Fig. 10.1
The “oxygen cascade” with partial pressure of oxygen (PO 2 ) falling from ambient air to the mitochondria. Factors affecting oxygenation at different levels are shown at the top (gray arrows). Modified from Meyhoff et al. [2]
However, oxygen is a drug that should be administered only when indicated (mainly for hypoxemia) and its dosage should be carefully evaluated with the aim to obtain a normal oxygen concentration in arterial blood [2].
Nevertheless, several studies published in the last few years demonstrated that high perioperative FiO2 reduces postoperative nausea and vomiting, promotes healing, and prevents surgical site infection (SSI) and morbidity following high-risk surgery [3].
Moreover, a recent meta-analysis of small randomized controlled trials suggested that perioperative supplemental oxygen might reduce mortality in colorectal surgery [4]. It would be very attractive if such a simple, worldwide applicable and considerably inexpensive (less than 0.001 cents/liter) [5] intervention could affect surgical mortality. Maybe, the matter is somewhat more complex. First of all, benefits and limitations of supplemental oxygen for preventing SSI are still controversial [6]. Therefore, the putative mechanism of reduced mortality is not clearly understood. Finally, some concerns exist on safety about breathing high oxygen fractions for several hours and new insights into the potential side effects of hyperoxia have been recently achieved.
10.2 Main Evidences
Despite advances in understanding perioperative factors affecting the risk of SSI, such as appropriate prophylactic antibiotic use, glycemic control, management of body temperature, and postoperative pain [6, 7], SSI remains very frequent, accounting for 15–17 % of all nosocomial infections [4, 7], and is still associated with higher costs, prolonged hospital stay, increased intensive care unit admissions, and higher morbidity and mortality [5, 8].
Given the existing relationship between pO2 and both wound infection and healing [2, 5, 8] (see below), several studies in the last years focused on the role of high perioperative FiO2 in reducing SSI rates.
The meta-analysis by Brar et al. [4] is the only investigation showing a survival benefit with perioperative supplemental oxygen [9]. It included five studies, published between 2000 and 2007, reporting its effects on the incidence of SSI in colorectal patients. In all studies, the treatment group received 80 % oxygen intraoperatively. This concentration was maintained postoperatively in four of the five studies, precisely for 2 postoperative hours in three of the studies and for 6 postoperative hours in one of them. Control groups received 30 % oxygen in four of the five investigations and 35 % oxygen in one of them. All papers but one reported data on mortality. These studies included 615 patients in the supplemental oxygen group and 589 in the control group, and the overall mortality rate was 0.2 and 2.0 %, respectively, with statistical analysis significantly favoring the treatment arm (p = 0.01).
Nevertheless, this meta-analysis failed to clearly demonstrate a reduction in SSI in the treatment population, and this is consistent with the conflicting results of previous and following literature.
In fact, despite previous meta-analyses [10, 11], a recent randomized controlled trial [12] and two subgroup analyses from a meta-analysis published in 2012 [7] reported a beneficial effect of supplemental oxygen on SSI rates, most of these results must be interpreted with caution. For example, the meta-analysis by Chura et al. [10] does not conform to QUOROM guidelines [4], the one by Al-Niaimi et al. [11] presents a high degree of heterogeneity, [7] while the trial by Bickel et al. [12], reporting a beneficial effect of 80 % oxygen given during and 2 h after appendicectomy, presents several questionable aspects, the most important of which is the early interruption of the study [2, 13]. Furthermore, many other investigations showed no benefits of supplemental oxygen in reducing SSI after abdominal, gynecologic, obstetric, breast, and cardiac surgery [6, 7, 14–16]. One of the largest trials to date [16] found no significant difference in SSI rate after randomizing 1,400 patients to receive 80 or 30 % oxygen during and 2 h after abdominal surgery. A multicenter trial [6] and a meta-analysis [7] published in 2012 showed no reduction in SSI rates with a perioperative FiO2 of 0.8, even if two subgroup analyses within the meta-analysis (trials that excluded neuraxial anesthesia and trials that involved colorectal surgery only) showed a decreased SSI rate with high FiO2.
Considering the lack of agreement among data regarding the beneficial effect of supplemental oxygen in preventing SSI, the reduced mortality observed by Brar et al. [4] cannot be clearly nor exclusively attributed to the reduction in SSI, although a contribution from a reduced SSI-related mortality cannot be excluded. However, as these authors themselves suggest, a plausible explanation for their finding may be the prevention of unrecognized hypoxia at the time of surgery or in the early postoperative period, that in a minority of patients not receiving “supplemental” oxygen may contribute to both regional and systemic imbalance between oxygen supply and demand (that is what the term “hypoxia” rather implies), possibly leading to focal ischemia–reperfusion injury with subsequent oxidative stress accounting for increased capillary permeability, organ failure, and cardiopulmonary complications [3]. Accordingly, several reports indicate that poor outcomes after major surgery are strongly associated with derangements in tissue oxygen delivery (DO2) [17], while many investigations suggest that perioperative interventions aimed to optimize oxygen delivery and especially utilization (expressed by parameters such oxygen extraction ratio, central and mixed venous oxygen saturation) (Fig. 10.2) may reduce mortality [17, 18].
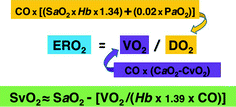

Fig. 10.2
Some parameters reflecting the balance between oxygen delivery (DO 2 ) and consumption (VO 2 ) and their determinants. ERO 2 Oxygen extraction ratio, SvO 2 Mixed venous oxygen saturation, CO Cardiac output, SaO 2 Arterial oxygen saturation, Hb Hemoglobin concentration, PaO 2 Arterial partial pressure of oxygen, CaO 2 Arterial oxygen content (Hb × 1.39 × SaO2), CvO 2 Mixed venous oxygen content (Hb × 1.39 × SvO2)
10.3 Pharmacologic Properties
The role of hyperoxia in reducing SSI is biologically plausible. In fact, the oxidative killing of bacteria by neutrophils, representing one of the major defenses against SSI, is dependent on a respiratory burst that involves the production of superoxide anion and other reactive oxygen species (ROS) (Fig. 10.3) [6–8]. Therefore, the bactericidal activity of neutrophils can be substantially impaired, because of limited substrate, in a low-oxygen environment such as the wound one, where local vascular supply is disrupted by surgical trauma, thrombosis, or edema. Conversely, hyperoxia increases ROS production [2, 5, 7]. Oxygen is also critical for wound healing because it enhances neovascularization, epithelialization, and collagen synthesis, since both hydroxylation of proline and lysine and cross-linking of collagen are pO2-dependent. Finally, hyperoxia may act as an important cell signal involved in the activation of immune response by interacting with tumor necrosis factor α [2, 5, 8].






