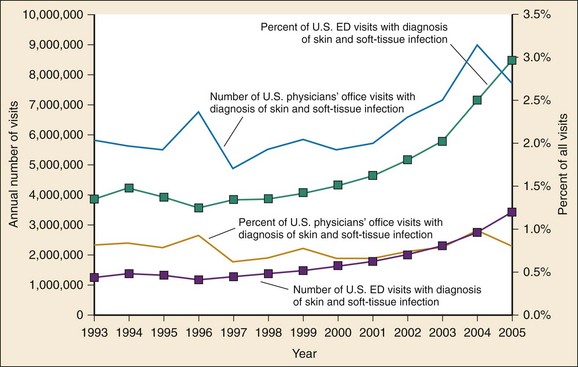
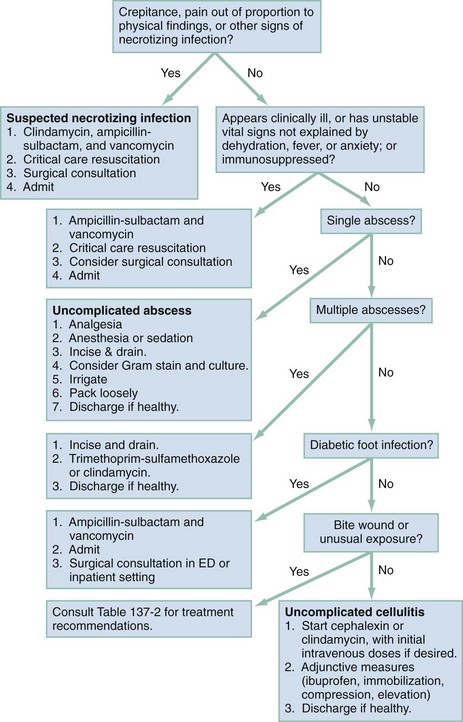
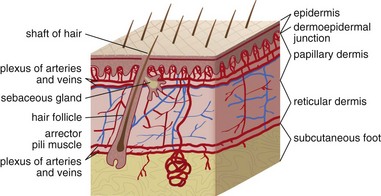
Chapter 137 Skin infections are among the most common problems treated by emergency physicians. In 2005 bacterial skin infections accounted for 3% of all emergency department (ED) visits in the United States, with more than 3,400,000 visits in that year alone.1 While treating many patients with mild skin infections, the emergency physician is vigilant for the rare occurrence of life threatening variants, such as necrotizing fasciitis. The epidemiology of skin infections has changed in the past decade with the emergence of a new organism, community-associated methicillin-resistant Staphylococcus aureus (CA-MRSA). Unlike most streptococci, S. aureus has usually been resistant to penicillin since penicillin was first commercialized because of the production of a β-lactamase (also known as penicillinase) enzyme that cleaves the penicillin molecule.2 Penicillinase-resistant penicillins typified by methicillin were developed in 1959, but strains of S. aureus resistant to these agents (MRSA) were identified by 1960.2 Resistance is due to an altered bacterial target, the cell wall building enzyme known as penicillin-binding protein. A new strain was identified in Chicago in the mid-1990s. It was characterized by resistance to methicillin and related antimicrobials but susceptibility to agents ineffective against the older “nosocomial MRSA”; it could not be traced to the hospital environment.3 The DNA fingerprinting technique known as pulsed-field gel electrophoresis identified the predominant CA-MRSA strain as USA-300.4 In contrast, the older nosocomial strain of MRSA is known as USA-100. A large, nationwide, ED-based study found CA-MRSA to be the leading identifiable cause of skin infections among ED patients.5 The number of ED visits for skin and soft tissue infections increased from 1.2 million in 1993 to more than 3.4 million in 2005; whether this was due to an increase in abscess, cellulitis, or both is unknown.1 Intriguingly, whereas CA-MRSA is now found in all populations of patients and in health care and community settings alike, the epidemic of skin infections it has caused is most apparent among ED patients for reasons that remain unclear.6 These data are summarized in Figure 137-1. The skin is the largest organ in the body and accounts for about 15% of total body weight.7 It has three layers, the hypodermis, the dermis, and the epidermis (Fig. 137-2). Hair is present throughout except on glabrous skin, which is the heavily keratinized skin found on the palms, soles, and parts of the genitals. Figure 137-2 Anatomy of the skin. (From Amirlak B: Skin anatomy. Available at http://emedicine.medscape.com/article/1294744-overview#a1. Accessed April 15, 2011.) The dermal-epidermal junction is a complex basement membrane whose disruption results in vesicles and bullae. The dermis consists of cells, fibers, and ground substance, which is an acellular material composed of glycoproteins and other macromolecules.7 The hypodermis, or subcutaneous tissue, is composed largely of adipocytes. For cellulitis and abscess, the only relevant laboratory test is a wound culture and Gram’s stain when pus is present. However, there is debate about the necessity of obtaining culture specimens from skin infections.8,9 A culture is prudent for complicated purulent infections, such as surgical wound infections that may involve deep structures or abscesses in immunocompromised patients. Uncomplicated skin abscesses are usually caused by CA-MRSA, and the need for culture is debatable. Nonpurulent cellulitis is not routinely cultured because of low yield.10 A stat Gram’s stain can be helpful in the setting of diagnostic uncertainty. Blood cultures are not indicated in patients with skin infection, with the following exceptions: septic shock, necrotizing infections, immunocompromise, multifocal infections suggesting hematogenous seeding, and infections complicating lymphedema. Early studies found pediatric facial cellulitis often to be accompanied by Haemophilus influenzae bacteremia.11,12 However, the relevant strain is type b, which is now covered by childhood vaccination. Moreover, this organism is typically be targeted presumptively in treatment of facial cellulitis, and there is no evidence that blood culture is of benefit. When a foreign body is suspected, plain radiographs are traditional, although they will miss foreign bodies that are small or radiolucent. Plain radiographs may reveal soft tissue gas but do not rule out necrotizing infection. Ultrasonography is promising for detection of foreign bodies associated with infection.11 Ultrasonography can also identify gas, which suggests necrotizing infection. Ultrasound examination is useful for differentiation of abscess from cellulitis, as discussed later.12 Cellulitis is an inflammatory abnormality of skin and subcutaneous tissue thought to be the result of bacterial infection.10 Cellulitis may be purulent or nonpurulent and may occur in the setting of wounds, foreign bodies, or impaired perfusion. Purulent cellulitis drains freely, in contrast to abscesses, which are walled off by fibrous tissue and epidermis. CA-MRSA is the leading cause of purulent skin and soft tissue infections in ED patients, but its role in nonpurulent cellulitis is unknown.1,5,6,10,13 When localized edema becomes severe, separation of epidermal layers can occur, leading to vesicles or bullae. This can make it difficult to distinguish cellulitis from other infectious and noninfectious causes of dermatitis. When the border of an area of cellulitis becomes well demarcated, raised, and palpable, the term erysipelas is used. This form of cellulitis is most often caused by Streptococcus pyogenes. The bacterial causes of cellulitis vary according to body site, comorbidities, and environmental exposures (see Table 137-2).10 Diabetic foot infections are the most common cause of hospitalization for patients with diabetes, and an infected wound precedes two thirds of lower extremity amputations in patients with diabetes.14 Neuropathy, vascular insufficiency, and hyperglycemia are important factors in the development of diabetic ulcers and foot infections. Nonetheless, it is important to avoid antibiotic overuse, and uninfected ulcers should not be treated with antibiotics. The most likely organisms in an acute diabetic foot infection are S. aureus and streptococci. Chronic wounds are more likely to be polymicrobial with both Gram-positive and Gram-negative organisms as well as anaerobes. Chronic wounds that have previously been treated with antimicrobials are more likely to involve multidrug-resistant organisms.15 Pseudomonas is a traditional concern but is not common.16 Deep tissue for aerobic and anaerobic culture or bone samples for biopsy should be obtained at the time of débridement if deep tissue infection or osteomyelitis is suspected, but organisms cultured from superficial swabs are not reliable for predicting the pathogens responsible for deeper infection. Osteomyelitis should be considered a potential complication of any deep or extensive ulcer, especially one that is chronic or overlies a bone prominence. In addition to antibiotics, diabetic foot infections require careful wound care and in some cases débridement, revascularization, or amputation. Exposure and travel history are important considerations in the evaluation of skin and soft tissue infections. Vibrio species, in particular Vibrio vulnificus, are associated with exposure to seawater and can cause severe soft tissue infections.17 Patients with liver disease, such as cirrhosis, are at risk. Infection occurs from contamination of open wounds by seawater or shellfish and rarely by hematogenous spread from ingestion of contaminated seafood such as raw oysters. Edwardsiella tarda is a rare cause of wound infection after seawater exposure; it has been implicated in serious soft tissue infections including myonecrosis, particularly in patients with liver disease. Erysipelothrix rhusiopathiae is usually associated with a localized erysipeloid eruption with minor trauma, often on the hands of seafood workers. Needle aspiration and even biopsy of cellulitis lesions are unlikely to reveal the etiology and are not recommended.10 Blood cultures are not recommended except in the presence of a presumed hematogenous source of infection, in septic shock, and in cellulitis complicating lymphedema. Blood cultures are also traditional for facial cellulitis but may be omitted in well-appearing patients who are treated with agents active against H. influenzae.18 The role of high-sensitivity pathogen identification, such as polymerase chain reaction analysis of surface skin swabs, has yet to be determined. Emergency physicians have changed their prescribing practices dramatically since CA-MRSA was first described.1 In 1993, antibiotics targeted to CA-MRSA were almost never prescribed for skin and soft tissue infections. By 2005, 38% of antibiotic regimens included an agent typically active against CA-MRSA. Multiple studies have shown that antibiotics do not make a difference in the treatment of abscess, and the effectiveness of agents targeting CA-MRSA in nonpurulent skin infections (i.e., cellulitis) is unknown. Table 137-1 summarizes relevant antibiotics. Oral agents with activity against CA-MRSA include trimethoprim-sulfamethoxazole, clindamycin, tetracyclines, and linezolid. The common strains of CA-MRSA (i.e., USA-300) are almost universally susceptible to trimethoprim-sulfamethoxazole in vitro. It is well tolerated and causes severe reactions like Stevens-Johnson syndrome no more commonly than do other agents, such as ampicillin. However, it is not considered effective against streptococci. Thus, when CA-MRSA is suspected but streptococci remain a possibility, the best therapy is most likely trimethoprim-sulfamethoxazole plus a β-lactam such as cephalexin or clindamycin monotherapy.18 Table 137-1 First-Line Oral Antibiotics for Skin and Soft Tissue Infection *Dose by amoxicillin component. Dicloxacillin is commonly suggested as an agent for treatment of cellulitis because of its appropriate spectrum and high potency. However, it must be taken four times daily on an empty stomach—a very challenging task—and causes gastrointestinal upset. Doxycycline is another option for coverage of CA-MRSA. However, this agent must also be taken on an empty stomach and causes gastrointestinal upset. Moreover, it is not thought to cover streptococci well.18 As with trimethoprim-sulfamethoxazole, when doxycycline is prescribed for skin and soft tissue infections, a β-lactam should be added to target streptococci unless the etiology of the infection is known. Doxycycline causes photosensitivity, and patients should be warned to avoid sun exposure. Clindamycin is an important agent because most CA-MRSA isolates are sensitive to it, and it is an excellent agent for streptococci. It can be taken with food. CA-MRSA can become resistant to clindamycin during a single course of treatment. This phenomenon is known as inducible resistance and occurs in about 2% of CA-MRSA isolates.5,19 Most microbiology laboratories in the United States now test for inducible resistance among MRSA strains that are susceptible to clindamycin in vitro. The test for inducible resistance is known as the D-test. The name comes from a D-shaped pattern of the area of clindamycin’s inhibition of bacterial growth, which results from resistance induced by a pellet (“disk”) of erythromycin that is placed next to the clindamycin pellet.20 Early reports of susceptibility of CA-MRSA to rifampin gave way to later reports of widespread resistance, both inducible and de novo. This agent is not recommended for skin infections.18 Vancomycin is the standard parenteral agent for MRSA. Others include linezolid, daptomycin, tigecycline, and telavancin. No penicillin or cephalosporin is active against CA-MRSA, except ceftaroline, a parenteral cephalosporin approved by the U.S. Food and Drug Administration in 2010 and indicated for complicated skin infections.21 Nonpurulent cellulitis is treated with coverage for streptococci and methicillin-sensitive S. aureus. When the infection is purulent, a culture and Gram’s stain may be performed and coverage for CA-MRSA provided. Otherwise, agents effective against CA-MRSA are not recommended except in cases not initially responsive to first-line therapy and when septic shock is present.18 The first-line agent is cephalexin, which is safe, well tolerated, and well absorbed and need not be taken on an empty stomach. Inadequate dosing and failure to adhere to recommended adjunctive measures may lead to treatment failure. Relatively severe infections may be managed with one or more initial doses of the intravenous equivalent cefazolin, which, like cephalexin, should be given in maximal doses (e.g., 2 g every 8 hours for adults). Nafcillin is an equivalent option. In patients allergic to cephalexin, clindamycin is an excellent choice. Table 137-2 provides further treatment recommendations. Table 137-2 Bacteriology of and First-Line Antibiotic Therapy for Skin Infections CA-MRSA, community-associated methicillin-resistant Staphylococcus aureus. *For life- or limb-threatening infections, use intravenous equivalents and add vancomycin. Adjunctive measures are important in the treatment of cellulitis. Extremity cellulitis responds dramatically to compression and elevation. The extremity should be elevated above the level of the heart. A splint is useful as an anchor for elevation and can be hung from an intravenous pole. Patients with cellulitis complicating venous stasis or lymphedema should be educated about the importance of compression not only for the acute infection but also to prevent future episodes. Nonsteroidal anti-inflammatory agents (e.g., ibuprofen) are effective and should not be forgotten.22 As stated before, many cases of cellulitis treatment failure may be due to failure to adhere to these adjunctive measures rather than antibiotic choice. Figure 137-3 is a universal treatment algorithm for skin and soft tissue infections. The algorithm assumes no prior treatment and no toxic shock syndrome. Previously treated infections require broader spectrum antibiotic coverage and customized management decisions.
Skin and Soft Tissue Infections
Perspective
Principles of Disease
Clinical Features
Diagnostic Strategies
Cellulitis
Principles of Disease and Clinical Features
Diabetic Foot Infections
Water-borne Infections
Differential Considerations and Diagnostic Strategies
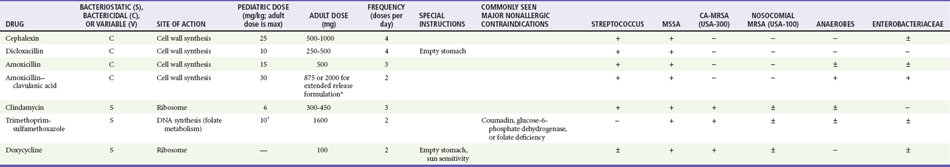
Management
ANATOMIC VARIANT OR PREDISPOSITION
LIKELY BACTERIAL CAUSE
FIRST-LINE THERAPY (NONTOXIC AND IMMUNOCOMPETENT)*
Uncomplicated cutaneous abscess
CA-MRSA, others
Incision and drainage without antibiotics
Nonpurulent bacterial skin infections
Various species in the Streptococcus genus, Staphylococcus aureus
Cephalexin or clindamycin
Adjunctive measures
Purulent cellulitis and wound infections
CA-MRSA, others
Cephalexin plus trimethoprim-sulfamethoxazole, or clindamycin monotherapy
Adjunctive measures
Diabetic foot infection
Mixed Gram-positive, Gram-negative, and anaerobes
Amoxicillin–clavulanic acid plus trimethoprim-sulfamethoxazole
Avoid antibiotics for uninfected ulcers
Any cat bite or infected dog bite
Pasteurella multocida, others
Amoxicillin–clavulanic acid
Human bite (treat presumptively)
Oral anaerobes, others
Amoxicillin–clavulanic acid
Erythema migrans
Borrelia burgdorferi (Lyme)
Doxycycline
Puncture wound through sole of shoe (treat presumptively)
Pseudomonas aeruginosa
Ciprofloxacin
Buccal cellulitis
Haemophilus influenzae type b (vaccine serotype)10
Ceftriaxone or ampicillin-sulbactam
Balanitis
Candida albicans or group A streptococcus
Fluconazole plus penicillin or amoxicillin
Consider diabetes
Liposuction
Peptostreptococcus (anaerobe), group A streptococcus
Ampicillin–clavulanic acid ± trimethoprim-sulfamethoxazole
Saltwater exposure
Vibrio vulnificus
Doxycycline
Freshwater exposure
Aeromonas species
Ciprofloxacin
Butcher, clam handler, veterinarian
Erysipelothrix rhusiopathiae
Amoxicillin
Black necrotic eschar with raised border and severe surrounding edema
Bacillus anthracis (anthrax)
Ciprofloxacin
< div class='tao-gold-member'>

Full access? Get Clinical Tree


Skin and Soft Tissue Infections
Only gold members can continue reading. Log In or Register to continue