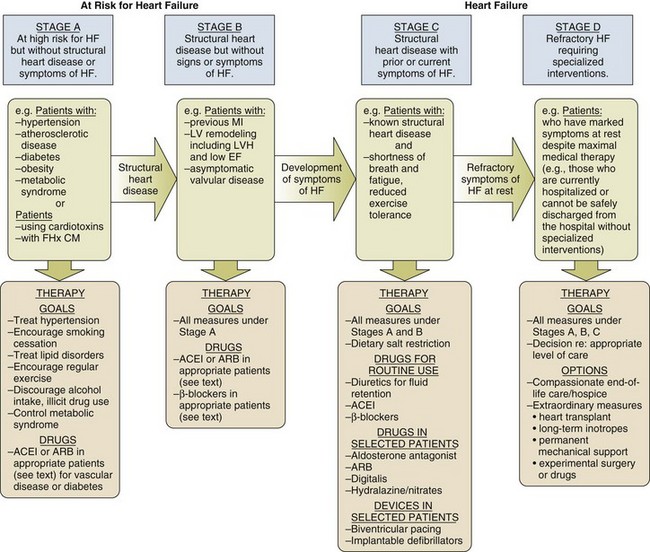
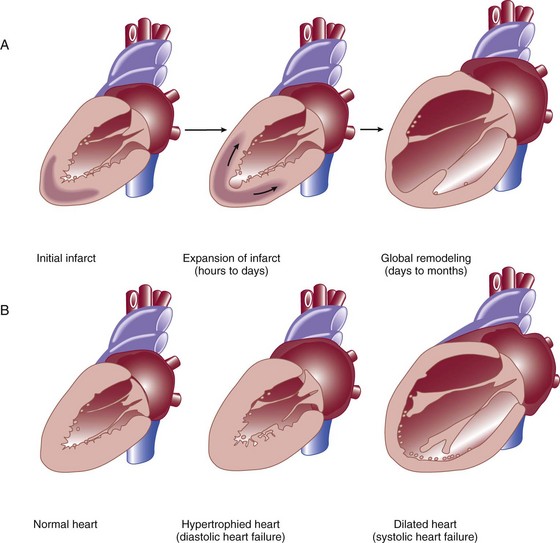
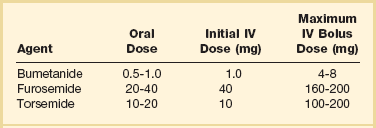
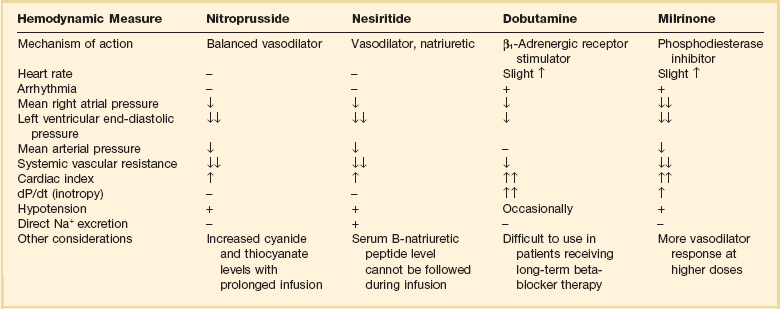
29 DEFINITION, EPIDEMIOLOGY, AND STAGING OF HEART FAILURE PROGNOSIS IN ACUTE HEART FAILURE PHARMACOLOGIC MANAGEMENT OF ACUTE HEART FAILURE TRANSITION TO CHRONIC PHARMACOLOGIC THERAPY FOR SEVERE HEART FAILURE CORONARY HEART DISEASE AND HEART FAILURE: SPECIAL CONSIDERATIONS HEART FAILURE WITH PRESERVED LEFT VENTRICULAR EJECTION FRACTION (DIASTOLIC HEART FAILURE) ACUTE MYOCARDITIS AND HEART FAILURE DEVICE THERAPY: IMPLANTED CARDIOVERTER-DEFIBRILLATORS AND CARDIAC RESYNCHRONIZATION THERAPY MECHANICALLY ASSISTED CIRCULATORY SUPPORT: VENTRICULAR ASSIST DEVICES Heart failure is a very common illness; 5.8 million Americans are affected.1,2 It is estimated that 1% of the population of Americans over the age of 65 are affected by heart failure, and 20% of hospital admissions in patients over age 65 are due to heart failure.3 Men and black Americans are affected more frequently. In 2008 670,000 new cases of heart failure were diagnosed in the United States, where there are nearly 1 million hospital discharges, 658,000 visits to the emergency department, over 3.4 million ambulatory outpatient visits, and 6.5 million hospital days annually for patients with a primary diagnosis of heart failure.1,2 Over 56,000 people in the United States died in 2007 with heart failure as the primary cause. The treatment of heart failure incurs a very large economic burden on the U.S. health care system. Estimated direct and indirect cost of heart failure in 2010 was $29 billion. The majority of this cost was related to the treatment of patients hospitalized with heart failure. Epidemiologic data showed significant increases in both the incidence and prevalence of heart failure in the U.S. population in the 1990s,3 likely influenced by the aging of the population, a high prevalence of hypertension, and improved treatment and survival of patients with ischemic heart disease.4 However, there is evidence that hospitalizations for heart failure have declined during the past decade. In the United States, the rate of hospitalization for heart failure declined from 2845 per 100,000 patient-years in 1998 to 2007 per 100,000 person-years in 2008, a decline of 29%.5 Patients hospitalized with acute heart failure are usually elderly, with a mean age in the early 70s. In the United States, roughly 80% of patients will have a previous history of heart failure,6 and in European studies one third of patients had a new diagnosis of heart failure.7 From 40% to 55% of patients will have normal or relatively normal left ventricular systolic function. This occurs more commonly in women than in men. Coronary heart disease is present in 50% to 60% of patients and hypertension in 72% of patients. Comorbid conditions are common, including renal disease in 30% of patients, diabetes mellitus in 43%, and chronic obstructive pulmonary disease (COPD) in roughly 30%.6–8 In the American College of Cardiology/American Heart Association Guidelines for the diagnosis and management of chronic heart failure, four stages in the development of heart failure are recognized (Fig. 29.1). This staging system emphasizes the progressive nature of left ventricular dysfunction and heart failure, and describes evidence-based guidelines for therapy for each stage.4 It is important to realize that heart failure may be preventable. Several common medical conditions place patients at high risk for developing left ventricular dysfunction and the heart failure syndrome. Attention to and appropriate management of these conditions may prevent the development of heart failure. In addition, in patients who have left ventricular dysfunction but who have not yet developed heart failure, appropriate therapy can improve prognosis and prevent the development of severe heart failure. Stage A represents patients at high risk for heart failure but without structural heart disease or symptoms of heart failure. These patients include those with hypertension, atherosclerotic heart disease, diabetes, obesity, or the metabolic syndrome; those exposed to cardiotoxic medications; and those with a family history of cardiomyopathy. About 75% of patients who develop heart failure have antecedent hypertension.2 Stage B patients have structural heart disease but without signs or symptoms of heart failure. This group includes patients who have suffered a myocardial infarction, those with abnormal left ventricular ejection fraction but no symptoms of heart failure, and those patients with asymptomatic valvular heart disease. Stage C patients have structural heart disease with prior or current symptoms of heart failure, such as shortness of breath, fatigue, and reduced exercise tolerance. Lastly, stage D patients have refractory symptoms of heart failure at rest despite maximal medical therapy. They require specialized or extraordinary interventions such as cardiac transplantation, mechanical circulatory support, or end-of-life care. Heart failure can be due primarily to left ventricular systolic dysfunction or diastolic dysfunction, although both abnormalities are often present together. Right ventricular dysfunction may accompany left ventricular dysfunction or may be the primary problem. Systolic heart failure results from inability of the heart to expel blood normally owing to depressed left ventricular contraction. The left ventricle is often dilated. There is loss of myocytes and fibrosis, resulting in reduced left ventricular ejection fraction. Diastolic heart failure is caused by a reduction in left ventricular compliance, which leads to impaired diastolic filling, higher left ventricular diastolic pressure, and elevated pulmonary capillary wedge pressure (PCWP). Diastolic heart failure is also called heart failure with preserved ejection fraction (HFpEF) or heart failure with normal ejection fraction (HFnEF) (Fig. 29.2A and B). Numerous conditions may cause damage to left ventricular myocardium and result in systolic heart failure. The most common of these conditions is atherosclerotic coronary artery disease with myocardial infarction and chronic myocardial ischemia. Other common causes include hypertension; familial and idiopathic cardiomyopathy; viral myocarditis; valvular heart disease, such as aortic stenosis, aortic insufficiency, and mitral regurgitation; peripartum cardiomyopathy; transient apical ballooning syndrome (takotsubo cardiomyopathy); and cardiomyopathy due to cardiotoxic cancer chemotherapy. Less common causes are alcoholic cardiomyopathy, diabetic cardiomyopathy, and a cardiomyopathy seen in some patients with hyperthyroidism or severe obesity.9–11 After myocardial damage and loss of myocytes occur, a process known as ventricular remodeling is initiated. This remodeling results in dilation of the ventricular chamber, a change in ventricular geometry from ellipsoid-like to a more spherical shape, worsening of left ventricular contractile force, and reduction in ventricular ejection fraction.3,12 In addition, the change in left ventricular geometry often leads to an increase in the size of the mitral annulus and altered physical relationships of the mitral valve structures. This results in increasing mitral regurgitation and worsening of the heart failure syndrome (see Fig. 29.2). Remodeling is mediated by a series of maladaptive systemic responses, commonly termed neurohormonal activation. Two major systems involved in neurohormonal activation are the renin-angiotensin-aldosterone system and the sympathetic nervous system. Activation of the renin-angiotensin-aldosterone system leads to elevated levels of renin, angiotensin II, and aldosterone, which has deleterious consequences on cardiac function and hemodynamics.3,12 These effects include salt and fluid retention, endothelial dysfunction, vasoconstriction, myocyte hypertrophy, myocardial fibrosis, and myocyte apoptosis (programmed cell death). Sympathetic nervous system activation is in part mediated via decreased cardiac output, which results in tachycardia, increased myocardial oxygen consumption, and peripheral vasoconstriction.13 Renal effects of sympathetic nervous system activation lead to further activation of the renin-angiotensin-aldosterone system. Increased circulating norepinephrine levels also contribute to myocyte injury and death. Arrhythmias become common. A detrimental positive feedback loop is established, causing progressive deterioration in left ventricular structure and performance over time with progressive worsening of chronic heart failure. Heart failure is a clinical diagnosis, determined after evaluation of a patient’s symptoms and physical examination and supported by results of ancillary testing, such as chest x-ray and echocardiography. Typical symptoms of heart failure include shortness of breath at rest or with exertion, cough, swelling, orthopnea, and paroxysmal nocturnal dyspnea, which are due to pulmonary or systemic venous congestion. Fatigue, anorexia, and change in mental status are symptoms that may be caused by low cardiac output. Physical examination often reveals pulmonary rales; elevated jugular venous pressure; signs of cardiomegaly; cardiac murmurs, especially mitral regurgitation and third heart sound (S3) gallop; hepatic enlargement; and ascites and edema. An S3 gallop and elevated jugular venous pressure are the most specific signs of heart failure; absence of rales is not infrequent in patients with acute heart failure, and chest radiograph may not show obvious congestion.14 Manifestations of more severe heart failure include marked dyspnea at rest, possibly resulting in respiratory failure, cyanosis, cool extremities, reduced urine output and altered mental state. A careful evaluation for the presence of pulsus paradoxus is important if cardiac tamponade is suspected. However, these signs and symptoms are not specific for heart failure, and coexisting conditions such as obesity, chronic lung disease, and deconditioning can add uncertainty to the clinical diagnosis. Heart failure is often both underdiagnosed and overdiagnosed in outpatient and emergency department settings. Studies have shown that serum assays of B-natriuretic peptide (BNP) and N-terminal pro-BNP are very helpful in improving the accuracy of diagnosing heart failure. BNP is a 32–amino acid peptide produced by cardiac myocytes in response to pressure-induced wall stretch and tension.15 Physiologic actions of BNP include arterial and venous dilation and natriuresis. A study of 1530 patients presenting to the emergency department with dyspnea showed that knowledge of BNP serum levels resulted in improved accuracy of the diagnosis of heart failure when compared to clinical judgment alone, from an initial range of 65% to 74%, up to 81% accuracy.16 BNP levels above 100 pg/mL had a sensitivity of 90% and a specificity of 76% for the diagnosis of heart failure and were very useful for discriminating patients with dyspnea due to uncomplicated lung disease who had BNP values below 100 pg/mL.17 Patients with BNP values below 100 pg/mL were very unlikely to have heart failure as the cause of dyspnea. Levels between 100 and 400 pg/mL can be seen in dyspneic patients with cor pulmonale, pulmonary hypertension not due to left ventricular failure, and acute pulmonary embolism. Heart failure is the likely diagnosis when levels are above 400 pg/mL. It should be emphasized that BNP levels should always be used in conjunction with all other clinical data to arrive at the correct diagnosis. Evaluating BNP levels in heart failure patients in the emergency department was shown to decrease the need for hospitalization and decrease the need for intensive care unit (ICU) admissions, without affecting 30-day mortality rates.18 Total hospital stay was shortened by 3 days, cost of treatment was significantly reduced, and time to initiation of definitive therapy in the emergency department was shortened by 30 minutes. BNP levels correlate with disease severity, as values are higher in patients with more severe heart failure and with worse left ventricular systolic function. Higher levels also have been correlated with a poorer prognosis, and can predict an increased rate of functional deterioration and a higher mortality rate.19 In a study of 114 patients admitted to the hospital with class IV heart failure, of all variables evaluated, predischarge BNP level was most strongly associated with death or readmission within 6 months, with BNP levels greater than 350 pg/mL having impressive sensitivity and specificity.20 BNP levels may also be elevated in patients with diastolic heart failure, those with renal failure, and stable patients with chronic left ventricular systolic dysfunction with compensated heart failure. BNP levels may initially be low in patients with “flash” pulmonary edema, as they may present to the hospital more quickly than the time required for significant rises in serum BNP to occur. Serum levels of N-terminal (NT) pro-BNP can also be used for the diagnosis of heart failure. A precursor hormone, pro-BNP, is cleaved to form BNP and NTpro-BNP, which is physiologically inactive. NTpro-BNP has a longer half-life than BNP. It is cleared from the serum by the kidneys, so levels are higher in patients with coexisting renal disease. Levels of NTpro-BNP rise significantly in older populations. A cut off level of NTpro-BNP below 300 pg/mL yielded a negative predictive value of 98% for exclusion of the diagnosis of heart failure.21,22 The diagnosis of heart failure is very likely at levels over 450 pg/mL for patients under age 50, above 900 pg/mL in patients between 50 and 75 years old, and above 1800 pg/mL in patients over the age of 75. Hospitalization for acute heart failure is associated with a poor prognosis. In-hospital mortality rate is high, reported at 4% to 8%. There is a 9% mortality rate at 60 to 90 days, and a 1-year mortality rate of 29%.1,6,7,23–25 The 90-day rehospitalization rate is around 30%, although only half of these rehospitalizations are caused by heart failure. In a European heart failure database, in-hospital mortality rate was 6.9%, 12-week readmission rate was 24%, and total mortality rate at 12 weeks was 13.5%.8 Randomized trials of pharmacologic therapy reported an annual mortality rate of 10% in patients with class II-III symptoms and 20% to 50% in class IV patients. In the most severe chronic heart failure group, patients awaiting cardiac transplantation, 1-year mortality rate was 75% with 2-year mortality rate of 92%.26 More recent data indicate a decline in the risk-adjusted inpatient mortality rate, from 5.5% in 2000 to 2.8% in 2007. Similar reductions were seen in both sexes and across all age groups.27 The prognosis for patients with cardiogenic shock due to acute myocardial infarction continues to be poor. Mortality rate remains 40% to 50% even with aggressive supportive therapy and emergency revascularization strategies.28 Several clinical factors have been shown to identify patients with a poorer prognosis and include lower left ventricular ejection fraction, low blood pressure on admission, and higher PCWP. In the United States ADHERE database registry of 62,275 admissions for heart failure, blood urea nitrogen (BUN) greater than 43 mg/dL, admission systolic blood pressure under 115 mm Hg, and creatinine level greater than 2.75 mg/dL were the three factors that indicated a poor prognosis for patients admitted with acute heart failure. The presence of an elevated BUN was associated with a fourfold increase in hospital mortality rates to 8.35%. The presence of all three factors yielded an in-hospital mortality rate of 19.8%24 (Box 29.1). Other factors associated with increased mortality rates include hyponatremia, higher serum BNP level, and elevation of serum troponin.6,29,30 In ADHERE, higher levels of serum BNP on admission were associated with a higher in-hospital mortality rate, ranging from 1.9% in the lowest quartile to 6.0% in the highest quartile (BNP over 1730 pg/mL). The ability of BNP to predict prognosis was present even after multivariate adjustment for coexisting conditions, and was also true for patients with normal and abnormal left ventricular ejection fraction.31 Serum troponin is elevated in 6% to 10.4% of patients admitted with acute heart failure (with serum creatinine <2.0 mg/dL). Troponin elevation was associated with a lower blood pressure, lower ejection fraction, and longer hospital length of stay. Hypothetical mechanisms of troponin release in chronic heart failure include ischemia, cytokine activation, oxidative stress, and apoptosis. Hospital mortality rate was 8.0% in troponin-positive patients and 2.7% if troponin-negative. The ability of troponin elevation to predict mortality rate also was independent of other variables and was true even in patients with nonischemic causes of heart failure.32 The coexistence of kidney disease significantly worsens the prognosis of patients with heart failure. Kidney disease aggravates the tendency to volume overload and heart failure decompensation, and heart failure often worsens renal function. This complex interaction of severe heart failure and worsening kidney function is called the cardiorenal syndrome. In addition, high-dose diuretic therapy may also temporarily worsen renal function. There is increasing evidence that elevated systemic venous pressure causing renal venous congestion plays an important role in the pathogenesis.33 In a retrospective study of patients with acute heart failure who had right-sided heart catheterization, elevated central venous pressure was associated with reduced glomerular filtration rate (GFR) and higher all-cause mortality rate. These findings were independent of the measured cardiac output.34 Up to 50% of patients hospitalized with heart failure demonstrate a GFR less than 60 mL/minute/m2, and renal function may worsen in up to 30% of patients admitted with heart failure.35 Patients at particular risk for developing worsening renal function are those with lower left ventricular ejection fractions, lower blood pressure, diabetes mellitus, a history of hypertension, and older age. These patients have longer hospital stays and higher readmission and mortality rates. A meta-analysis of 16 large studies of heart failure patients revealed that 29% of heart failure patients had moderate to severe impairment of renal function. These patients had more than 100% increased relative mortality risk. Any degree of renal impairment had an approximately 50% increased relative mortality risk.36 The best treatment strategy for these patients is not clear, as patients with significant renal impairment have generally been excluded from large randomized pharmacologic heart failure trials. Acute heart failure is defined as the rapid onset of severe symptoms of heart failure, usually within hours to several days. Acute heart failure can occur with predominant systolic or diastolic dysfunction. Acute heart failure is often life-threatening and requires urgent diagnostic and therapeutic interventions, often simultaneously.23 Acute myocardial ischemia is a common cause and should always be considered in the differential diagnosis of this syndrome. Several distinct clinical syndromes of acute heart failure can be identified23 (Table 29.1). Table 29.1 Adapted from Nieminen MS, Bohm M, Cowie MR, et al: Executive summary of the guidelines on the diagnosis and treatment of acute heart failure. The Task Force on Acute Heart Failure of the European Society of Cardiology. Eur Heart J 2005;26:384-416; and Gheorghiade M, Zannad F, Sopko G: Acute heart failure syndromes. Circulation 2005;112:3958-3968. 1. Acute worsening or decompensation of chronic heart failure symptoms, either in the setting of known chronic cardiovascular illness or de novo. These patients do not have shock or pulmonary edema. This is the most common presentation of acute heart failure requiring admission to hospital, occurring in approximately 70% of patients with acute heart failure. 2. Acute pulmonary edema with normal blood pressure, often caused by acute myocardial infarction or acute coronary ischemia. 3. Acute pulmonary edema associated with elevated blood pressure, often in the setting of chronic severe hypertension and chronic kidney disease. Pulmonary edema accounts for roughly 25% of acute heart failure admissions. 4. Cardiogenic shock with heart failure, usually due to acute myocardial infarction. This syndrome is the most severe presentation of acute heart failure and is associated with high in-hospital mortality rates. This accounts for about 5% of acute heart failure cases. 5. “High cardiac output heart failure” often induced by sepsis, hyperthyroidism, or cardiac arrhythmia. This type is the least common presentation, occurring in a small percentage of patients. 6. Acute right ventricular failure, occurring with acute right ventricular myocardial infarction, massive pulmonary embolism, or cardiac tamponade. Physical examination often shows pulmonary rales and wheezes, an S3 gallop, and elevation of jugular venous pressure. Peripheral pulses are weak and thready with diminished cardiac output states. A low cardiac output is reliably predicted by a low “proportional pulse pressure,” which is calculated by the pulse pressure (systolic blood pressure minus diastolic blood pressure) divided by systolic blood pressure. A ratio below 0.25 predicts a cardiac index below 2.2 L/minute/m2.37,38 The skin may be cool and clammy and there may be evidence of cyanosis. Peripheral edema and ascites may indicate concomitant right ventricular failure of longer duration. Chest radiography is done urgently. An electrocardiogram (ECG) is needed to assess for signs of ischemia and infarction, and to evaluate for arrhythmia. Cardiac rhythm needs to be monitored continuously. Laboratory examination includes evaluation of hemoglobin and hematocrit, electrolytes, renal and liver function, thyroid profile, and cardiac biomarkers (troponin I or troponin T) to look for evidence of myocardial necrosis. BNP level assists in the diagnosis of heart failure in patients presenting with dyspnea, and can be followed serially to assess effectiveness of therapy. Pulse oximetry helps to assess oxygenation and pulmonary function. An arterial line is helpful in managing patients with hypotension or cardiogenic shock. Urgent two-dimensional echocardiography is essential to evaluate left ventricular size and function, right ventricular function, valve function, and the presence of pericardial effusion. Doppler echocardiographic assessment of valve stenosis and regurgitation and of hemodynamics is invaluable. When patients are admitted to hospital with acute decompensation of chronic systolic heart failure, the specific reason for a patient’s deterioration must be searched for and corrected when possible (Box 29.2). Environmental factors such as excessive salt and fluid intake or alcohol consumption are common. Patient adherence to outpatient therapies must be assessed, as heart failure regimens often involve numerous medications. Emotional and physical stressors should be corrected when feasible. Concomitant administration of medications for noncardiac conditions can have detrimental effects. A partial listing includes corticosteroids and nonsteroidal anti-inflammatory drugs (NSAIDs). These drugs can cause fluid retention and can aggravate hypertension. NSAIDs also interfere with the beneficial renal effects of angiotensin-converting enzyme (ACE) inhibitors and can interfere with the action of loop diuretics.39 The use of NSAIDs has been reported to increase the risk of hospitalization for heart failure by tenfold in patients with a history of heart failure.8 Metformin and thiazolindinediones can contribute to water retention and aggravate the symptoms and signs of heart failure. Cancer chemotherapies can cause myocardial damage. Cardiac toxicity due to anthracycline chemotherapy is well described. Tyrosine-kinase inhibitors, a newer class of cancer chemotherapy, are also being recognized as agents that can aggravate heart failure and cardiomyopathy.40 Cardiac medications such as calcium channel blockers and antiarrhythmic drugs can also have direct negative effects on left ventricular contractility. Calcium channel blockers are generally contraindicated in patients admitted with acute decompensated heart failure.23 Type 1A and 1C antiarrhythmic drugs are also contraindicated in patients with abnormal left ventricular systolic function. There are few controlled-trial data to arrive at evidence-based guidelines for the treatment of acute heart failure. Many recommendations are based on small studies, experience, observation, and a general consensus of opinion.41 The goals of initial therapy are to improve symptoms, optimize blood pressure, lower PCWP, and improve cardiac output. Treatments to reverse or prevent myocardial injury are instituted, and a search for reversible causes of heart failure needs to occur. Optimization of other comorbid conditions is important, including hyperglycemia, renal disease, and pulmonary function. Initial therapy includes supplemental oxygen and assessment of the need for ventilatory assistance with noninvasive positive airway pressure ventilation or endotracheal intubation. Noninvasive ventilation improves oxygenation and pulmonary compliance and decreases work of breathing. Endotracheal intubation may be required for patients with severe hypercapnia, acidosis, and respiratory muscle fatigue. The use of continuous oxygen administration alone was prospectively compared with the use of continuous positive airway pressure ventilation (CPAP) and noninvasive intermittent positive pressure ventilation (NIPPV) in 1069 patients who presented with acute cardiogenic pulmonary edema. No difference among these therapies was noted in the primary end point of death at 7 days, or in the secondary end point of death plus endotracheal intubation at 7 days. Noninvasive ventilation did result in more rapid improvement in dyspnea, tachycardia, hypercapnia, and acidosis. There was no difference in safety or efficacy between CPAP and NIPPV.42 Treatment of arrhythmias is essential. Rapid atrial fibrillation is a common problem in these patients. In the Euroheart Failure Study, 9% of patients hospitalized with acute heart failure had atrial fibrillation during the hospitalization, and 42% had a history of paroxysmal atrial fibrillation.43 Other studies report atrial fibrillation in 25% to 30% of hospitalized heart failure patients.36 Control of the ventricular response to atrial fibrillation is vitally important, especially in patients with diastolic heart failure. This control can be achieved rapidly with the use of intravenous beta blockers such as metoprolol or esmolol, parenteral digoxin, or intravenous amiodarone. Intravenous diltiazem can be used in patients whose left ventricular systolic function is known to be normal or near normal. Placement of a pulmonary artery (PA) catheter enables the clinician to accurately measure PCWP, cardiac output, and mixed venous oxygen saturation. It can also help assess the effectiveness of therapy. Whether to routinely use PA catheters to assess and manage patients with acute heart failure has been long debated. The ESCAPE trial evaluated the routine use of PA catheterization in patients hospitalized with acute exacerbation of chronic heart failure and left ventricular systolic dysfunction.44 There was no difference in the primary end point of days alive out of the hospital during 6 months after discharge in groups managed with or without a PA catheter. There were no significant adverse effects with PA catheter use in this study. There were no subgroups identified in which use of the PA catheter was beneficial. However, there was a trend noted in improving the initial diuresis, with less deterioration of renal function, in the PA catheter group. The authors concluded that there was no indication for the routine use of PA catheters in the setting of acute heart failure. However, a PA catheter is often essential for the management of patients with acute severe heart failure. Findings on physical examination are often insensitive indicators of hemodynamic status. Indications for PA catheter use include cardiogenic shock, differentiating pulmonary from cardiac causes of dyspnea, hemodynamic assessment if one is unsure of the diagnosis or severity of heart failure by clinical assessment, worsening renal function, guiding parenteral vasodilator therapy, and in patients who are not improving with initially prescribed therapy.37 PA catheter placement is also necessary as part of the evaluation for cardiac transplantation or the implantation of a ventricular assist device (VAD) (Box 29.3). Consecutive patients with severe heart failure were evaluated and classified according to hemodynamic measurements of PCWP and cardiac index. Patients were described as “dry” with average PCWP less than 17 mm Hg, or “wet” with PCWP reading of 29 mm Hg on average. The patients were also described as “warm” versus “cold,” based on a cardiac index of greater than 2.1 L/minute/m2 versus an index of less than 1.6 L/minute/m2. The severity of symptoms and findings on physical examination did not predict the hemodynamic status as defined by invasive monitoring. In addition, the hemodynamic picture did not predict the response to therapy and survival was similar in all four groups, except that patients with higher cardiac output and lower PCWP had slightly better outcomes than patients with low cardiac output and high PCWP.45 Tailoring pharmacologic therapy to hemodynamic measurements is often helpful and necessary to determine precise measurements of cardiac output and left ventricular filling pressure, in order to guide intensive intravenous drug therapy. Aggressive therapy tailored to the response in hemodynamic measurements has been advocated as an effective method to obtain more rapid and sustained improvement in patients with the most severe heart failure.46 When PCWP is reduced to less than 16 mm Hg, and right atrial pressure is reduced to less than 8 mm Hg, most patients will improve acutely and for the remainder of their hospitalization. Additional hemodynamic goals include reducing systemic vascular resistance to less than 1200 dynes × sec/cm5, raising cardiac index to greater than 2.6 L/minute/m2, and maintaining systolic blood pressure over 80 mm Hg. PCWP can be lowered to a normal value of 10 to 12 mm Hg in many patients with significant left ventricular dysfunction without untoward effects.46,47 In a group of patients referred for cardiac transplantation, a combination of aggressive parenteral therapy, targeted to optimal hemodynamics, followed by conversion to appropriate oral therapy, resulted in clinical improvement so that 30% of these patients were able to be removed from transplant lists.46 Goals of treatment of heart failure can be defined as short-term, to relieve dyspnea and reverse acute hemodynamic decompensation, and long-term, to prevent rehospitalization, improve functional status, and prolong survival. Additional goals include preserving renal function, preventing arrhythmias, and preventing myocardial necrosis in patients with ischemic and nonischemic disease. Pharmacologic therapies to prevent or attenuate chronic remodeling should be instituted or strengthened prior to discharge from the hospital, as these have been shown to improve long-term survival (Box 29.4). Although no randomized clinical trials exist, the use of loop diuretics is supported by a long history of clinical success. These agents increase renal excretion of salt and water. The onset of action of intravenous bolus furosemide is 30 minutes, and the drug peaks at 1 to 2 hours. The half-life of the medication is 6 hours, so twice daily dosing is usually required.39 Other loop diuretics often used are bumetanide and torsemide (Tables 29.2 and 29.3). Several small clinical trials suggested that a constant infusion of a loop diuretic resulted in superior diuresis when compared to intermittent bolus dosing,48,49 although other studies did not confirm this.50 A large, randomized controlled study was performed in patients hospitalized with acute decompensated heart failure who were taking a high dose of furosemide prior to admission. Patients were randomized within 24 hours of admission to bolus dosing or constant infusion, and usual daily outpatient dose (given intravenously) versus high dose (2.5 times their usual daily dose). No significant differences were noted in predefined outcomes between bolus and infusion administration. However, the high-dose group had better relief of dyspnea and more fluid and weight loss than the lower-dose group; also, 23% of the high-dose group had a significant deterioration in renal function, but at 60 days there was no difference in renal function between the two groups.51 Table 29.3 Treatment of Refractory, Diuretic-Resistant Heart Failure An association between high-dose loop diuretics and a worse prognosis has been observed. There is concern that this may be mediated by further activation of the renin-angiotensin-aldosterone system by loop diuretics. However, in a retrospective analysis utilizing propensity matching, hospital mortality rate was not different between low-dose and high-dose diuretic groups.52 It is likely that poorer outcomes associated with high-dose diuretics is not due to the drug itself but reflects a greater severity of heart failure illness with concomitant renal disease.53 Patients with chronic heart failure with or without renal dysfunction may exhibit resistance to loop diuretics, defined as an acute reduction in diuretic efficacy after repeated loop diuretic dosing. With chronic loop diuretic use, there is an increase in sodium reabsorption in the distal nephron and stimulation of aldosterone release. In edematous states there is delayed oral absorption of the drug. With renal dysfunction there may be reduced levels of drug delivered to the renal tubule. This resistance is associated with a poorer prognosis.23 Diuretics that act distally in the renal tubule, such as metolazone or hydrochlorothiazide, or aldosterone blockers such as spironolactone can be added.4 Combination diuretic therapy induces a greater diuresis than simply increasing the dose of loop diuretic further. The response may be delayed for 48 to 72 hours. Combining diuretics often augments diuresis even in the setting of significant chronic kidney disease, and a good response can be expected in over 70% of patients. Particular attention must be given to following potassium, sodium, chloride, and magnesium levels when combination diuretic therapy is prescribed54 (Table 29.4). Table 29.4 Continuous Intravenous (IV) Infusion of Loop Diuretics Adapted from Nieminen MS, Bohm M, Cowie MR, et al: Executive summary of the guidelines on the diagnosis and treatment of acute heart failure. The Task Force on Acute Heart Failure of the European Society of Cardiology. Eur Heart J 2005;26:384-416. The use of novel diuretics, vasopressin inhibitors, has been evaluated in clinical trials of treatment of acute heart failure.55 Vasopressin is a hormone synthesized in the hypothalamus; its major effect is to control free water clearance. It acts through V1a receptors in vascular smooth muscle and myocardium, leading to peripheral and coronary vasoconstriction, myocyte hypertrophy, and positive inotropy. Vasopressin also acts through V2 receptors at the renal tubule collecting ducts to cause free water retention and hyponatremia. Levels of vasopressin are increased in patients with chronic heart failure, and higher vasopressin levels correlate with worse heart failure severity. Vasopressin release is stimulated by changes in serum osmolality and cardiac output and leads to further vasoconstriction and retention of free water.56 Inhibition of vasopressin’s effects would have theoretic benefits in patients with heart failure.57 In contrast to loop diuretics, inhibition of vasopressin theoretically would not cause hypotension or neurohormonal activation, and would not aggravate cardiac arrhythmias due to electrolyte depletion. Conivaptan is a vasopressin antagonist that inhibits V1a and V2 receptors. Tolvaptan and lixivaptan are antagonists selective for the V2 receptor. These medications increase urine volume and free water excretion, with a rise in the serum sodium concentration. The use of conivaptan in patients with class III-IV heart failure was associated with increased urine output, and decreases in PCWP and right atrial pressure, without changes in cardiac output.8 Oral use of tolvaptan was associated with fluid loss and diuresis without change in heart rate, blood pressure, or serum creatinine. In a large, multicenter, placebo-controlled randomized trial called EVEREST, tolvaptan was administered to patients hospitalized with heart failure. There were no adverse consequences on heart rate, blood pressure, or serum electrolytes and there was more rapid improvement in dyspnea and signs of heart failure when compared to usual therapy.58 Hyponatremia improved. Hemodynamic effects included rapid reduction in PCWP and right atrial pressure.59 However, at 10-months follow-up after hospitalization, there was no improvement in mortality rates or readmission rates.60 Vasopressin inhibitors are approved for treatment of severe hyponatremic states but are not approved for use in heart failure. Intravenous vasodilator therapy is often added to diuretic therapy to obtain more rapid improvement in severe heart failure. A clear indication for vasodilators is in patients with severe hypertension and pulmonary edema. Their use should also be considered in patients who are not responding to intravenous diuretics combined with standard oral therapies. Blood pressure response to these medications needs to be carefully monitored because hypotension is a common effect. Improvement in hemodynamics has been obtained with aggressive intravenous vasodilator therapy using intravenous nitroprusside, intravenous nitroglycerin, or nesiritide. The choice of agents depends on matching the patient’s clinical picture and hemodynamics with the predicted effects of each vasodilator.61,62 Intravenous sodium nitroprusside is a powerful venous and arterial dilator. It is a drug of choice in treating hypertension-related heart failure with pulmonary edema and severe heart failure due to acute mitral regurgitation. The use of nitroprusside requires hospitalization in the ICU and invasive monitoring with a PA catheter and arterial line. This drug causes a significant reduction of afterload and preload, leading to decreased right atrial pressure, decreased systemic vascular resistance, decreased mean systemic blood pressure, decreased PCWP, and increased cardiac index in patients with heart failure and left ventricular dysfunction. Limitations of nitroprusside use include inducing a coronary “steal” syndrome in patients with active coronary ischemia.39 In addition, toxic metabolites can accumulate with more prolonged administration. In patients with significant hepatic dysfunction, thiocyanate levels rise, and in patients with renal dysfunction, cyanide is generated. Dosage range is 0.3 to 5.0 µg/kg/minute. Human BNP can be manufactured by recombinant DNA technology and is available as an intravenous medication, nesiritide, for heart failure therapy. BNP is a hormone produced by ventricular and atrial myocytes in response to stretch from cardiac chamber dilatation. Hemodynamic effects include venous and arterial dilation, coronary vasodilation, and natriuresis. Reduction in PCWP and right atrial pressure exceeding the effects of intravenous nitroglycerin, when compared directly, was reported.61 It is not proarrhythmic and does not induce tolerance.63 It may potentiate the effects of loop diuretics. Significant hypotension may limit its use in some patients.39 Because the hypotensive effects of nesiritide are less marked than nitroprusside, nesiritide can be used without invasive hemodynamic monitoring and can be initiated in emergency department settings. Nesiritide is initiated as an intravenous bolus dose of 2 µg/kg followed by infusion of 0.01 µg/kg/minute. Nesiritide was compared to dobutamine in patients with severe heart failure. Nesiritide infusion was associated with less tachycardia and ventricular arrhythmia.64 Other nonrandomized studies suggested a trend toward improved survival and lower rehospitalization rates with nesiritide.63 A meta-analysis of three randomized trials of nesiritide suggested a slight increase in mortality rates in patients given nesiritide versus a placebo control group, possibly mediated through an adverse effect on renal function.65,66
Severe Heart Failure
Definition, Epidemiology, and Staging of Heart Failure
Pathophysiology
Diagnosis
Prognosis in Acute Heart Failure
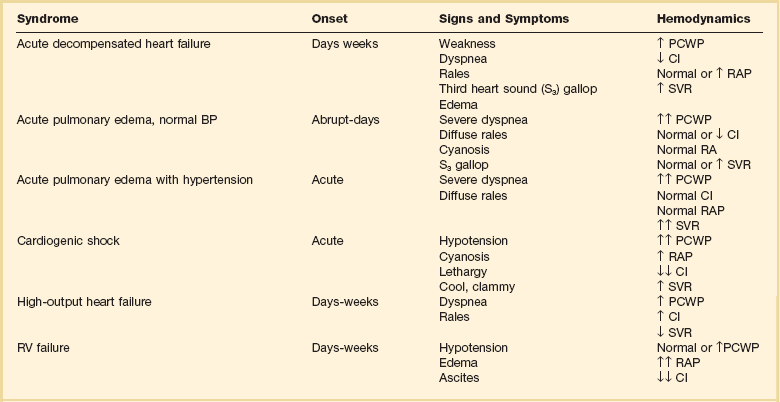
Acute Heart Failure Syndromes
Initial Evaluation and Therapy
Indications for Invasive Hemodynamic Monitoring
Pharmacologic Management of Acute Heart Failure
Intravenous Diuretics
To Loop Diuretic, add:
Dose
Hydrochlorothiazide
25-50 mg once or twice daily
or metolazone
2.5-5.0 mg once or twice daily
or spironolactone
12.5-50 mg once daily
Diuretic
Dose
Bumetanide
1 mg IV load, then 0.5-2 mg/hr infusion
Furosemide
40 mg IV load, then 10-40 mg/hr infusion
Torsemide
20 mg IV load, then 5-20 mg/hr infusion
Vasopressin Inhibitors
Parenteral Vasodilators (Table 29.5)
Nitroprusside
Nesiritide (B-natriuretic Peptide)

Full access? Get Clinical Tree


Severe Heart Failure