81 Sudden Cardiac Death
Implantable Cardioverter-Defibrillators
Since its initial development in the 1970s1 and its introduction to clinical practice in the 1980s,2 the implantable cardioverter-defibrillator (ICD) has revolutionized the management of patients with or at risk for life-threatening ventricular arrhythmias. Large randomized controlled trials3–13 have shown that these devices prevent death from ventricular tachycardia (VT) or ventricular fibrillation (VF). Device-based treatment of recurrent VT or VF is the initial treatment of choice for many patients who have experienced or are at high risk for experiencing these rhythm disturbances.14 Device complexity makes a detailed understanding of ICD technology challenging for practitioners, but a general understanding of these devices and associated clinical problems is increasingly important because of their widespread use.
 Epidemiology of Sudden Cardiac Death
Epidemiology of Sudden Cardiac Death
Sudden cardiac death, arbitrarily defined as death from a cardiac cause occurring within 1 hour of cardiovascular symptom onset or without preceding symptoms,15 is a major public health problem responsible for approximately 450,000 deaths annually in North America alone.16 Out-of-hospital cardiac arrest carries a dismal prognosis, with reported rates of survival to hospital admission of 5% to 10% and minimal improvement in survival rates over the past several decades.17 This poor outcome occurs despite public health efforts to improve public recognition of cardiac symptoms and shorten the time to therapy by means of bystander cardiopulmonary resuscitation (CPR) and better access to emergency medical services.18 Among patients who survive to hospital admission, mortality and morbidity remain exceedingly high,19,20 highlighting the need for preventive efforts.
A significant proportion of sudden cardiac deaths are due to a treatable arrhythmia such as VT or VF,18,21 with the remainder being due to asystole or pulseless electrical activity (PEA). In autopsy studies, a majority of sudden cardiac death victims have pathologically apparent structural heart disease, particularly coronary atherosclerosis.22 In many cases, recent unstable coronary disease can be demonstrated by pathologic evidence of recent plaque rupture, with or without thrombosis.23 In cases in which cardiac monitoring was in place at the time of death, arrhythmia is commonly present.24
A significant proportion of sudden cardiac death occurs in patients without previously identified cardiac disease.19,25 Currently there is no feasible means of screening the population at large to identify all individuals who are at risk for this catastrophic event. Prediction and prevention strategies have therefore focused on identifying patients with clinical characteristics that place them at particularly high risk for sudden cardiac death.26,27 From the public health perspective, the most important conditions that predispose to a high risk of sudden cardiac death include cardiovascular risk factors, coronary artery disease, and left ventricular (LV) dysfunction of ischemic etiology and a variety of hereditary conditions that are listed in Box 81-1.
Approximately 50% of deaths in patients with heart failure are sudden.27,28 The majority of these are due to ventricular tachyarrhythmias.24 However, asystole and PEA are more common modes of sudden unexpected death in patients with end-stage heart failure.29 Among the factors that predict sudden cardiac death, severity of LV systolic dysfunction and age are by far the strongest predictors.30–32 Trials of ICD therapy have largely focused on patients with LV dysfunction, coronary disease, and spontaneous or inducible ventricular arrhythmias.33
 Prevention of Tachyarrhythmic Sudden Cardiac Death: Non-Device Therapy
Prevention of Tachyarrhythmic Sudden Cardiac Death: Non-Device Therapy
Previously, antiarrhythmic drugs were the cornerstone of treatment and prevention of recurrent VT and VF. However, it is recognized that these drugs are intrinsically hazardous, given their arrhythmogenicity and other adverse effects.34–39 Currently, antiarrhythmic drugs retain a primary role in patients with other conditions for which these agents are indicated (e.g., concurrent atrial fibrillation) or to decrease the frequency of ICD shocks. In this instance, D–L sotalol, dofetilide, or amiodarone are most often utilized.
Although class IC antiarrhythmic drugs, including encainide, flecainide, and moricizine, are effective at suppressing ventricular ectopy, they have been shown to significantly increase mortality in the landmark Cardiac Arrhythmia Suppression Trials.34,36 D-Sotalol, a pure class III antiarrhythmic agent, was evaluated in a randomized controlled trial and, similar to class IC agents, was found to increase mortality.40 The L-isomer that confers the beta-blocking effect may attenuate this hazard.41 Dofetilide, a class III agent, has been shown to be safe in patients with symptomatic heart failure and LV dysfunction when initiated in the hospital.42 In contrast, dronedarone, a newer antiarrhythmic agent, was found to increase mortality in patients with advanced heart failure.43 Thus, its role in the management of arrhythmias in patients with heart failure is unclear. Newer antiarrhythmic agents including azimilide, celivarone, and vernakalant are under investigation.
Amiodarone is the only available empirical choice for arrhythmia prevention in patients with heart failure or LV dysfunction. Several trials have shown decreased risk of death among patients treated with amiodarone after myocardial infarction (MI).35,44 Among patients at risk for arrhythmic death, a meta-analysis of controlled trials showed a reduction in total, cardiac, and sudden cardiac deaths with amiodarone therapy.45 In patients with heart failure, emperic amiodarone does not increase the risk of death (in contrast to class IC agents).10,37
Guided approaches to antiarrhythmic drug choice have also been evaluated.46 This can be done noninvasively using serial ambulatory cardiac monitoring to assess the response to specific drug choices, or invasively using serial programmed electrical stimulation to evaluate the drug effect on inducibility of VT or VF. Both approaches have been evaluated and can predict response to medical treatment reasonably well.47–49
The high recurrence rates of VT/VF and medication-related adverse events limit both empirical and guided therapies.38,50,51 For example, although amiodarone is the most effective antiarrhythmic drug for preventing the recurrence of VT and VF, a substantial proportion of patients (up to 20%) treated with amiodarone are unable to continue therapy in the long term owing to cumulative side effects, recurrent arrhythmia prompting a change in therapy, or death.38,52
Medications other than antiarrhythmic drugs have also been evaluated. Beta-blockers clearly reduce the risk of death among patients with recent MI53,54 and LV dysfunction,55–57 and it appears that approximately 50% of this decreased risk is due to reductions in sudden death.53 Beta-blockers have been shown to suppress ventricular arrhythmias among patients at elevated risk58,59 and may reduce death when used as primary antiarrhythmic therapy.60 Use of HMG-CoA reductase inhibitors (“statins”) has been associated with a lower risk of sudden death compared with nonuse in several studies.61–63 However, there are no large randomized controlled trials to confirm this finding. Trials of angiotensin-converting enzyme inhibitors and angiotensin receptor blockers in patients with heart failure and coronary disease have shown reductions in the risk of sudden cardiac death in these populations.64 Omega-3 fatty acids (“fish oils”) appear to reduce the risk of sudden cardiac death in epidemiologic studies41,65 and in prospective randomized trials.66,67 A report68 has raised methodological concerns on one of these prospective trials.67 A recent randomized trial designed to look at the effect of highly purified omega-3 fatty acids on secondary prevention of sudden cardiac death after MI showed no benefit,69 possibly related to a low event rate in both groups. Aldosterone inhibition (spironolactone, eplerenone) has also been shown to be useful in preventing sudden death in patients with heart failure and after MI. While more widespread use of automated external defibrillator (AED) therapy was hoped to have a significant benefit in the prevention of sudden death, the Home AED Trial (HAT) failed to show a survival benefit of an AED in addition to CPR versus CPR alone among a large group of patients with a history of prior MI.70
Catheter ablation and surgery are often effective in preventing recurrent VT in patients who are difficult to treat by other means. Both techniques attempt to destroy or “ablate” involved myocardial tissue to interrupt reentrant VT circuits, thus preventing the development of sustained arrhythmias. In the past, VT surgery was considered a primary form of therapy in experienced centers, as it could offer a cure to patients with few other therapeutic options.71–74 Currently, VT surgery has a limited role owing to very high operative morbidity and mortality and improved nonsurgical approaches. Catheter ablation is a technique using intracardiac catheters to induce VT, map the pathologic circuits or substrate, and ablate small areas of involved myocardial tissue with radiofrequency energy.75,76 Ablation may carry a lower procedural risk than open surgical approaches, but a substantial number of patients have recurrent ventricular arrhythmias.74,77 Thus, it is presently not a replacement for ICD therapy. VT related to ischemic heart disease may be difficult to manage with catheter ablative procedures,77,78 owing to multiple pathologic intracardiac circuits. Like antiarrhythmic drugs, VT ablation is used as an adjunct to decrease the frequency of ICD therapy rather than a means to prevent sudden death.79
Revascularization is of primary importance in patients with coronary artery disease and malignant ventricular arrhythmias. One study evaluated the role of ICD in patients undergoing coronary artery bypass grafting (CABG) and showed no benefit in this population.80 Other studies have demonstrated an association between CABG and decreased risk of sudden death.11,81,82 Two randomized trials of ICD therapy early following MI found no difference in mortality with usual medical care versus an ICD9,11 (see Clinical Trials).
Lifestyle factors have been associated with lower risks of sudden death. Tobacco avoidance, exercise, moderate alcohol consumption,83 and a diet rich in fish65 have all been shown to be protective, and lifestyle modification programs may prevent sudden death.84,85
 Implantable Cardioverter-Defibrillator Therapy
Implantable Cardioverter-Defibrillator Therapy
Device Basics
The pulse generators of early devices were large (approximately 250 cm3) and required surgical implantation in the abdomen. Leads were large (150 to 180 cm2) epicardial pads placed via a thoracotomy. Separate epicardial screw-in sensing leads were also required. Implantation was associated with significant perioperative morbidity and mortality. Rhythm analysis was rudimentary and relatively insensitive. Only medium- or high-energy shock therapy was available, and data storage capacity was limited to information regarding the number of shocks. When intracardiac electrogram storage and analysis became available, it was apparent that inappropriate shocks, predominantly for atrial fibrillation, were common.86,87
The initial primary purpose of the ICD was to detect VT and VF and terminate these arrhythmias with effective defibrillation. Reports of early experiences suggested a substantially lower annual mortality among ICD recipients versus similar historical comparative groups.88 Recent refinements in ICD technology have improved the safety and tolerability of the devices substantially, but effective defibrillation remains the crucial lifesaving feature.
Current devices are much smaller, allowing subpectoral or subcutaneous implantation. Using nonthoracotomy lead systems, implantation methods are identical to permanent pacemaker implantation. Local anesthetic with mild sedation is used for implantation; heavy sedation or a brief general anesthetic is needed to test defibrillation thresholds. Operative mortality for nonthoracotomy systems is less than 0.5%.89 The risk of defibrillator-threshold or safety-margin testing is estimated to be less than 0.05% for death or stroke and less than 0.2% for necessitating prolonged resuscitation, based on a large series of registry data.90 This risk is higher in patients with severe LV dysfunction where even a brief induction of VF can have persistent and detrimental efftects.90,91 Obesity, cachexia, limited vascular access, pulmonary hypertension, anticoagulation, bleeding disorders, and vascular or cardiac anomalies may increase the technical challenge of implantation. Tricuspid valve prosthesis or significant tricuspid valvular disease may preclude use of endocardial lead systems. Features of contemporary ICD systems are listed in Table 81-1.
TABLE 81-1 Features of Current Implantable Cardioverter-Defibrillators
| Size | 30-45 cm3 |
| Weight | 70-100 g |
| Batteries | Low-resistance lithium or silver vanadium for charging defibrillation capacitor; separate battery for pacing functions |
| Leads | Steroid-eluting, silicone- or polyurethane-coated, 4-9F (1.3–3 mm) caliber, depending on type; ports for ventricular, atrial, left ventricular (coronary sinus), and superior vena cava leads |
| Output, charge | 30-39 J (delivered), 750-800 V |
| Battery life | 3-8 yr, depending on manufacturer, device, and use |
| Arrhythmia detection | Rate-based; enhanced ventricular tachycardia detection features vary by device and manufacturer |
| Arrhythmia management | Defibrillation with biphasic waveform, low-energy cardioversion, antitachycardia pacing (ATP) features; atrial therapies, including ATP and cardioversion; bradycardic ventricular and dual chamber pacing; biventricular pacing |
| Storage capabilities | Device and lead identification, implantation date, physician contact; arrhythmia event data, including date and time, onset, heart rate, therapies delivered, shock counters, rate histograms, electrograms, marker channel; pacemaker functions, including pacing thresholds, lead impedances, R-wave and P-wave amplitude, percent pacing, heart failure diagnostic information |
| Programmable functions | Pacing parameters, tachyarrhythmic therapies, tiered therapy algorithms; many other refined programmed functions vary by manufacturer |
Complications Related to Transvenous Icd Placement
Although placement of a transvenous ICD system is routine in many centers, complications related to system placement do occur. Common procedural complications are summarized in Box 81-2.
Therapeutic Functions
Bradycardia and Pacing
Patients with significant heart failure commonly have symptomatic bradycardia due to conduction disturbances, inadequate chronotropic responses, and medications that induce bradycardia.29 Moreover, postcardioversion and postshock bradycardia is common among ICD patients. To meet these needs, all current ICDs have pacing capabilities. ICD systems are available with ventricular, dual-chamber, or biventricular pacing modalities.
Although patients who receive an ICD may have an indication for single or dual-chamber pacing, there are concerns about the potential adverse effects of right ventricular pacing. One major trial showed that atrioventricular sequential pacing at a rate of 70 beats per minute was associated with higher rates of heart failure, hospitalization, or death when compared with backup ventricular pacing at 40 beats per minute.92 This effect was ascribed to the untoward hemodynamic effects of right ventricular pacing. Other studies have supported this finding.93 Furthermore, pacing can precipitate ventricular tachyarrhythmias in some patients.94 Thus, the pacemaker backup rate should be turned down to the lowest acceptable rate in patients with LV dysfunction.
Biventricular pacing, or resynchronization therapy, is a pacing modality incorporated in some devices. The intent of biventricular pacing is not to treat bradycardia per se. Instead, it coordinates synchronous left and right ventricular contraction.95 In the presence of left bundle branch block or right ventricular pacing, the interventricular septum moves rightward during systole. This decreases the contribution of septal contraction to LV output, leading to less efficient LV systolic function. Biventricular pacing coordinates left and right ventricular contraction to minimize this effect. The left ventricle is approached through the venous system (coronary sinus) using specially designed leads to allow epicardial LV pacing.
Several studies evaluated biventricular pacing in patients with advanced symptomatic heart failure (NYHA III-IV) and significant intraventricular conduction delay (QRS duration ≥ 120 milliseconds).13,96–98 Results show improvements in symptoms, exercise tolerance, and quality of life99 among a significant proportion of these selected patients. A survival benefit has also been demonstrated (Table 81-2).13,98,100 More recent studies looking at less severe heart failure (NYHA I-II) have demonstrated a decrease in symptomatic heart failure episodes and favorable LV remodeling without a survival benefit.101,102 Another trial in less symptomatic patients (RAFT) will be reported later this year.103 Heart failure patients with QRS durations less than 120 milliseconds have not been shown to benefit from cardiac resynchronization therapy (CRT),104 but studies addressing methods other than QRS duration are ongoing (EchoCRT).
Tachyarrhythmia Detection
The primary method of detecting sustained VT is assessment of ventricular rate and duration of the tachycardia. Therapy is delivered for persistent heart rates exceeding a cutoff that is manually programmed. Different algorithms can be programmed for different rates (Figure 81-1). The major limitation of an exclusively rate-based rhythm analysis is that tachycardias other than VT (e.g., supraventricular tachycardia [SVT]) cannot be distinguished by rate alone.
Enhanced arrhythmia detection features in current dual-chamber systems enable sensitive and specific detection of VT and VF, decreasing the occurrence of inappropriate therapies.105–110 Onset criteria allow the distinction between sinus tachycardia, which generally has a gradual onset, and VT, which is abrupt. Rate stability criteria distinguish irregular atrial fibrillation from VT. Devices also use the intracardiac electrogram to identify VT. Analysis of QRS morphology during tachycardia compared with a sinus rhythm template is a feature found in many single and dual-chamber devices. Dual-chamber devices use atrial lead sensing to evaluate the relationship between ventricular and atrial activity to distinguish supraventricular tachycardia from VT.109 Judicious use of these features is highly sensitive for VT and specific for discrimination of SVT. Another method used to limit ICD shocks is to increase the number of intervals to detect before the device treats the arrhythmia. This prevents unnecessary therapies for arrhythmias that would otherwise have self-terminated, but with the tradeoff of an increased likelihood of syncope from the delay in administration of therapy.111,112 Trials assessing the utility of delayed detection are ongoing.113 Combining multiple algorithms to withhold unnecessary ICD shocks (SVT, noise, and more frequent use of antitachycardia pacing [ATP]) also holds promise.
Tachyarrhythmic Therapies: Tiered Therapy Algorithms
Using the methods outlined previously, the ICD detects arrhythmias and administers therapies as programmed. In contrast to early devices, current ICDs can deliver therapies other than defibrillation, including lower-energy cardioversion and ATP. Some devices also have atrial antitachycardia and cardioversion features, whose clinical benefit remains to be proven.114,115 A tiered therapy algorithm (see Figure 81-1) uses different “zones” of detection to preferentially administer ATP or shocks depending on the rapidity of the detected rhythm.
High-energy defibrillation is the primary and most important function of the ICD. It is highly effective for VF or very rapid VT. Other therapies are intended to abort hemodynamically tolerated VT to obviate a painful high-energy shock. Typically, tachycardias above 200 beats per minute are promptly treated with high-voltage shocks. If the ICD detects a ventricular rhythm in the “VF zone,” the battery charges the capacitor, which then discharges, or “shocks,” if a second rhythm analysis confirms ongoing VF. Current is transmitted between the right ventricular lead and either the device itself (“active” or “hot” can) or other electrodes or coils.116 The current passes through ventricular myocardium and depolarizes a proportion of myocytes with 27 to 35 J of energy, depending on the manufacturer and configuration. This depolarized mass of myocardium interrupts the fibrillating electrical wavefronts and terminates VF. After each therapy, the device reinstates a diagnostic algorithm to detect ongoing VT/VF. If the arrhythmia persists, the capacitor recharges, discharges, and continues this cycle of behavior until another rhythm is detected or the therapies are exhausted (e.g., 4–6 consecutive high-energy shocks for a single episode).
The major limitation of high-energy shocks is the associated discomfort experienced if the patient remains conscious during the arrhythmia. Many patients report that shocks are painful and are associated with fear, embarrassment, or other unpleasant emotions.117 Quality of life is significantly impaired in patients who receive ICD ≥ 5 shocks, from either the shock itself or the health condition necessitating the shock.118,119 It is important to prevent ICD shocks, given that both appropriate and inappropriate shocks have been associated with an increased risk of death.120 However, it is unclear whether the shock itself is responsible for the increased risk of death, or changes in the underlying condition both increase the occurrence of arrhythmias and the risk of death.
Low-energy cardioversion is an established method of terminating hemodynamically tolerated VT, with a success rate greater than 80%.121,122 When the device detects a rhythm in the VT zone, it charges the capacitor and delivers a lower-energy shock synchronized to the R wave (see Figure 81-1). Energy outputs of 0.1 to 5 J can terminate some VT events. Patient discomfort increases substantially with increased output, particularly above 0.5 to 1 J. Above 5 to 10 J, no benefit is gained with low-energy cardioversion versus defibrillation in terms of patient comfort, although avoidance of high-energy output may prevent long-term device dysfunction123,124 and prolong battery life. The other major risks of low-energy cardioversion are acceleration of the tachycardia rate, which occurs in up to 10% of cases, and delay of definitive therapy.122 Less commonly, cardioversion can cause the rhythm to degenerate to polymorphic VT or VF, necessitating defibrillation. ATP is generally favored over shocks to limit the problem.
ATP, when effective, is ideal therapy for terminating hemodynamically tolerated VT. ATP is painless, although awareness of palpitations can occur. ATP is usually the initial therapy attempted for episodes of VT, because success rates are similar to those obtained with low-energy cardioversion; up to 90% of VTs can be terminated with pacing.125–127
ATP is more complex than defibrillation or cardioversion. The principle is to deliver pacing stimulation to the ventricle to gain control over the reentrant circuit that is perpetuating the tachycardia (overdrive suppression). If pacing is effective in entering the VT circuit, when pacing is terminated, the patient’s native or paced control over ventricular depolarization is restored. In order to enter the circuit, pacing must occur in the excitatory gap when the ventricle is not refractory to stimulation, and the device must pace at a rate faster than the VT rate. Rates with a cycle length between 70% and 90% of the VT cycle length (i.e., approximately 10% to 40% faster) are most effective in terminating the tachycardia.125,127 ATP techniques intended to improve entry into the circuit and termination of the tachycardia have been developed. Manufacturers do not share a standard nomenclature to describe ATP algorithms, but each method employs several comparatively simple principles. Burst pacing delivers a series of several beats at a fixed cycle length. Ramp pacing progressively shortens cycle length (i.e., accelerates). Adaptive therapy modes allow pacing at differing rates, depending on the VT rate. Scanning allows the device to introduce pacing at varying points in the VT cycle. In the setting of VT, the device delivers several different ATP protocols in an attempt to terminate the tachycardia.
Atrial therapies incorporated in some devices include ATP and cardioversion. Their effectiveness in preventing and terminating atrial arrhythmias has been demonstrated,114,128,129 but the clinical value of this approach remains controversial. It is very uncommon to implant a device to treat atrial arrhythmias solely, but this is occasionally done in highly symptomatic patients who are intolerant of medical therapy.
 Clinical Trials
Clinical Trials
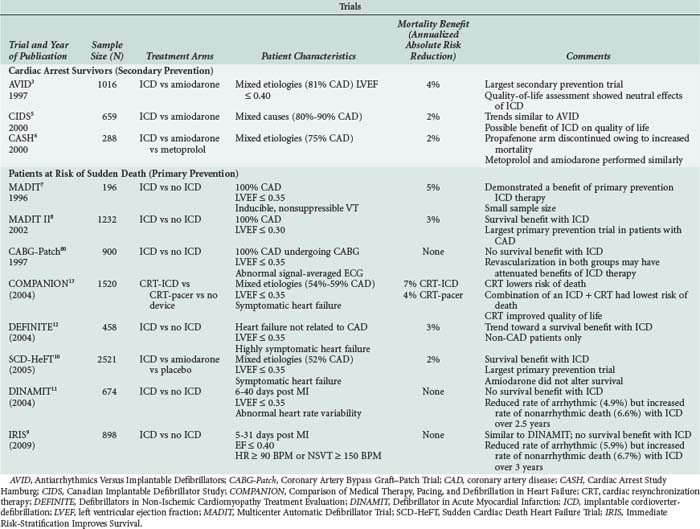
Many large (N > 100) randomized controlled trials assessing the efficacy of ICD therapy have been completed (see Table 81-2).3–1380 Three large trials assessed the role of ICD therapy as secondary prevention of sudden cardiac death among patients with ischemic LV dysfunction and sustained, hemodynamically significant ventricular arrhythmias.3,5,6 The largest of these trials (Antiarrhythmics versus Implantable Defibrillators [AVID]) randomized 1016 patients with symptomatic VT or VF and LV dysfunction (LV ejection fraction < 0.40) to therapy with ICD versus antiarrhythmic drugs (82.4% amiodarone).3 This study was stopped before completion of enrollment because of a statistically significant survival benefit (11.3% absolute risk reduction at 3 years) of the ICD. The Canadian Implantable Defibrillator Study (CIDS)5 and the Cardiac Arrest Study Hamburg (CASH)6 demonstrated trends toward decreased mortality, but these findings were not statistically significant. Meta-analysis of these three randomized trials supported data consistency, with a significant relative reduction in mortality risk of 28% (95% confidence interval [CI] 13%–40%).130
Several primary prevention trials assessed the role of ICD therapy among patients at risk for but without clinically sustained VT or VF.4,7,8,80 Although inclusion criteria varied, enrollment in these trials focused on patients with LV dysfunction. Similar to the secondary prevention trials, results of the primary prevention trials were consistent. Mortality reductions in the primary and secondary prevention trials have demonstrated similar results (see Table 81-2). From these studies it is clear ICD therapy reduces annual mortality by 2% to 7% in most patient groups. These studies also indicate that patients with both ischemic and nonischemic etiologies of LV dysfunction benefit from ICD therapy and that amiodarone has a limited role in the prevention of sudden death in patients with heart failure.
All but three of the primary prevention trials demonstrated a mortality benefit from ICD therapy. As previously discussed, routine aggressive coronary artery revascularization was likely responsible for the lack of benefit from routine ICD therapy in the CABG-Patch Trial.80 This inference is supported by a lower than anticipated mortality rate in that trial and the fact that the ICD resulted in a significantly lower rate of arrhythmic death.82 ICD therapy also did not reduce the risk of death in DINAMIT or IRIS (see Table 81-2). Similar to CABG-Patch, the proportion of arrhythmic deaths to the total deaths in these trials was also lower than anticipated.9,11 The lack of benefit from ICD therapy in these three studies illustrates that when considering a patient for an ICD, careful thought must be given to the long-term risk of arrhythmic death and the competing modes of death. ICDs have less impact with reduced rates of arrhythmic death.
A marked increase in the number of ICDs is occurring because of these trials. It is worth emphasizing that ICD therapy is costly,131,132 and the magnitude of benefit is sensitive to baseline risk.133 Studies to date have assessed ICD therapy in relatively high-risk populations, but even within these populations, risk appears to vary substantially. For example, in AVID, no benefit was observed among the subgroup of patients with an LV ejection fraction greater than 0.35.30 Whether ICD therapy is appropriate in lower-risk high-risk patients, particularly those with relatively preserved LV ejection fraction, remains to be determined. Further studies will aid in determining whether ICD therapy in such patients provides no benefit, small but costly benefit, small but clinically important benefit, or harm.
 Device-Related Issues Among Patients in Intensive Care
Device-Related Issues Among Patients in Intensive Care
Lead Failure
Lead failure due to dislodgment, fracture, or insulation breach occurs in 5% to 10% of patients, and lead replacement is usually required.134–136 Risk of lead failure is higher with a subclavian route compared with a cephalic vein approach, owing to the compressive effects of the clavicle and first rib on the subclavian vein.135 Lead failure is also more likely in younger patients, as well as certain specific leads that have been subject to manufacture advisory.137 Presenting complaints include inappropriate shocks, syncope or presyncope from device failure to deliver therapy, or proarrhythmia. Increased defibrillation thresholds can occur in the absence of lead defects, dislodgment, or change in physiologic conditions from ischemia, electrolyte abnormalities, or antiarrhythmic medications. This is thought to be due to myocardial fibrosis at the point of contact of the defibrillation lead. Frequent shocks appear to exacerbate this response. Steroid-eluting leads attenuate the inflammatory-fibrotic myocardial response and the associated increase in thresholds.
< div class='tao-gold-member'>

Full access? Get Clinical Tree