although Leroy modified the bellows to be safer, the Royal Humane Society retracted their support of artificial ventilation, temporarily delaying the advancement of airway management and the use of positive pressure ventilation [11,64].
Scottish surgeon William Macewen pioneered elective tracheal intubation for anesthetic delivery [60,65–67]. In 1878, he practiced blind digitally assisted orotracheal and nasotracheal intubation on cadavers. He abandoned nasotracheal intubation as impractical, probably because of the size and stiffness of his metal tubes [33,60,66]. He hoped to deliver anesthetic without contaminating the surgical field and to protect the lungs from aspiration without performing a tracheostomy. Orotracheal intubation would also resolve the conflict between surgeon and anesthesiologist for access to the mouth and airway [1,33,61,66]. He used his index finger to depress the epiglottis and guide a rigid, curved brass tracheal tube of his design [1,61,68] over his finger and into the larynx [33]. In July 1878, he performed his first tracheal intubation in a patient undergoing removal of an epithelioma of the tongue, right anterior pillar of fauces and posterior pharyngeal wall [33,60,68]. Topical anesthesia awaited discovery, and Macewen practiced several awake tracheal intubations in the unanesthetized patient before the day of surgery [33,61,66,68]. Other than bouts of coughing both during and immediately after placement of the tracheal tube, the patient tolerated the intubation, and Macewen proceeded with anesthesia [33,60]. With sponges packed into the pharynx to prevent aspiration, the extensive surgical procedure, including sawing through the lower jaw, concluded successfully [1,33,60,66]. The patient maintained spontaneous ventilation and showed no signs of pain [60]. As did others, Macewen advocated viewing the glottis via indirect laryngoscopy to determine oropharyngeal pathology before performing intubation.
Between 1878 and 1880, Macewen described two additional successful tracheal intubations for glottic obstruction [60,66,68]. His fourth patient, the second scheduled for anesthesia and surgery following tracheal intubation, resulted in a fatality [33,60,68]. Like the first case, the patient had an epithelioma of the tongue’but also had bronchitis [60]. Although Macewen successfully practiced awake intubations in the patient before the day of surgery, on the day of surgery the patient pulled out the tube and pleaded for its placement after induction with chloroform [60,66,68]. The patient struggled with induction, and once achieved, Macewen inserted the tube [60,66]. Within minutes, the radial pulse and spontaneous ventilation ceased [36,60,66]. An autopsy revealed cerebral hemorrhage, probably stress-related [36,66]. This was the last tracheal intubation reported by Macewen [33,60,66,68].
Joseph O’Dwyer, a physician at the Foundling Hospital in New York, found the mortality from diphtheria to be unacceptable. He studied autopsies to define alternatives to tracheostomy [2,60,69], and from these investigations developed the O’Dwyer endolaryngeal tubes. He first used these clinically in 1882 and by 1885 had perfected his skills and tubes. The metal tubes came as a set in a range of sizes and featured a collar, designed to rest within the pharynx against the false vocal cords, to prevent the tube from advancing down the trachea [1,68]. O’Dwyer is credited with publicizing tracheal (actually endolaryngeal) intubation, albeit not for anesthetic purposes [4,60].
In 1885, a young woman with known laryngeal stenosis, approached O’Dwyer requesting an alternative to tracheostomy. O’Dwyer accommodated her request, by inserting one of his tubes. Similar cases followed [33]. In 1887, the New York physician George Fell modified his breathing apparatus supplying positive pressure ventilation using a bellows and mask, by attaching it to a tracheotomy tube. Fell’s apparatus was specifically used to treat narcotic overdose. Learning of Fell’s modifications, O’Dwyer altered his tube to permit controlled ventilation with Fell’s device, hence the Fell-O’Dwyer Apparatus [1,4,68]. (Fig. 54.7) Theodore Tuffier, a French surgeon, and Rudolph Matas, a Texas and Louisiana surgeon, made further modifications for positive pressure ventilation during intrathoracic surgery [1,33,68].
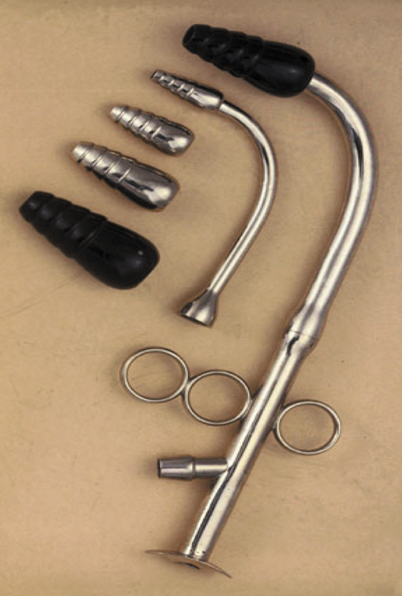
Fig. 54.7
Modified O’Dwyer’s tubes intended for insertion into the larynx. Combined with Fell’s foot bellows and air hose, they comprised the Fell-O’Dwyer apparatus. (Courtesy of Science Museum London/Science and Society Picture Library, London, England)
In 1893, Karl Maydl, Professor of Surgery at the University of Prague, suggested that intubation of the larynx, and packing gauze around the glottic opening would prevent aspiration of blood [33,66]. Although Macewen had advocated this technique, little was known of Macewen’s work [33]. Maydl knew however, of O’Dwyer’s work and adopted O’Dwyer’s intubation techniques for otorhinolaryngological surgical procedures [1,33,66].
In 1896, German surgeon Franz Kuhn found that rigidity of existing tracheal tubes was limiting [68,70]. Using coiled metal similar to that found in spouts of old gasoline cans, he made a tracheal tube that was flexible, yet resisted compression [36,68,70]. Like Macewen, Kuhn performed blind, digitally-assisted awake tracheal intubation with the aid of a rigid curved introducer [60,68,70]. His method was easier than Macewen’s because topical anesthesia with cocaine had been discovered. Topical anesthesia allowed great advances in nasotracheal intubation after World War I [1,66,70].
Kuhn surrounded his tracheal tube with a supralaryngeal flange to prevent over-insertion [60,66,68]. Kuhn acknowledged the existence of tracheal tube cuffs but preferred to pack the larynx with oil-soaked gauze [30,36,60,68] which also helped secure the tube in place [60]. Kuhn pioneered suctioning of tracheal blood and secretions [68,71]. Most tubes at this time were narrow and were used for insufflation (blowing air plus ether or chloroform vapor into the trachea). He preferred larger tubes because they allowed bidirectional respiration. During World War II, English prisoners of war found Kuhn’s technique and tracheal tubes beneficial. One advantage touted for this technique was that batteries which were in short supply were not required [30,60].
In 1902, Kuhn pioneered nasotracheal intubation in living patients [66,69,72]. He argued that the anatomy of the nasal route provided a more direct approach to the glottic opening [66], prevented the tracheal tube from being bitten, and left the mouth unoccupied [66,68]. However, he ultimately viewed orotracheal intubation as better suited to his patients’ needs, probably because the metallic composition of his tubes might traumatize the nasal mucosa despite its flexibility [66].
Before 1900, placement of a tracheal tube usually required a blind digitally assisted orotracheal intubation, a difficult maneuver requiring extensive practice and involving significant risk to the intubator whose finger might be bitten and become infected [2,36]. Widespread use of blind nasotracheal intubation awaited development of soft flexible tracheal tubes. The introduction of direct laryngoscopy during the 1910s overcame these limitations.
Laryngoscopy
Direct laryngoscopy originated with Alfred Kirstein’s 1895 article describing his “autoscope” (Fig. 54.8) [58,73,74]. A colleague’s accidental placement of an esophagoscope into the trachea sparked Kirstein’s interest. Initially, he directly visualized the larynx by shortening an esophagoscope and attaching an electric lamp to the handle, deflecting light down the lumen by a prism taken from Caspar’s electroscope. The enclosed “O” shape of this original autoscope limited the view of the glottis, leading to a redesigned blade with an open crescent shape and an improved distal width and thickness for direct or indirect positioning of the epiglottis [33,59,68]. Notwithstanding this work, Kirstein never performed tracheal intubation or bronchoscopy. But in 1897, his student, Gustav Killian, used the autoscope to extract a foreign body from a patient’s bronchus. This case and his subsequent work made Killian the “father of bronchoscopy.” [59]
By 1910, Chevalier Jackson had invented a U-Shaped direct laryngoscope (Fig. 54.9) for examining the esophagus and airway. He became renowned for his ability to remove foreign bodies from the airway, and patients came from all over the world to obtain his services’often performed at little to no cost to the patient. In 1912, Charles Elsberg used the Jackson laryngoscope for tracheal intubation and insufflation. This prompted Jackson to publish a paper advocating examining vocal cords for pathology before intubation and ascertaining the proper size of tube to be used. His work furthered the popularity that laryngoscopy gained over the succeeding decades [15,75,76].

Full access? Get Clinical Tree