Fig. 38.1
Degrees of nerve injury that leave the nerve in continuity . The mildest injury is neurapraxic, which represents a loss of electrical function with little, if any, anatomic change. More severe is an axonotmetic injury with disruption of the axonal membrane but minimal disturbance to connective tissue. Wallerian degeneration of the axon occurs. Progressively more severe is the mild neurotmetic injury (mild connective tissue damage) and then severe neurotmetic injury (severe connective tissue damage and severe scarring). The latter will usually require a complete surgical repair
The anesthetic considerations associated with operative recording from peripheral nerve are minimal. General anesthetics do not affect peripheral nerve directly and the CNAP is minimally affected by halogenated agents, nitrous oxide, and intravenous anesthetics . If the patient becomes significantly hypothermic, there may be a mild to moderate decrease in nerve conduction velocity, but the amplitude of the CNAP would not be significantly affected. In the protocol described here, the stimulating electrodes and the recording electrodes are both placed on peripheral nerve. Therefore, neuromuscular junction blockade would not affect the CNAP. In some instances, though, it is reassuring to see evoked movement when stimulating peripheral nerve. It is not advisable to use evoked movement instead of a CNAP to evaluate nerve function. The reason for this is described elsewhere in this chapter.
The method of stimulating and recording CNAPs is straightforward and requires little experience to become proficient [12]. Since the size of the CNAP is considerably larger than the brain potentials recorded in operative monitoring, the process of signal averaging is not required. The necessary instrumentation is commonly available. Either equipment used for operative monitoring or EMG instrumentation can be used successfully. There are very few anesthetic considerations that would affect peripheral nerve recordings. Perhaps the most important would be the use of a tourniquet on the limb from which recordings will be made. If a tourniquet is used, the tourniquet should be released at least 20 min prior to any recordings. This would provide ample opportunity for perfusion to restore normal function prior to any recordings.
Stimulation of nerve through electrodes in direct contact is safe and effective. Stimulation parameters vary somewhat depending upon the size of the nerve being studied. For example, the normal, healthy ulnar nerve at the elbow requires an intensity of approximately 4–6 mA (8–10 V) when pulse duration is very short (<0.1 ms). We use these very-short-duration stimulus pulses to determine the presence of larger, myelinated fibers that can mediate motor function. Very small, unmyelinated fibers will not respond to these short-duration pulses [14]. However, nerve that has been chronically compressed or nerve that is highly scarred may require very high levels of stimulation at this pulse duration [11, 13]. In this case, it may be necessary to use intensities as high as 30–40 mA (60–80 V) at very short pulse durations (0.02 ms) to insure stimulation of fibers embedded in scar [14]. It should be noted that very-short-duration stimulus pulses (on the order of 0.02 ms) preferentially activate larger, myelinated fibers that will mediate motor function. While there may be some concern for possible damage to nerve stimulated directly at these intensities, it should be pointed out that even with these high intensity levels there is little risk of damage to the nerve itself. Long periods of stimulation may induce a variety of factors that damage nerve. However, damage produced by short-term stimulation results primarily from the heat due to electrical currents or from electrolysis, the “plating” effect of salt deposition. Both of these processes require significant electrical p ower. One can easily see that the average electrical power, expressed in watt-seconds, conveyed by 30 mA of current flowing for 0.1 ms and a repetition rate of 5 pulses/s is very small and very little heat is produced. (Power = I 2 RT = (current)2 × resistance × time. Assuming current = 30 mA, resistance = 2000 Ω, and 5 pulses at 0.1 ms, P = 0.032 × 2000 × 0.0005 = 0.0009 W-S of average power delivered under these conditions). There is also very little electrolysis over the short term (several seconds of stimulation) because of a small amount of power. Kline and Hackett [11] histologically examined peripheral nerves from primates used in nerve research and have never found evidence of damage with stimulation well beyond these intensities. Further, in operative studies on human subjects, there have been no reports of postoperative nerve dysfunction as a result of this stimulation [11]. Under the conditions described here, electrical stimulation remains a safe mechanism for the evaluation of peripheral nerve.
Recording the CNAP from normal peripheral nerve is also very easy since the response is large in size (0.2–1 mV). Since the response is large in size, it does not require averaging to enhance the response. In fact, averaging is to be discouraged since it could lead to a deceptive large appearance of a response when, in reality, the response is extremely small. In the absence of averaging, it has been determined that a minimum of 4000 fibers of adequate size must be present in order to produce a clear CNAP [15]. This number of fibers has the potential to mediate significant function and therefore the appearance of a clear CNAP is a good prognostic indicator.
Figure 38.2 illustrates the type of electrodes that I have successfully used to stimulate and record from peripheral nerve. Stainless steel electrodes represent a good cost-effective means of providing stimulation.
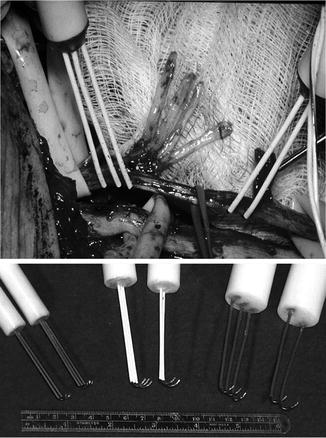

Fig. 38.2
Electrodes used for the stimulation and recording of peripheral nerve CNAPs. (Top) Electrodes applied directly to nerve. The stimulating electrode is a tripolar electrode and the recording electrode is bipolar (see text). Note that the electrodes can be used over just a few centimeters of nerve length. (Bottom) Selection of different size electrodes to be applied to various size peripheral nerves. The gap between contacts can be varied with the size of the nerve to be studied
Of great importance, however, is that silver electrodes not be used to provide stimulation. When silver electrodes are used, there is the possibility that silver salts could be deposited and these are very toxic. The stimulation of an intact nerve in situ presents an unusual set of circumstances [10]. The application of a potential difference with electrodes along the length of a nerve creates two current paths. First, there is a current in the gap between the two electrodes; second, there is a current flowing away from the electrodes, through the nerve, through the tissues of the body, and back to the other electrode. The latter current path is problematic as a current along the length of the nerve will be recorded together with the CNAP. This produces excessive stimulus artifact and, when the distance between stimulating and recording electrodes is short, may obscure the action potential. A solution to this problem is to use a tripolar electrode with the outermost terminals connected together. In this arrangement, the application of a potential difference between the inner and outer electrodes still produces two currents; neither involves the whole nerve. The reduction in stimulus artifact achieved in this way permits even very small action potentials in injured or regenerating nerves to be seen. The use of a tripolar electrode also serves to limit the spread of stimulating current longitudinally along the length of nerve [10]. The latter is undesirable as misleading observations are made when sufficient current spreads several centimeters away from the stimulating point. While there may be no viable axons at the point of stimulation, the spread of current may excite distant viable axons, leading to the production of a CNAP. This phenomenon could lead to the false inference of excitable axons at the point of stimulation. A further exposition of this problem has been published previously [10].
For recording, a bipolar pair of electrodes is used. The configuration of the recording electrodes is also important to the successful evaluation of the electrical characteristics of peripheral nerve. Two stainless steel wire electrodes along the length of the nerve with a separation of at least 3–5 mm work well. If these electrodes are moved too close together, the amplitude of the recorded action potential will be reduced; thus, a larger distance of 5 mm is recommended, particularly for larger nerves. Further description of the features of recording electrodes has also been published previously [10].
Figure 38.3 demonstrates the appearance of the normal CNAP and to contrast this with the appearance of a very abnormal CNAP. The normal CNAP shows relatively large amplitude and a very short duration. The short duration indicates that the conduction velocities of the numerous fibers within the nerve are very similar. By contrast, the low amplitude seen in the abnormal CNAP indicates fewer fibers present and these show extensive temporal dispersion due to wide variations in conduction velocities [16]. These findings will be referred to in the practical illustrations.
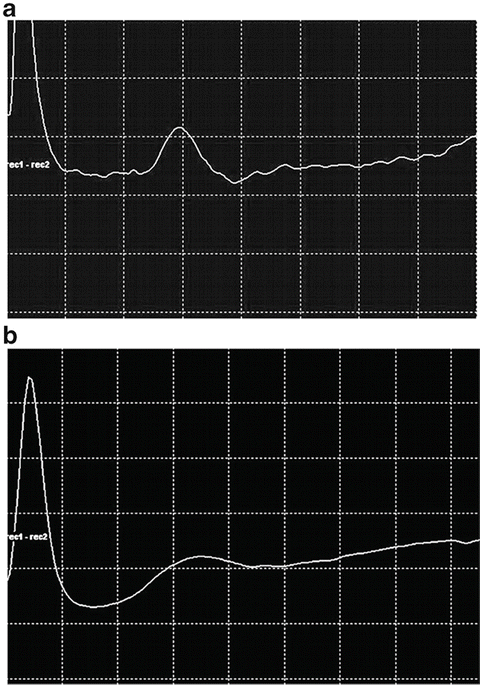

Fig. 38.3
Normal CNAP (a) recorded over a length of 15 cm through the brachial plexus. Note the shorter duration of the CNAP when compared to the abnormal response below. Calibration: 200 μV/div, 1 ms/div. Abnormal CNAP (b) recorded over a length of 7 cm through the brachial plexus. The lower amplitude is a reflection of fewer fibers and the broad base indicates dispersion of conduction velocities. Calibration 200 μV/div, 1 ms/div
It is very important to note that the patient’s history should be placed into context with the operative peripheral nerve recordings. An example of this relevance would be the case of a stretch injury to the brachial plexus. When the more proximal elements are stretched, there is the possibility of an avulsive injury to the nerve roots. In a severe avulsion, the nerve roots are torn loose from the spinal cord in such a way that the dorsal root ganglia are separated from the spinal cord leaving them connected to the sensory fibers of peripheral nerve. In this case, the sensory fibers of peripheral nerve will remain normal, but are disconnected from the spinal cord. The motor fibers of the same root are separated from their cell body in the anterior horn of the spinal cord and these fibers will undergo Wallerian degeneration. With operative stimulation and recording during brachial plexus exploration, a large CNAP such as that seen in Fig. 38.5 becomes the indicator of a poor prognosis. It shows normal sensory fiber activity in the clinical situation with no motor activity. These are the characteristics of an avulsive injury. In this case, a repair would not be effective.

Full access? Get Clinical Tree








