CHAPTER 77
Neurolytic Blocks
INTRODUCTION
Dogliotti1 first described the technique of subarachnoid chemical neurolysis using alcohol for the treatment of sciatic pain more than 80 years ago. That same year, Suvansa2 described intrathecal carbolic acid for the treatment of tetanus. A quarter of a century later, Maher3,4 in his 2 landmark articles described his experience with hyperbaric phenol and silver nitrate for subarachnoid neurolysis, stating, “It is easier to lay a carpet than to paper a ceiling,” an obvious reference to his understanding of the baricity properties of alcohol and phenol (carbolic acid). Over the ensuing years, however, lack of experience with either technique and fear of the anticipated complications resulted in underuse of this neurolysis, especially when applied to the subarachnoid space. Better understanding and increased use of neuraxial opiates for cancer pain since the 1980s have decreased the use of subarachnoid chemical neurolysis even further. Nonetheless, because of the physical separation of the sensory and motor roots of spinal nerves within the spinal canal, intrathecal dorsal rhizotomy (more appropriately called rhizolysis) is the only neurolytic procedure that allows sensory block without concomitant motor block. Because of this and because of the relative precision with which the affected nerve roots can be blocked, the technique is particularly useful for treating cancer pain in an extremity, where preservation of motor function is so important. In short, the physical separation of motor and sensory fibers in the subarachnoid space preserves forever a small but unique role for subarachnoid neurolysis in the management of cancer pain in carefully selected patients. The present chapter will describe the techniques of:
• Trigeminal neurolysis
• Lumbar sympathetic (LSNB)
• Celiac plexus (CPB) neurolysis
• Ganglion impar and superior hypogastric plexus neurolysis
• Sacroiliac joint (SIJ) neurolysis
• Spinal and epidural including transforaminal neurolysis
INDICATIONS
Neurolysis may be used to manage recalcitrant pain syndromes of both a malignant as well as benign etiology. Techniques have been described for blocking different types of pain:
• Pain from the cranial nerves (particularly the trigeminal nerve; CN V)
• Pain originating in the major plexuses (brachial and lumbar)
• Pain associated with sympathetic-medially pain (especially the celiac plexus, lumbar sympathetic ganglia, ganglion impar, and superior hypogastric plexus)
• Pain originating in the periphery (sacroiliac joints)
Before considering the use of chemical or thermal neurolysis, however, it behooves the clinician to ascertain that every alternative means of providing analgesia has been investigated with the patient. By virtue of their irretrievable nature, chemical neurolytics are exceedingly unforgiving when placed in contact with nonintended, nontargeted tissues. For these reasons, a caveat to the successful use of neurolytic blocks is a predetermined analgesic response, which is unequivocal following a local anesthetic block of the same targeted structure(s).
AGENTS
• In general, absolute alcohol or phenol may be selected for chemical neurolysis.
• Alcohol has been the drug of choice for subarachnoid block, cranial nerve block, and celiac plexus block.
• Phenol finds utility for lumbar sympathetic nerve blocks, peripheral neurolysis like SIJ block, Ganglion impar, and superior hypogastric plexus blocks, although various authorities have used either drug for any of the aforementioned indications.
SELECTION CRITERIA
Because the duration of action of neurolytic agents is finite but unpredictable, great care must be exercised in choosing appropriate candidates for these procedures. Neurolytic blockade is especially suited to patients in whom conventional treatment regimens have failed and who have a short life expectancy, usually less than 1 year, when the subarachnoid or epidural route is selected. More often than not, neurolysis at other sites has been successfully used in individuals who do not have an imminently grave prognosis. As with all neurolytic procedures, including electrical and thermal neurolysis, patients must be completely apprised of the possibility of debilitating side effects and other serious associated complications that can follow even a successful block, most notably, motor weakness and incontinence. The selection criteria for subarachnoid neurolysis include the following:
• The diagnosis is well established.5
• The patient’s life expectancy is short, usually 6 to 12 months.
• The patient’s pain is unresponsive to antineoplastic therapy (chemotherapy, radiation).
• The patient’s pain has failed to respond to adequate trials of analgesic agents and adjunctive drugs.
• The pain is localized to 2 or 3 dermatomes.
• The pain is predominantly somatic in origin.
• The pain is unilateral (neurolytic blocks for bilateral pain should be staggered).6
INFORMED CONSENT
• It is crucial that not only the patient, but also the patient’s family fully understand the anticipated procedure, its potential risks, the alternative forms of therapy available, and, most importantly, the possibility of serious complications.
• It is important that they understand that the procedure does not simply “take away pain,” but rather substitutes numbness (loss of sensation) for the pain.
• So important is this concept that, with rare exceptions, before the decision is made to proceed with a subarachnoid neurolytic block, a prognostic block should be carried out using a local anesthetic so that the patient can experience the pain relief that may be anticipated after a neurolytic block and the accompanying sensory block.
• Although an occasional patient may decide that he or she cannot tolerate the numbness, most patients prefer this lack of sensation to the pain and choose to proceed with the neurolytic procedure
PREOPERATIVE CONSIDERATIONS
Neurolytic blocks are usually considered in patients for whom the analgesic ladder has failed to provide adequate analgesia. However, the pros and cons for each procedure, including a detailed knowledge of potential complications, and other factors such as the speed of pain relief, and the improvement of quality of life need to be weighed before making the decision. In most scenarios, diagnostic block performed prior to the therapeutic neurolytic blocks is recommended. Temporary unexpected adverse effects can occur due to unexpected spread of local anesthesia during diagnostic block; however, serious complications may follow injection of neurolytic agents.
The following procedures need to be done routinely for preoperative considerations:
• Obtaining an informed, written consent.
• Ascertaining the absence of contrast allergies and no use of anticoagulant medications.
• It is useful to refer to standard references such as those published by the American Society of Regional Anesthesia (ASRA) concerning anticoagulants and regional anesthesia prior to commencing to determine the suitability of prospective block candidates prior to proceeding.
• Standard American Society of Anesthesiologists (ASA) monitors are applied and baseline vital signs are assessed and documented.
• An intravenous canula should be placed for the purposes of administering intravenous fluids and supportive drugs in the event of either an inadvertent event or following the expected orthostatic hypotension resulting from a successful block.
DOCUMENTATION FOR OFF-LABEL INDICATIONS
All candidates for whom peripheral or central neurolysis is contemplated must understand that the use of phenol and alcohol for these procedures represents an off-label use of these chemical agents. This critical information must be made part of the written informed consent process.
TRIGEMINAL NEUROLYSIS (FIGURES 77-1 TO 77-9)
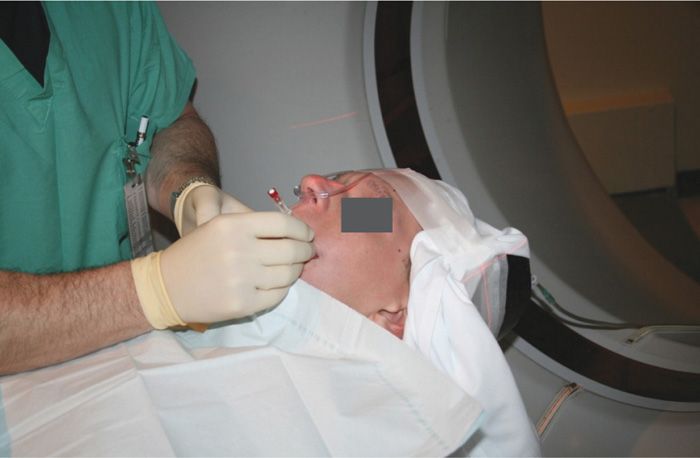
Figure 77-1. Neurolysis using CT-scan guidance for trigeminal neuralgia. Patient in position to enter the CT scanner. A left-sided subzygomatic approach to the foramen ovale is demonstrated. (Used with permission from Kenneth D. Candido, M.D.)
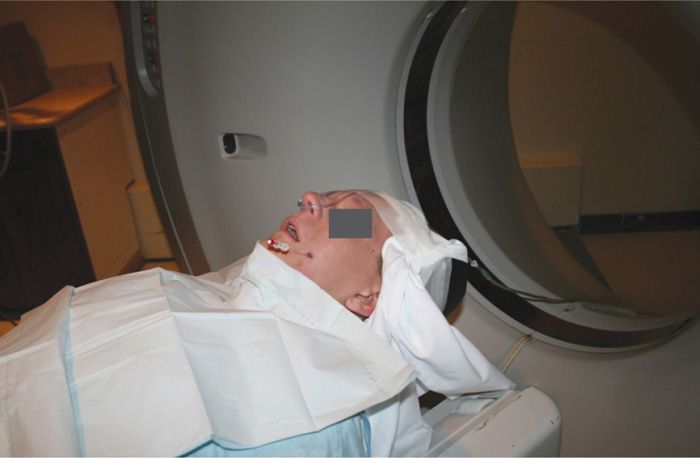
Figure 77-2. The needle has been placed for left-sided CT-scan guided neurolytic TGN block; patient about to enter the scanner. (Used with permission from Kenneth D. Candido, M.D.)
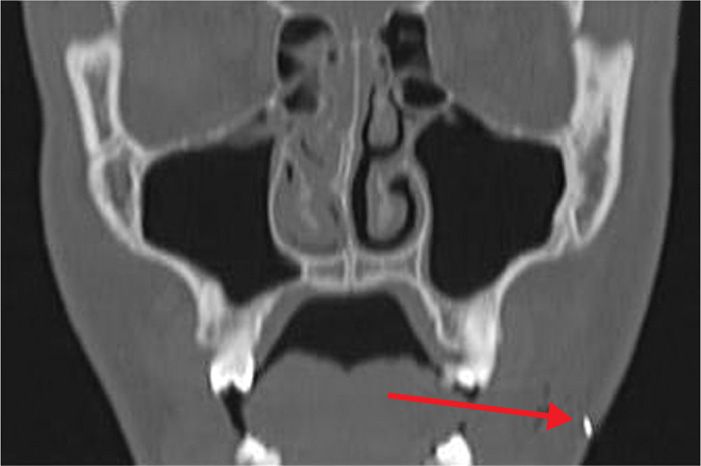
Figure 77-3. Coronal CT scan view of advancing needle tip for CT-guided neurolytic TGN block, demonstrated by arrow (coronal view). (Used with permission from Kenneth D. Candido, M.D.)
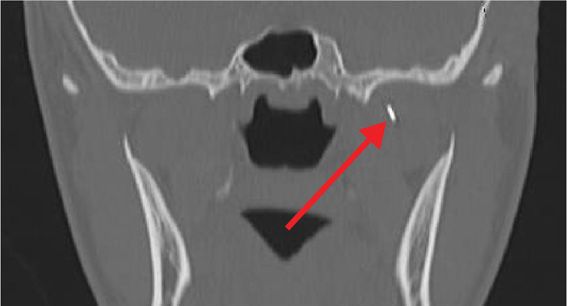
Figure 77-4. Coronal view of needle tip advancing toward foramen ovale for neurolytic TGN block; tip denoted by arrow (coronal view). (Used with permission from Kenneth D. Candido, M.D.)
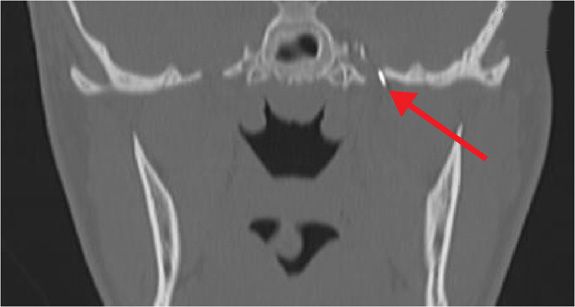
Figure 77-5. Needle tip entering foramen ovale for neurolytic TGN block (coronal view). (Used with permission from Kenneth D. Candido, M.D.)
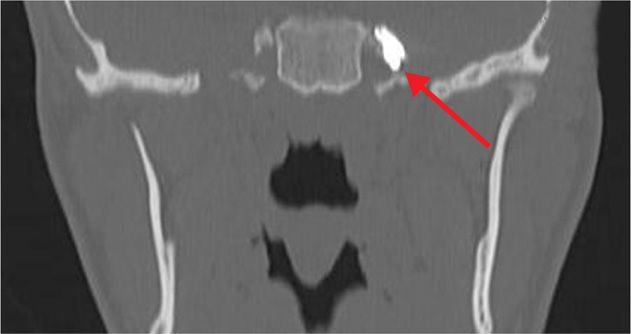
Figure 77-6. 1-mL iodine-based, water-soluble contrast injected through foramen ovale into Meckel cave as prelude to neurolytic TGN block (coronal view). (Used with permission from Kenneth D. Candido, M.D.)
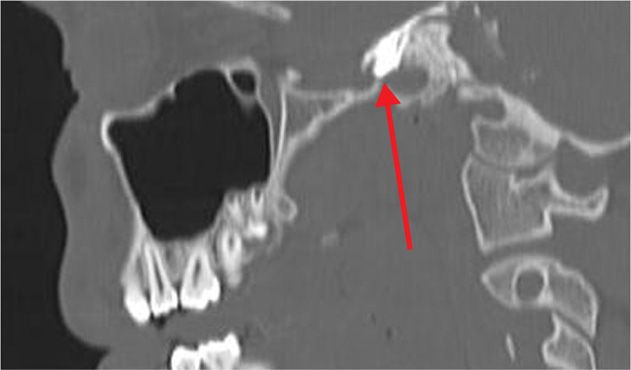
Figure 77-7. Sagittal CT scan view of the same view as in Figure 77-6 with contrast injected through the foramen ovale as a prelude to neurolytic TGN block (sagittal view). (Used with permission from Kenneth D. Candido, M.D.)
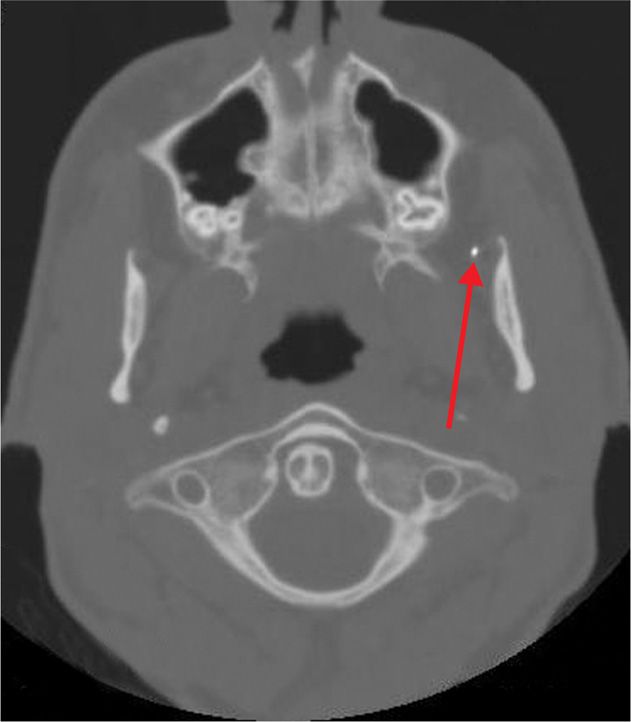
Figure 77-8. Axial CT view of same patient showing advancing needle tip heading toward foramen ovale for neurolytic TGN block (axial view). (Used with permission from Kenneth D. Candido, M.D.)
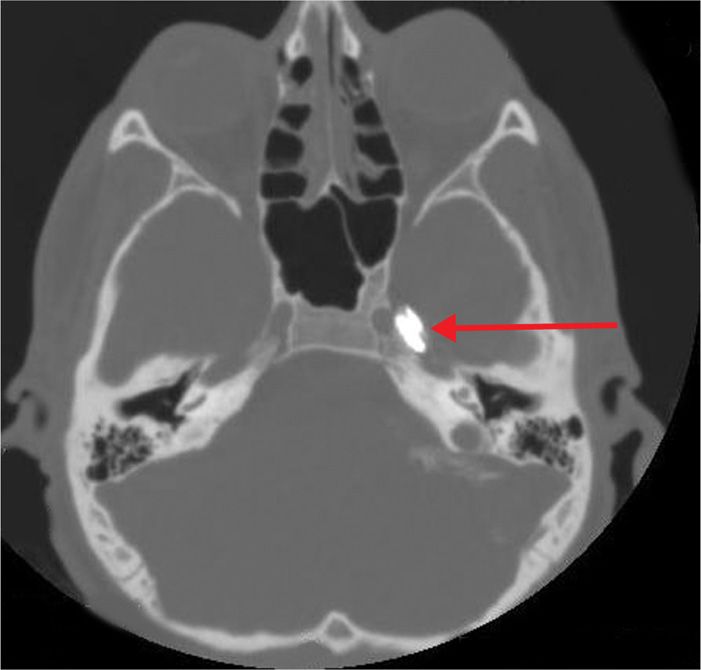
Figure 77-9. Contrast in Meckel cave as prelude to neurolytic TGN block; same patient as in Figure 77-8 (axial view). (Used with permission from Kenneth D. Candido, M.D.)
Relevant Anatomy
The trigeminal nerve resides in Meckel ave, located within the foramen ovale. Recalcitrant V1, V2, or V3 pain which has been shown to be responsive to local anesthetic blocks may be an indication for alcohol neurolysis of the Gasserian ganglion.
Preoperative Considerations
• It is imperative that patients are offered surgical options including microvascular decompression before seeking to undertake neurolysis.
• An informed consent must include discussion of the risks of bleeding, paralysis, and death. Patients need to be evaluated by physicians to exclude all contraindications and must have responded favorably to one or more local anesthetic diagnostic blocks performed at previous visits.
Contraindications
• Patient who has bleeding problems
• Taking irreversible inhibitors of platelet aggregation
• Has history of allergy to iodine-based contrast agents
• Has an infection at the injection site
• Patient who is unable to remain motionless during the procedure, which may cause unexpected injury during the intervention
Fluoroscopic Views
• The procedure may be performed using either fluoroscopic or CT guidance, and rarely by using ultrasound guidance.
• It is the recommendation of the present authors that only CT guidance is used for these patients, both to ascertain that the medication is successfully placed at the target site as well as to preserve a clear and unmistakable medical record in the case of an unforeseen outcome or inadvertent spread of agent to nontargeted tissues.
Positioning of Patient
• Supine position
Selection of Needles, Medications, and Equipment
• A fine-gauge hypodermic needle and 2 mL of 1% lidocaine plain will be used for local anesthesia.
• A 22- or 25-gauge Whitacre-type subarachnoid needle, 10 to 15 cm long with a distal curve and 1 mL of absolute alcohol will be used for the neurolysis block.
Intraoperative Technique Steps
• The patient is placed supine and a full complement of American Society of Anesthesiologists (ASA) monitors is applied. Baseline vital signs are assessed and recorded.
• The skin is cleansed using chlorhexidine.
• A skin wheal is raised approximately 3 cm lateral to the corner of the closed mouth using a fine-gauge hypodermic needle and 2 mL of 1% lidocaine plain.
• A 22- or 25-gauge Whitacre-type subarachnoid needle, 10 to 15 cm long with a curve placed at the distal tip is then advanced from anterior to posterior, and slightly medial and cephalad, aiming toward the ipsilateral pupil.
• Once the needle has been advanced more than 4 cm, the patient is scanned to identify the direction of the needle in relationship to the foramen ovale (Figures 77-3 to 77-9).
• Rostral, axial, and sagittal views with reconstruction are used to properly position the needle tip.
• Once within Meckel ave, the stylet of the needle is withdrawn, and an assessment is made for cerebrospinal fluid.
• If there is CSF aspirated, the needle is slightly withdrawn until the flow ceases.
• If there is no CSF aspirated, one mL of high-density, water-soluble, iodine-based contrast is injected.
• An assessment is made to determine the spread of contrast.
• If the spread appears adequate and there is no vascular or CSF run-off, 1 mL of absolute alcohol may now be injected, slowly and incrementally.
• The patient must be cautioned that this may burn somewhat, in the distribution of the ophthalmic, maxillary, or mandibular nerves, respectively.
• The needle is then flushed with 0.5 mL of air and is withdrawn.
• Following the procedure, the patient must be observed in a monitored setting for no less than 1 hour to assess the full effects of the procedure and to determine if there have been any adverse effects from the block.
Postprocedure Considerations
• The patient must be observed in a monitored setting for at least 1 hour to assess the full effects of the procedure and to determine if there have been any adverse effects from the block.
Monitoring of Potential Complication
The following complications from the blocks which may occur include:
• Numbness, hypesthesia, or dysesthesia in the entire trigeminal nerve or in one of its branches (V1, V2, V3)
• Reactivation of a dominant herpes simplex
• Corneal abnormalities such as absence of corneal reflex, ulceration, and keratitis
• Bleeding at the injection site or localized pain
• Intracranial hemorrhage
• Infection
• Injection into the foramen spinosum in lieu of the ovale
LUMBAR SYMPATHETIC NEUROLYSIS (FIGURES 77-10 TO 77-13)
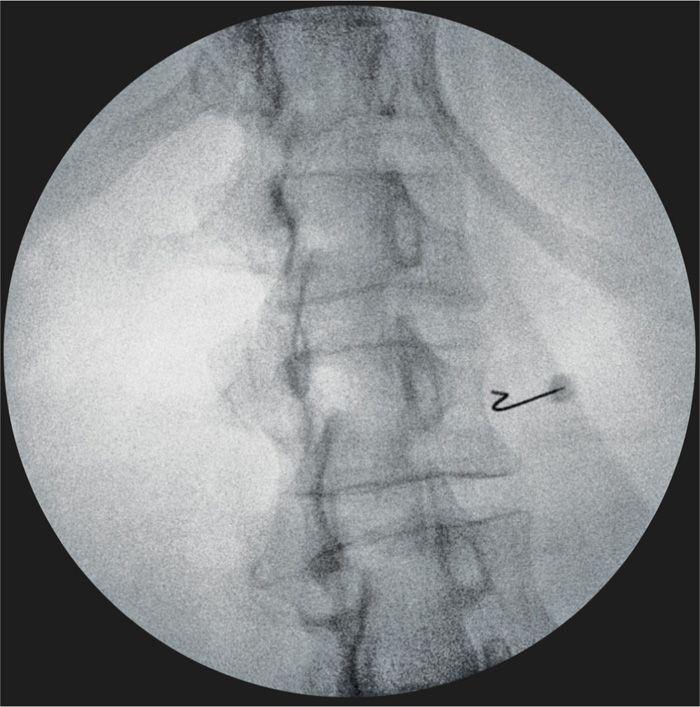
Figure 77-10. Lumbar sympathetic neurolytic block at L2. Curved needle advancing around lateral aspect of the right L2 vertebral body; oblique fluoroscopy image. (Used with permission from Kenneth D. Candido, M.D.)
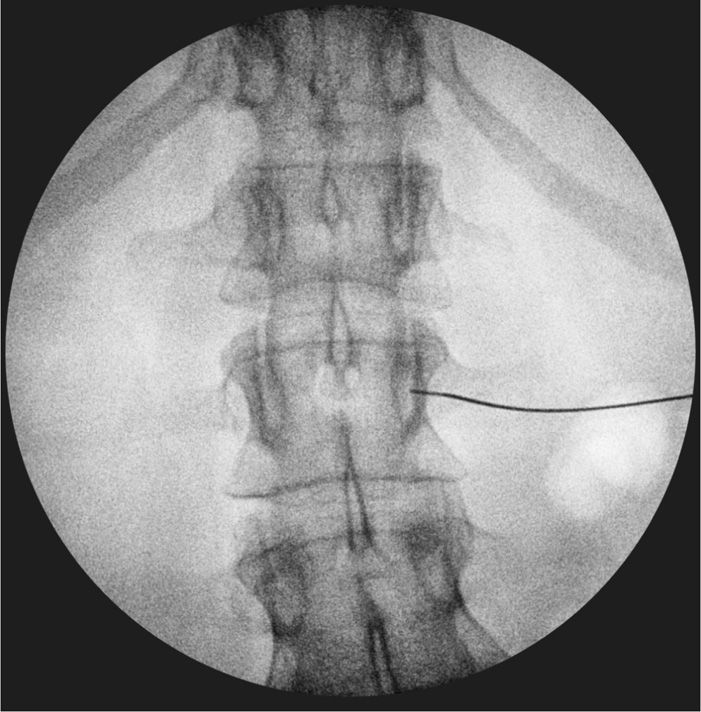
Figure 77-11. Anteroposterior fluoroscopy view of needle seated at level of pedicle, anterior, and lateral to vertebral body at the right L2 for neurolytic LSNB. (Used with permission from Kenneth D. Candido, M.D.)
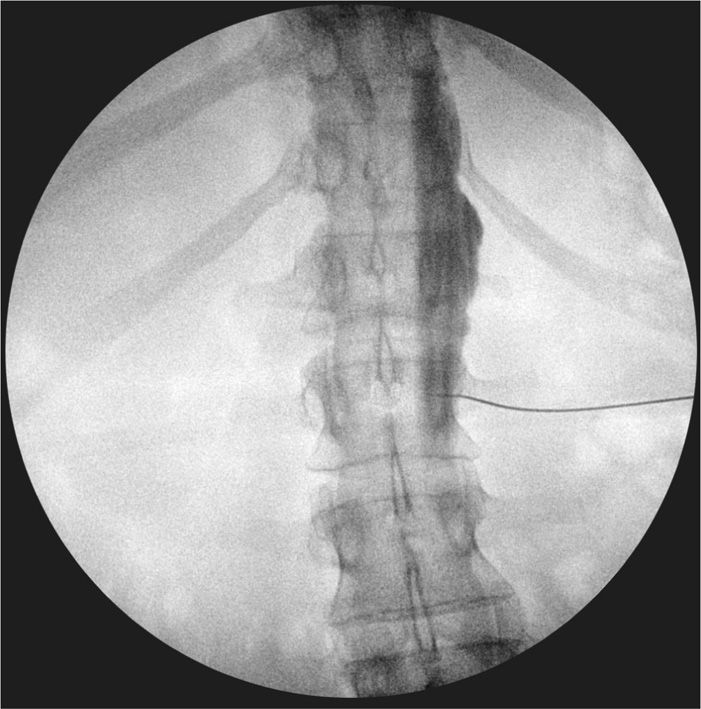
Figure 77-12. Anteroposterior view of 5-mL iodine-based, water-soluble contrast injected at L2 for neurolytic LSNB on the right side demonstrating right-sided spread. (Used with permission from Kenneth D. Candido, M.D.)
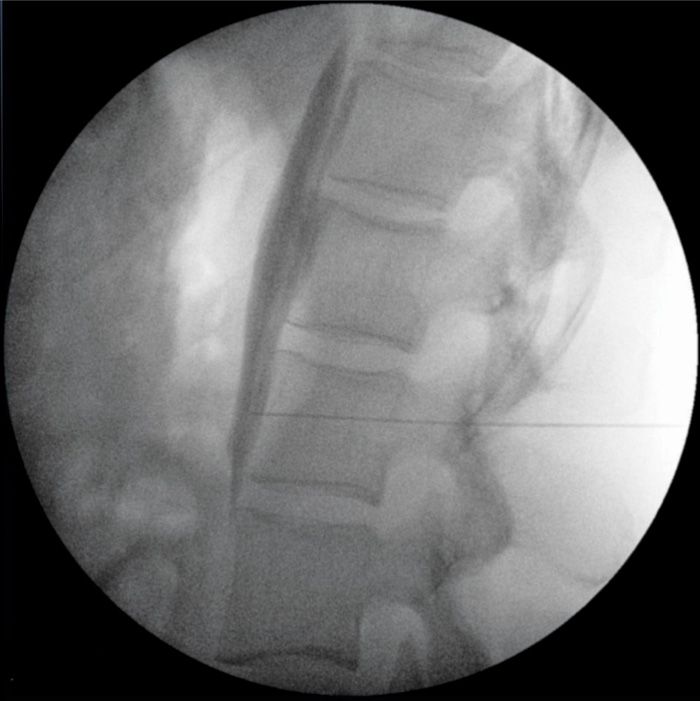
Figure 77-13. Lateral fluoroscopic view, neurolytic LSNB; contrast injected as a prelude to injecting phenol; tight linear spread along ventral margin of multiple vertebral bodies without extravasation or spread ventrally or dorsally. (Used with permission from Kenneth D. Candido, M.D.)
Relevant Anatomy
Individuals with sympathetically-mediated pain occasionally fail to derive benefit from repeated local anesthetic nerve blocks of the sympathetic ganglia. The ganglia, which are paired, may number from 3 to 4 and may be as long as 1.5 cm.
Preoperative Considerations
When patients have transient responses to local anesthetic blocks and wish not to undergo trial spinal cord stimulation, they may occasionally be considered candidates for lumbar sympathetic neurolysis. An informed consent must include discussion of the risks of psoas muscle injection, hematoma, femoral nerve neuropathy with quadriceps weakness, and vascular injury. Male patients should be advised that they may also develop retrograde ejaculation after procedure as genitofemoral neuralgia may occur up to 25% of the time.
Contraindications
• Patient who has bleeding problems
• Patient who is taking irreversible inhibitors of platelet aggregation
• Patient who has history of allergy to iodine-based contrast agents
• Patient who has an infection at the injection site
• Patient who is unable to remain motionless during the procedure which may cause unexpected injury during the intervention
Fluoroscopic Views
• While ultrasound may be used, due to the severe potential consequences of a misplaced agent, it is likely that fluoroscopy or CT imaging provide a more detailed assessment of the anatomical confines required for successful blockade.
• A scout anteroposterior (AP) film of the lumbar spine will be obtained, and then the 20-degree oblique orientation fluoroscope view will be taken.
Positioning of Patient
• Prone position; alternative is lateral decubitus or modified Sims positioning
Selection of Needles, Medications, and Equipments
• A fine-gauge hypodermic needle and 1% lidocaine, 2 to 5 mL will be used for raising a skin wheal.
• A 22- or 25-gauge Whitacre-type subarachnoid needle with a curve at the distal tip and 5 to 15 mL of 5% to 7% phenol in glycerin is used for neurolytic block.
Intraoperative Technical Steps
• The patient is placed in the prone position; lateral decubitus; or modified Sims position.
• An intravenous cannula is placed and standard ASA monitors are applied.
• A full surgical sterile skin preparation is undertaken.
• While ultrasound may be used, due to the severe potential consequences of a misplaced agent, it is likely that fluoroscopy provides a more detailed assessment of the anatomical confines required for successful blockade.
• A scout anteroposterior (AP) film of the lumbar spine is obtained (Figure 77-10), and then the fluoroscope is placed in the 20 degree oblique orientation (Figure 77-11).
• Blocks may be performed at L2, L3, or L4, with the most traditional level being selected as L2.
• After raising a skin wheal using a fine-gauge hypodermic needle and 1% lidocaine, 2 to 5 mL, a 22- or 25-gauge Whitacre-type subarachnoid needle with a curve at the distal tip is advanced from posterior to anterior and medially until the tip sits at the anterior-lateral margin of the L2 vertebral body, juxtaposed at the ventral one-third of the vertebral body.
• At this point, water-soluble, iodine-based contrast is injected.
• If the contrast does not adhere to the ventral surface of the vertebral column in a tight, linear array, the needle should be positioned until such a result is attained.
• Once needle placement is considered successful, 5 to 15 mL of 5% to 7% phenol in glycerin is incrementally injected, the final volume being determined to mimic the volume which was used to create the observed pattern of x-ray contrast spread.
• The needle is then flushed with 1 mL of air and is withdrawn.
Postprocedure Considerations
• Upon completion of the procedure, the patient is observed in a monitored setting for a minimum of 1 hour and is cautioned to arise slowly from a supine position to minimize the likelihood of developing orthostatic hypotension, a common finding following even unilateral LSNB neurolysis.
Monitoring of Potential Complications8
• The most common complication is neuralgia, which occurs due to unexpected spread of the neurolytic agents over the genitofemoral nerve
• Postdural puncture headache
• Bleeding at the injection site or localized pain
• Epidural, subdural, or subarachnoid injection
• Vascular complication such as hypotensive collapse
• Intra-aortic or intra-vena caval injection
• Thrombosis, embolization, infarction
• Paralysis
• Bladder, bowel, or sexual dysfunction
• Death
CELIAC PLEXUS NEUROLYSIS (FIGURE 77-14)
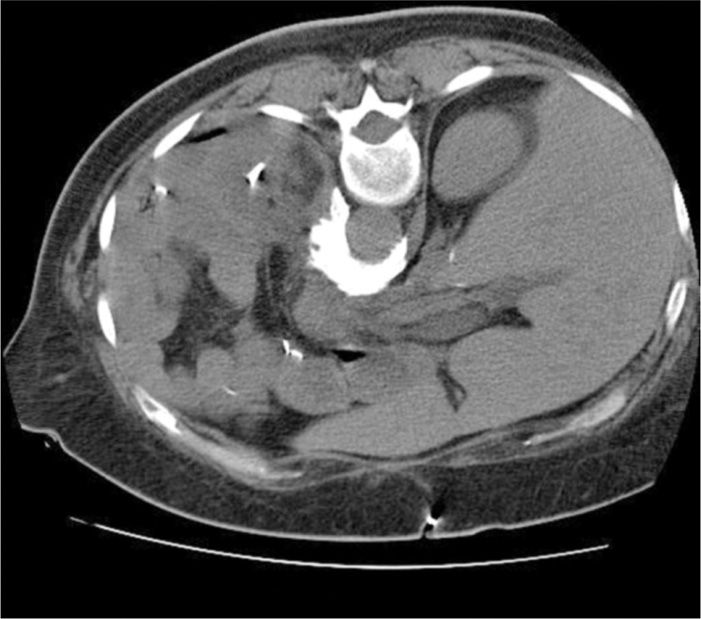
Figure 77-14. Axial CT-scan image of contrast spread following 20-mL injection; neurolytic CPB (refer to chapter on Celiac Plexus Block for additional details and images). (Used with permission from Kenneth D. Candido, M.D.)
Celiac plexus blocks, including neurolytic CPB, have been detailed in the chapter dedicated to that procedure. CPB neurolysis may be used for both malignant as well as for benign conditions, most commonly chronic pancreatitis. Fluoroscopy, CT-scan imaging, or endoscopic ultrasound use may be employed as adjuncts to the block. Each of these modalities has been well described.
GANGLION IMPAR NEUROLYSIS (FIGURES 77-15 TO 77-17)
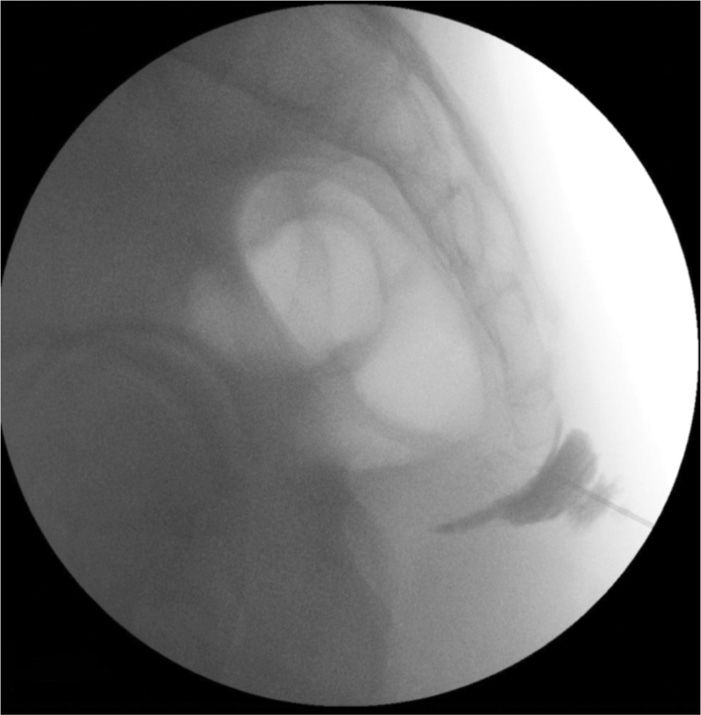
Figure 77-15. Ganglion impar neurolytic block using fluoroscopic guidance; needle approaches sacrococcygeal junction; contrast injected outside junction. (Used with permission from Kenneth D. Candido, M.D.)
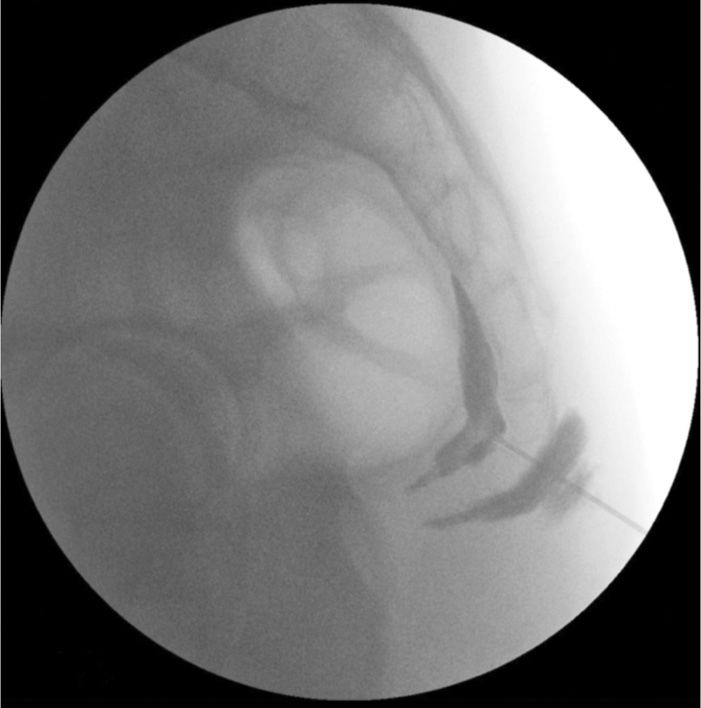
Figure 77-16. Ganglion impar neurolytic block after needle has passed through sacrococcygeal junction; additional contrast injected outlining ventral sacral margin; rectal gas pattern undisturbed by needle tip. (Used with permission from Kenneth D. Candido, M.D.)
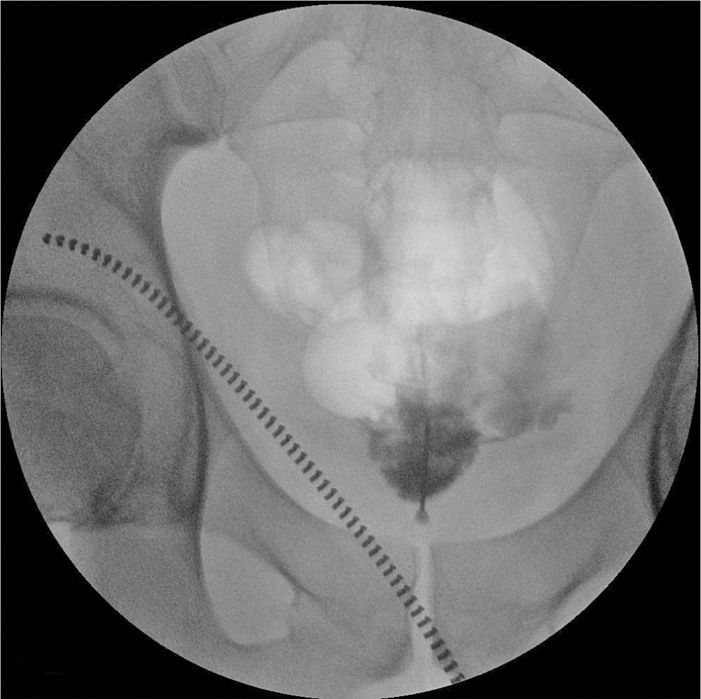
Figure 77-17. Ganglion impar neurolytic block; anteroposterior fluoroscopic view. (Used with permission from Kenneth D. Candido, M.D.)
Occasionally pelvic pain may prove resistant to conventional attempts to reduce its intensity and severity. Under these circumstances, or in the case of a malignant process, such as rectal, bladder, or cervical cancer causing the pain, a neurolytic procedure may be considered. Visceral or SMP in the perineal area associated with malignancies of the pelvic area produces vague, burning, localized perineal pain; and often urgency of urination or defecation.
Relevant Anatomy
The Ganglion of Walther is the solitary terminal ganglion of the sympathetic chain located anterior to the sacrococcygeal junction.
Preoperative Considerations
When one or a series of local anesthetic diagnostic blocks prove successful in reducing the pain, albeit temporarily, neurolysis may be considered. An informed consent must include discussion of the risks of rectal perforation, infection, bleeding and hematoma formation, and neural injury.
Contraindications
• Patient who has bleeding problems
• Patient who is taking irreversible inhibitors of platelet aggregation
• Patient who has history of allergy to iodine-based contrast agents
• Patient who has an infection at the injection site
• Patient who is unable to remain motionless during the procedure which may cause unexpected injury during the intervention
Fluoroscopic Views
• The most commonly used technique for ganglion impar neurolysis is the trans-sacrococcygeal approach with fluoroscopy by introducing the needle through the sacrococcygeal joint (SCJ).
• Ultrasound guidance is not widely used as it cannot accurately identify the depth of needle penetration because of some sacral and coccygeal bony artifacts. In addition, impacted stool or gas in the rectum is possible to make it difficult to recognize the SCJ in the anteroposterior fluoroscopy view. However, there is literature support recommending using the lateral fluoroscopy view to identify the depth of needle and the spread of the neurolytic agents.
Positioning of Patient
• Jack-knife position
Selection of Needles, Medications, and Equipments
• A fine-gauge hypodermic needle and 3 to 5 mL of 1% lidocaine with epinephrine 1:200,000 (5 μg/mL) will be used to raise a skin wheal.
• A 22-gauge, 3.5-in (8.89-cm) Quincke-type subarachnoid needle and 3 to 5 mL of 5% to 7% phenol in glycerin will be used for neurolytic blocks.
Intraoperative Technical Steps
• The procedure is performed with the patient prone, preferably in the jack-knife position.
• As the anatomical boundaries are an extension of those required to perform caudal epidural injections, most interventionalists ought to be able to identify the sacrococcygeal junction. This is situated distal to the sacrococcygeal ligament, along the same cephalad-caudal axis.
• Due to the proximity to the perineum, a comprehensive skin cleansing needs to be undertaken, using chlorhexidine.
• A skin wheal is raised after first identifying the anatomical boundaries of the sacrum, using a fine-gauge hypodermic needle and 3-5 mL of 1% Lidocaine with epinephrine 1:200,000 (5 μg/mL).
• Using a 22-gauge, 3.5 inch (8.89 cm) Quincke type subarachnoid needle, the sacrococcygeal junction is penetrated, taking care to not penetrate the rectum.
• Alternatively, a double-curved needle may be used to enter the skin more distally and advance through the anococcygeal ligament (between the anus and tip of the coccyx) toward the ventral surface of the sacrum, under continuous fluoroscopic guidance.
• Once the needle is appropriately situated, 3 to 5 mL of contrast may be injected.
• If the contrast spread adheres tightly to the ventral surface of the sacrum, an equivalent volume of 5% to 7% phenol in glycerin may then be incrementally injected.
• The needle is then flushed with 1 mL of air and is withdrawn.
Postprocedure Considerations
• The patient should be observed for 30 to 60 minutes in a monitored setting following the completion of the procedure to be certain that there have been no adverse effects from the neurolytic agent.
Monitoring of Potential Complications
• Even though there was no definitive literature description of the serious complication in patients who received the ganglion impar neurolytic block, as with most procedures, there is the theoretical risk of pain at the injection site, localized bleeding, infection, nerve injury, puncture of internal vessels or organs, paralysis, and death.
SUPERIOR HYPOGASTRIC PLEXUS NEUROLYSIS (FIGURES 77-18 TO 77-21)
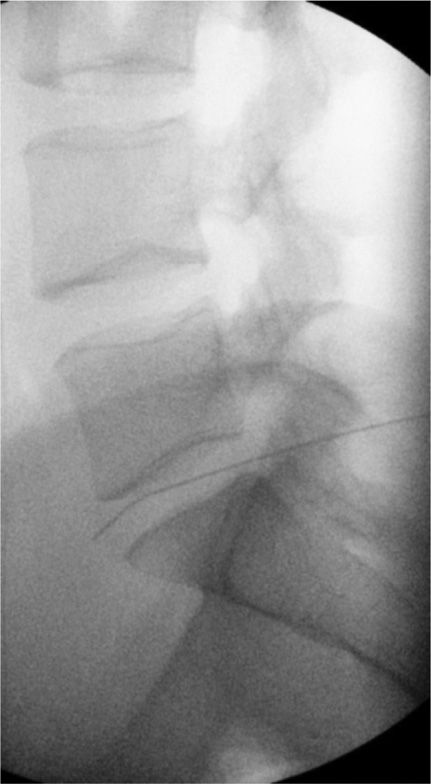
Figure 77-18. Superior hypogastric plexus neurolytic block; right-sided: Needle seen on lateral view. (Used with permission from Kenneth D. Candido, M.D.)
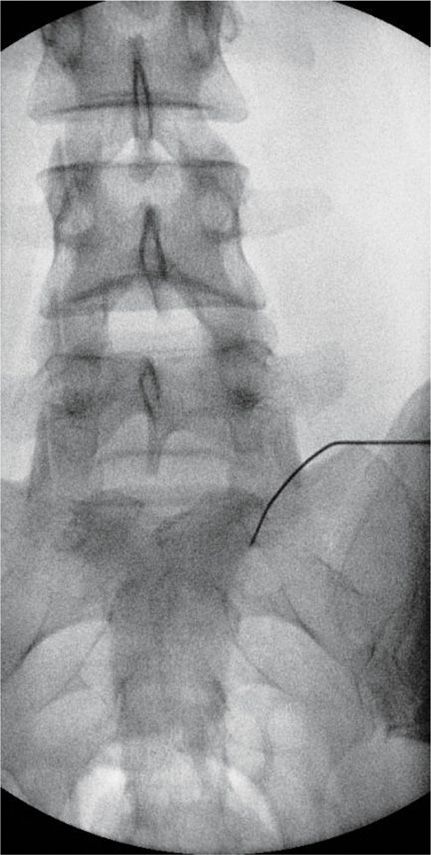
Figure 77-19. Superior hypogastric plexus neurolytic block; right-sided: Needle seen on AP view. (Used with permission from Kenneth D. Candido, M.D.)
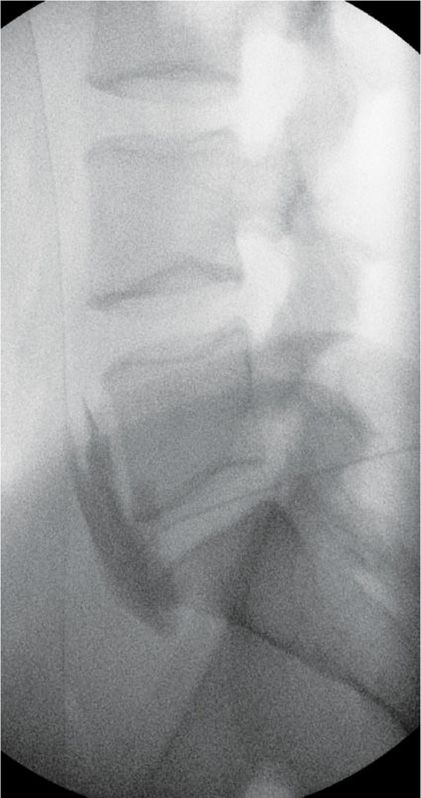
Figure 77-20. Superior hypogastric plexus Neurolytic block; right-sided: Needle seen on lateral view after injection of 5 mL of contrast. (Used with permission from Kenneth D. Candido, M.D.)
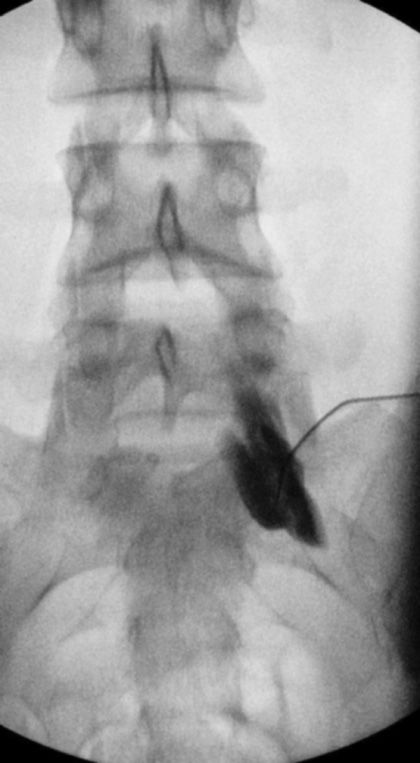
Figure 77-21. Superior hypogastric plexus neurolytic block; right-sided: Needle seen on AP view after injection of 5 mL of contrast. (Used with permission from Kenneth D. Candido, M.D.)
Pain arising from pathologic processes of the large colon and occasionally from other pelvic viscera is modulated by the superior hypogastric plexus. Blockade of the plexus using neurolytic agents is occasionally effective for visceral and sympathetically-mediated pain in the pelvis where local anesthetic diagnostic blocks have proved temporarily effective. The performance of the block is not dissimilar to performing provocation discography at L5-S1 and requires an appreciation of the relevant anatomy needed to perform this as well as perhaps an L5-S1 transforaminal injection. Unlike the situation noted above for ganglion impar block, the superior hypogastric plexus is a bilateral, paired structure, and so if the pain is diffuse and not unilateral, bilateral blocks will need to be performed to successfully modulate this type of sympathetic or visceral pain.
Relevant Anatomy
The superior hypogastric plexus is a bilateral, paired structure and sits in the retroperitoneum at the lower third of the fifth lumbar vertebra, at times extending down as low as the upper one-third of the first sacral level.
Preoperative Considerations
When one or a series of local anesthetic diagnostic blocks prove successful in reducing the pain, albeit temporarily, neurolysis may be considered. An informed consent must include discussion of the risks of rectal perforation; infection; bleeding and hematoma formation; and neural injury.
Contraindications
• Patient who has bleeding problems
• Patient who is taking irreversible inhibitors of platelet aggregation
• Patient who has history of allergy to iodine-based contrast agents
• Patient who has an infection at the injection site
• Patient who is unable to remain motionless during the procedure which may cause unexpected injury during the intervention
Fluoroscopic Views
• It is likely that fluoroscopy use is superior to ultrasound in terms of identifying the boundaries of the vertebral body and the intradiscal space.
• A scout film of the lower lumbar spine and a lateral oblique view is employed to identify the intradiscal space at L5-S1.
Positioning of Patient
• Prone position
Selection of Needles, Medications, and Equipments
• A 5- or 6-in (12.7- or 15.4-cm) 22- or 25-gauge Whitacre- or Quincke-type subarachnoid needle, curved along its mid-shaft, and 3 to 7 mL of 5% to 7% phenol in glycerin per side is used for neurolytic block.
Intraoperative Technical Steps
• The patient is placed prone, as for lumbar provocation discography.
• The usual precautions are taken in terms of ASA monitoring, intravenous access, and sterile skin preparation.
• Ultrasound or fluoroscopy may be used as adjuncts to identifying the relevant anatomy. However, it is likely that fluoroscopy use is superior to ultrasound in terms of identifying the boundaries of the vertebral body and the intradiscal space.
• A scout film of the lower lumbar spine is obtained, and a lateral oblique view is then employed to identify the intradiscal space at L5-S1.
• A 5- or 6-in (12.7- or 15.4-cm) 22- or 25-gauge Whitacre- or Quincke-type subarachnoid needle, curved along its mid-shaft, is advanced with caution taken to avoid striking the iliac crest or placing the needle tip into the intervertebral disc itself.
• Switching to a lateral view is then undertaken to ensure needle tip is immediately ventral to the anterior margin of the L5 vertebral body, at the level of the disc space.
• 3 to 7 mL of water-soluble, iodine-based contrast is next injection.
• If there is no vascular uptake or intradiscal spread, an equivalent volume of 5% to 7% phenol in glycerin per side is incrementally injected with frequent aspiration performed every 1 to 2 mL.
• The needle is flushed with 1 mL of air and is withdrawn.
• Digital subtraction angiography (DSA) may help identify vascular spread and should be considered, if available.
Postprocedure Considerations
• The patient should be observed for 30 to 60 minutes in a monitored setting following the completion of the procedure to be certain that there have been no adverse effects from the neurolytic agent.
Monitoring of Potential Complications
• Possible risks that might occur following the procedure is urinary and bowel problems due to neural damage.9
• In addition, as with most procedures, there is the theoretical risk of pain at the injection site, localized bleeding, infection, never injury, puncture of internal vessels or organs, distal ischemia (if the iliac artery is penetrated and the needle dislodges an atherosclerotic plague).10
SACROILIAC JOINT NEUROLYSIS (FIGURES 77-22 TO 77-24)
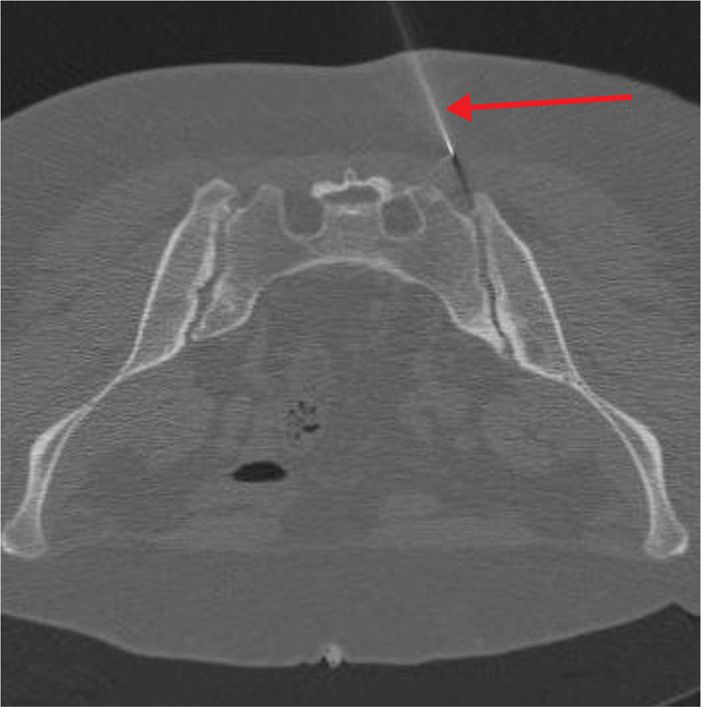
Figure 77-22. Sacroiliac joint neurolysis using phenol: CT-guided; axial image demonstrating advancing needle tip. (Used with permission from Kenneth D. Candido, M.D.)
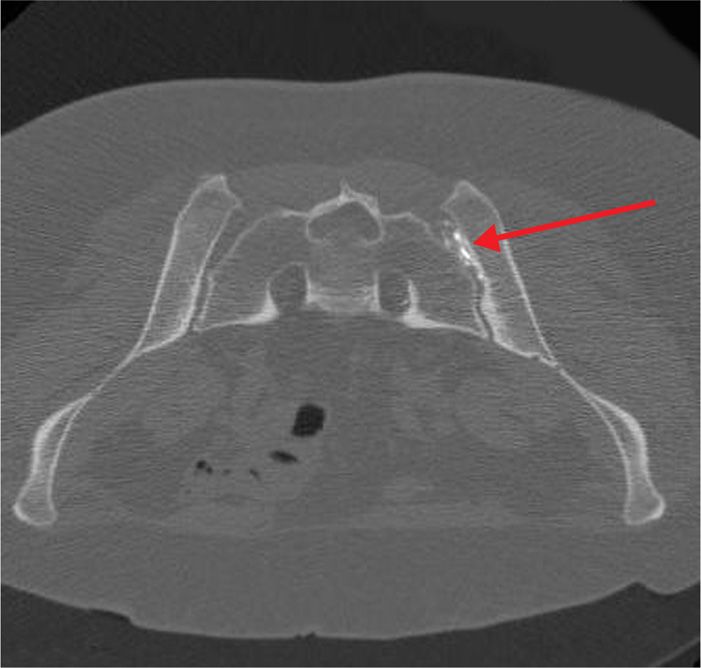
Figure 77-23. Sacroiliac joint neurolysis using phenol: CT-guided; axial image demonstrating advancing needle tip now situated into center of SIJ. (Used with permission from Kenneth D. Candido, M.D.)
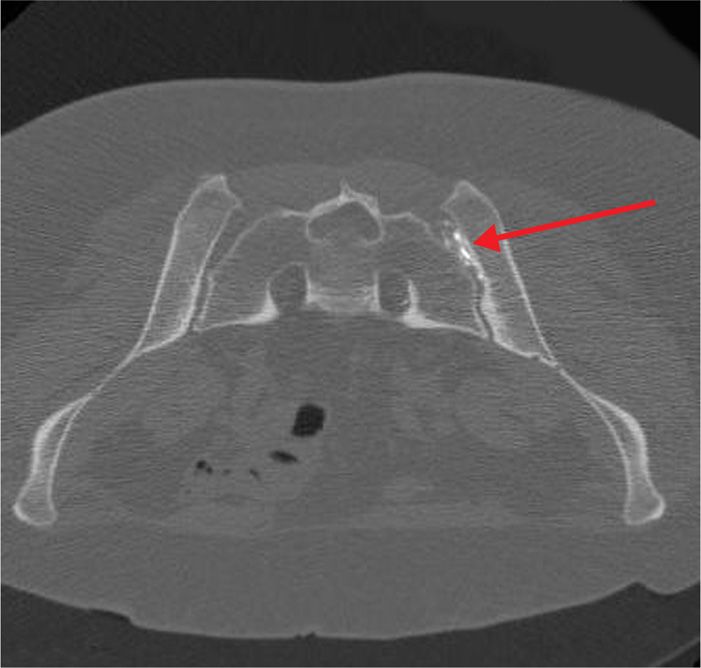
Figure 77-24. Sacroiliac joint neurolysis using phenol: CT-guided; axial image demonstrating contrast injected in anticipation of phenol injection into center of SIJ. (Used with permission from Kenneth D. Candido, M.D.)
There are occasions when neurolysis may be considered for peripheral pain issues, such as pain arising from the SI Joint which has proven to be responsive in the short-term to local anesthetic blocks, but for which analgesia has not been sustained. Although in contemporary practice, interventionists are increasingly turning towards radiofrequency procedures for these scenarios, there remains a small, but finite role for chemical neurolysis of the joint in selected cases.
Relevant Anatomy
As a true synovial joint, the SI joint is a pain-sensitive structure richly invested by unmyelinated free nerve endings in conjunction with the posterior primary rami of L2-S3. Blockade using dilute phenol may represent an option in cases where radiofrequency ablative techniques have either failed, or have provided non-sustained analgesia in an otherwise uncomplicated clinical situation.
Preoperative Considerations
When one or a series of local anesthetic diagnostic blocks prove successful in reducing the pain, albeit temporarily, neurolysis may be considered. An informed consent must include discussion of the risks of rectal perforation, infection, bleeding and hematoma formation, and neural injury.
Contraindications
• Patient who has bleeding problems
• Patient who is taking irreversible inhibitors of platelet aggregation
• Patient who has history of allergy to iodine-based contrast agents
• Patient who has an infection at the injection site
• Patient who is unable to remain motionless during the procedure which may cause unexpected injury during the intervention
Fluoroscopic Views
The use of CT-scan imaging is preferred over fluoroscopy or ultrasound to be able to make an unequivocal determination of proper needle placement.
Positioning of Patient
• Prone position
Selection of Needles, Medications, and Equipments
• A fine-gauge hypodermic needle and 3 to 5 mL of 1% lidocaine with epinephrine, 1:200,000 (5 μg/mL) is used for raising the skin wheal.
• A 22-gauge, 3.5-in (8.89-cm) Quincke-type subarachnoid needle and 2 to 3 mL of 3% to 5% phenol in glycerin is used for neurolysis block.
• Additionally, it is probably prudent to use a lower concentration of phenol (3%-5%) than that used for spinal neurolysis (5%-7%) as the risk of spillover onto the S2-S4 nerve roots (the pudendal nerve) needs to be accounted for and the appropriate precautions taken.
Intraoperative Technical Steps
• The patient is placed prone.
• The usual monitoring, intravenous access, and sterility precautions are undertaken.
• The skin is infiltrated near the anatomical midline using a fine-gauge hypodermic needle and 3 to 5 mL of 1% Lidocaine with epinephrine, 1:200,000 (5 μg/mL).
• A 22-gauge, 3.5-in (8.89-cm) Quincke-type subarachnoid needle is advanced through the skin in a slightly lateral direction until it is submerged 2 to 3 cm.
• Then, the patient is scanned to determine if the angle of entry and advancement is likely to be successful in placing the needle tip into the SI joint.
• Intermittent advancement and scanning is undertaken until the needle reaches the mid-point of the joint.
• Then, 2 to 3 mL of dense, water-soluble, iodine-based contrast is injected.
• If there is no demonstrated spillover of contrast towards the sacral neural foramina, an equivalent volume of 3% to 5% phenol in glycerin is incrementally injected.
• The needle is flushed with air and is withdrawn.
Postprocedure Considerations
• The patient should be observed for 30 to 60 minutes in a monitored setting following the completion of the procedure to be assessed for perineal numbness or urinary incontinence.
Monitoring of Potential Complications
• Even though there was no definitive literature describing serious complications in patients who received Sacroiliac Joint Injection,11 as with most procedures, there is the theoretical risk of pain at the injection site, localized bleeding, infection, never injury, puncture of internal vessels or organs.
SPINAL AND EPIDURAL (INCLUDING TRANSFORAMINAL) NEUROLYSIS (FIGURES 77-25 TO 77-32 AND TABLE 77-1)
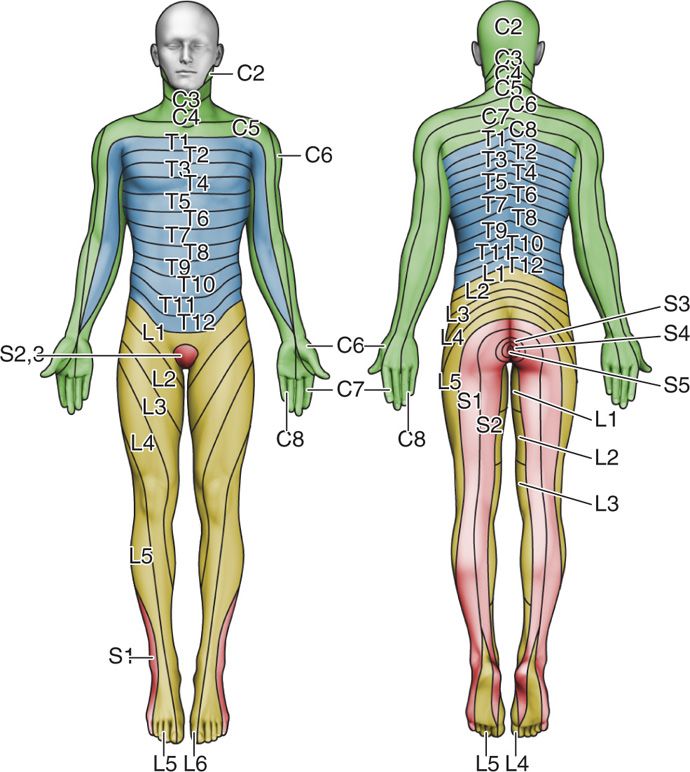
Figure 77-25. Dermatome chart. (Reprinted from K. Candido, R. Stevens Best Practice & Research Clinical Anaesthesiology, 17:3, 407-428, 2003, with permission from Elsevier.)
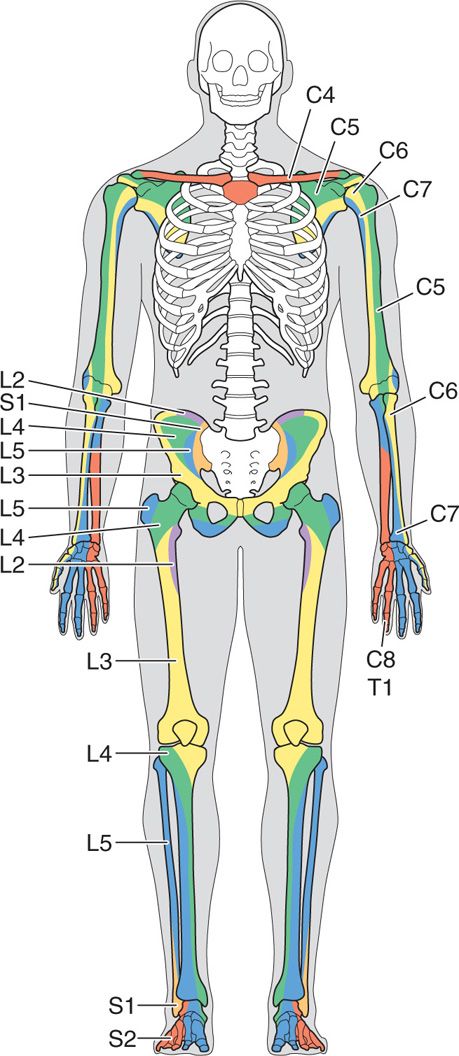
Figure 77-26. Sclerotome chart. (Reprinted from K. Candido, R. Stevens Best Practice & Research Clinical Anaesthesiology, 17:3, 407-428, 2003, with permission from Elsevier.)
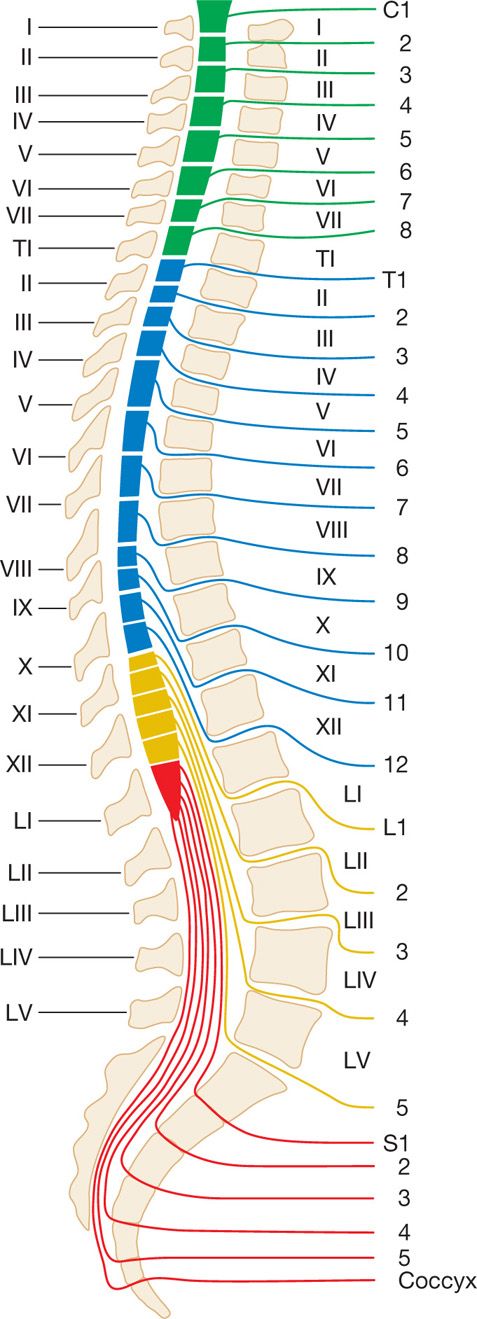
Figure 77-27. Spinal nerve root sites of exit from the spinal cord. (Used with permission from Kenneth D. Candido, MD.)
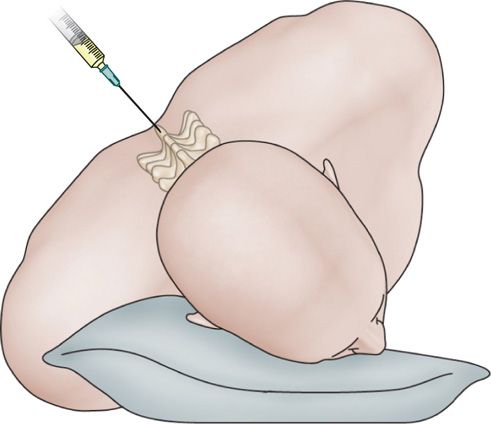
Figure 77-28. Patient positioning for hypobaric alcohol subarachnoid block. (Reprinted from K. Candido, R. Stevens Best Practice & Research Clinical Anaesthesiology, 17:3, 407-428, 2003, with permission from Elsevier.)
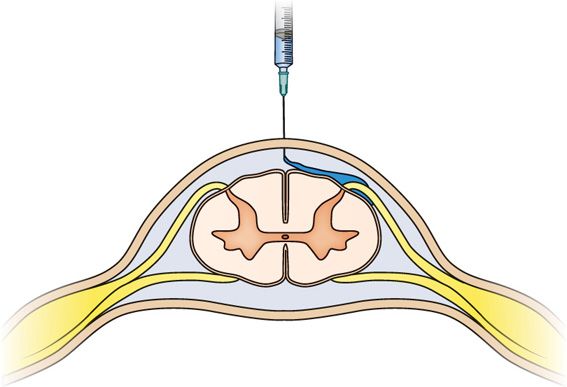
Figure 77-29. Hypobaric alcohol subarachnoid block: Bathing the fila radicularia. (Reprinted from K. Candido, R. Stevens Best Practice & Research Clinical Anaesthesiology, 17:3, 407-428, 2003, with permission from Elsevier.)
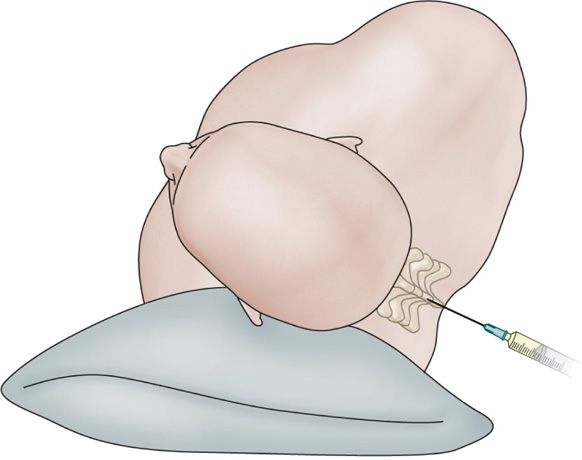
Figure 77-30. Hyperbaric phenol subarachnoid block: patient positioning. (Reprinted from K. Candido, R. Stevens Best Practice & Research Clinical Anaesthesiology, 17:3, 407-428, 2003, with permission from Elsevier.)
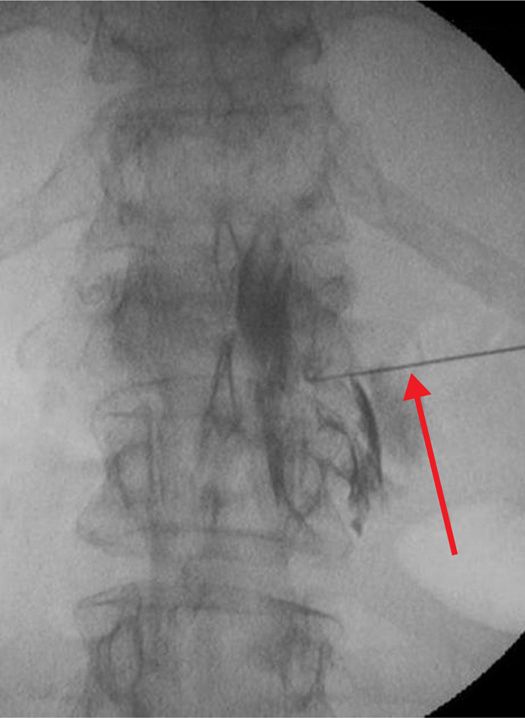
Figure 77-31. Neurolytic transforaminal phenol injection in a patient with prior decompression laminectomies and tumor growth prohibiting assuming lateral decubitus position as if for neurolytic subarachnoid block. Anteroposterior fluoroscopic view with arrow pointing to needle. (Used with permission from Kenneth D. Candido, M.D.)
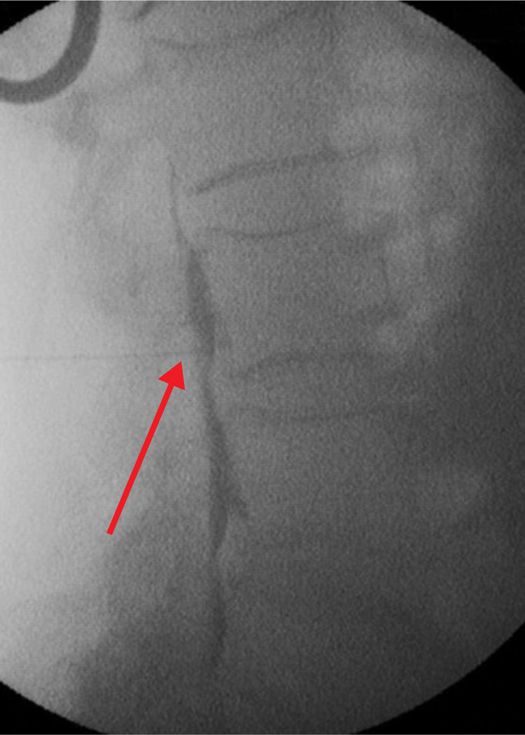
Figure 77-32. Neurolytic transforaminal phenol injection in a patient with prior decompression laminectomies and tumor growth prohibiting assuming lateral decubitus position as if for neurolytic subarachnoid block. Lateral fluoroscopic view. (Used with permission from Kenneth D. Candido, M.D.)
TABLE 77-1. Agents Used for Subarachnoid Neurolysis
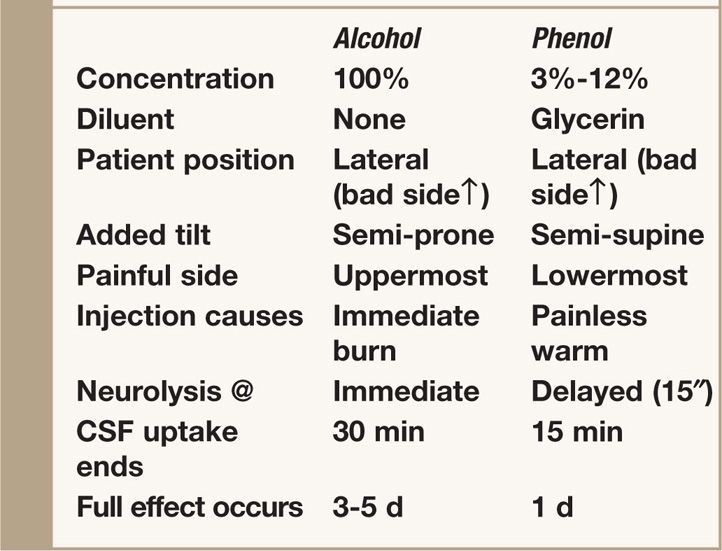
Unfamiliarity with the details of this technique has been a major obstacle to its use, and since proper execution of the technique determines its success and safety, the focus of the present discussion is on technical aspects of subarachnoid neurolysis.
• First, because of the “permanence” of the complications of this technique, subarachnoid neurolysis should be attempted only after careful review of a dermatome chart to determine precisely which nerve or nerves are subserving the patient’s pain.
• If the patient’s pain is due to one or more metastases to bone, it may be useful to refer to a sclerotome chart because the innervation of some parts of the skeleton differs from that of the overlying soft tissues.
• Because a neurolytic subarachnoid block must be carried out at the level where the dorsal root to be blocked leaves the spinal cord (to spare motor function), it is essential to determine which interlaminar foramen affords access to that root.
• Although the cervical nerves exit at a level higher than their respective vertebral bodies, all of the other nerves exit at a level below their respective vertebral bodies.12
• Finally, a choice must be made before the procedure is undertaken to determine whether a hyperbaric (phenol in glycerin) or hypobaric (absolute alcohol) technique is more appropriate.
• No controlled clinical trials have compared the outcomes with subarachnoid alcohol and phenol, but in our experience hypobaric alcohol has been the technique of choice in most cases because most patients with severe, intractable pain cannot lie on the painful side, a requirement when using hyperbaric phenol.
• Although at one time clinicians believed that phenol might exert a preferential effect on the small fibers subserving pain, it has been determined that neither alcohol nor phenol is a selective neurolytic agent, eliminating this as a rationale for choosing phenol over alcohol.13–15
Relevant Anatomy
• Spinal cord is continuous with the brainstem through the foramen magnum and terminates distally in the conus medullaris.
• The distal termination varies from L3 in infants to the lower border of L1 in adults.
• Surrounding of spinal cord in the bony vertebral column are three membranes (from within to the periphery): the pia mater, arachnoid mater, and dura mater.
• The pia mater is a highly vascular membrane that closely invests the spinal cord and brain.
• The arachnoid mater is a delicate and nonvascular membrane. It is closely attached to the dura (the outermost layer).
• Subarachnoid space contains the CSF, spinal nerves, a trabecular network, and blood vessels that supply the spinal cord and lateral extensions of the pia mater and dentate ligaments, which provide lateral support from the spinal cord to the dura mater. Although the spinal cord ends at the lower border of L1 in adults, the subarachnoid space continues to S2.
• The epidural pain is the area surrounding the dura mater anteriorly, laterally, and more usefully, posteriorly. The spinal epidural space extends from the foramen magnum to the sacral hiatus.
Preoperative Considerations
When one or a series of local anesthetic diagnostic blocks prove successful in reducing the pain, albeit temporarily, neurolysis may be considered. An informed consent must include discussion of the risks of rectal perforation; infection; bleeding and hematoma formation; and neural injury.
Contraindications
• Patient who has bleeding problems
• Patient who is taking irreversible inhibitors of platelet aggregation
• Patient who has history of allergy to iodine-based contrast agents
• Patient who has an infection at the injection site
• Patient who is unable to remain motionless during the procedure which may cause unexpected injury during the intervention
Fluoroscopic Views
• While ultrasound may be used, due to the severe potential consequences of a misplaced agent, it is likely that fluoroscopy provides a more detailed assessment of the anatomical confines required for successful blockade.
Positioning of Patient
• Lateral Decubitus (“bad side” up for alcohol injection and “bad side” down for phenol injection)
Selection of Needles, Medications, and Equipment
• A 22-gauge Whitacre- or Quincke-type subarachnoid needle
• 100% alcohol or 3% to 12% phenol
SUBARACHNOID NEUROLYSIS WITH ALCOHOL (INTRAOPERATIVE TECHNICAL STEPS)
• Because absolute alcohol is extremely hypobaric, when this agent is used, the patient is first placed in the lateral decubitus position with the painful site uppermost and is then rolled anteriorly approximately 45 degrees to place the dorsal (sensory) root uppermost.
• After the patient has been positioned properly, a 22-gauge Whitacre- or Quincke-type subarachnoid needle is inserted and advanced through the interlaminar space at the level of the dorsal root to be blocked.
• If the procedure is being carried out at a thoracic level, because of the long, caudally sloping spinous processes, a paravertebral approach is usually easier than a midline approach; whatever the approach, the needle tip should penetrate the dura in the midline.
• Needles smaller than 22-gauge should not be selected for this technique because the free flow of CSF is essential, and because postdural puncture headache is extremely rare after subarachnoid alcohol neurolysis.
• A prognostic block with local anesthetic should already have been carried out to determine whether the pain can be relieved by the technique, and, equally important, whether the patient can tolerate the attendant numbness.
• A test dose of local anesthetic should not be given when the needle is in place for a neurolytic block. The reason is that none of the available local anesthetics can be made as hypobaric as absolute alcohol.
• A local anesthetic should not be administered before the injection of the alcohol because the pain produced by the injection of the alcohol is an essential corroborating indicator that enhances the accuracy and effectiveness of the procedure.
• The physician must tell the patient to expect severe, localized, burning pain, but only for a fraction of a second after each injection, and to focus attention on whether that burning occurs at, above, or below the level of the pain.
• The patient also is instructed to report any other sensations, such as tingling, warmth, or numbness.
• Subarachnoid alcohol neurolysis is a precise procedure, and to ensure efficacy and safety, the alcohol should be injected in 0.1-mL aliquots using a tuberculin syringe.
• The syringe containing the alcohol should not be attached to the needle until the free flow of CSF indicates that the needle is definitely in the subarachnoid space.
• When the syringe has been attached to the needle, aspiration should not be carried out to verify proper needle placement because alcohol causes the CSF to form a white coagulum within the syringe.
• When the syringe has been attached to the needle, the subjective experience of each injection and the importance of the brief burning sensation to the success of the technique are reiterated one more time, after which sequential injection of 0.1-mL increments of alcohol begins.
• The first one or two increments of alcohol usually do not produce the expected burning pain simply because this volume is just enough to fill the hub and shaft of the spinal needle. The third or fourth 0.1-mL increment invariably produces the expected burning, however.
• When a nerve subserving a patient’s pain has been identified and blocked by this process, and a total of 0.7 mL of alcohol has been injected, the process is repeated above or below (or above and below) the level of the initial full injection to abolish the pain completely.
• No more than three or four nerves should ever be blocked at one session, but as indicated in the selection criteria, this procedure is best reserved for patients with pain limited to two or three dermatomes.
• In contrast to subarachnoid injections of local anesthetics for surgical anesthesia, the injection of alcohol for subarachnoid neurolysis must be made through a separate needle to block each nerve root.
• The reason that alcohol cannot be “floated” to a higher or lower level through a single needle, as is done when performing a hypobaric spinal for surgery, is that alcohol “fixes” too quickly and would not float far enough to block the adjacent dermatomes.
• Upon completion of the injection, 0.2 to 0.3 mL of air should be injected to clear the shaft and hub of alcohol to minimize the possibility that alcohol trickling from the needle as it is withdrawn from dura to skin will form a fistula.
SUBARACHNOID NEUROLYSIS WITH PHENOL (INTRAOPERATIVE TECHNICAL STEPS)
Intrathecal phenol in glycerin may be used as an alternative to alcohol for subarachnoid neurolysis. The technique is similar to that described earlier except that the patient must be positioned with the painful side down because phenol in glycerin is a hyperbaric solution.
• The patient must be tilted posteriorly with the back as close to the edge of the bed as possible.
• For this technique, a 22-gauge (or even better, a 20-gauge) spinal needle should be used because of the viscosity of the phenol-glycerin mixture.
• In a manner essentially opposite to the technique of subarachnoid alcohol neurolysis, the bevel of the spinal needle should be directed inferiorly (laterally toward the table).
• Because of the viscosity of phenol in glycerin, it takes significant pressure applied to the plunger of the syringe to force the phenol into the subarachnoid space, so the injection must be done slowly and carefully to prevent the escape of phenol from the syringe and onto the skin of the patient or the practitioner.
• Warming the phenol lessens its viscosity and makes it easier to inject.
• Because phenol has local anesthetic properties, its injection into the subarachnoid space is not accompanied by the burning pain produced by alcohol, although the patient may feel warmth, tingling, or even mild dysesthesia in the distribution of the nerve being injected.
• As with alcohol, after the subarachnoid injection of phenol in glycerin, 0.1 to 0.3 mL of air is injected to flush the lytic solution from each needle before it is removed.
EPIDURAL AND TRANSFORAMINAL NEUROLYSIS (INTRAOPERATIVE TECHNICAL STEPS)
• Occasionally, patients cannot lie on either side, due to abject misery from their underlying disease states.
• In such cases, epidural or transforaminal injections might prove useful.
• Under these circumstances, 5% phenol in glycerin has been demonstrated to be a superior agent compared to alcohol use, and small doses titrated to effect may provide significant analgesia in cases of intractable pain which has been demonstrated to be responsive to local anesthetic diagnostic blocks.16
Postprocedure Considerations
• The patient should be observed for 30 to 60 minutes in a monitored setting following the completion of the procedure to be certain that there have been no adverse effects from the neurolytic agent.
Monitoring of Potential Complications
From the point of view of complications, neurolytic subarachnoid blocks may be safest when undertaken in the midthoracic region because this region is relatively distant from the fibers that subserve limb, bowel, and bladder function, so any motor loss would be of little consequence. Conversely, in the lumbosacral region, owing to the proximity of sensory and motor fibers to each other (because of the decreasing size of the conus medullaris, the dorsal and ventral roots are very close together) and to the proximity of both to the autonomic fibers subserving bowel and bladder function, lumbar subarachnoid neurolysis usually is reserved strictly for select individuals in whom the risk-to-benefit ratio has been clearly delineated. For patients who already have compromised sphincter function, lumbosacral subarachnoid phenol neurolysis has been advocated for rectal and pelvic malignancies because of the tendency of phenol to spare motor function.
Complications of central neuraxial blocks include the following:
• Paralysis/paraplegia
• Bladder dysfunction
• Bowel dysfunction
• Sexual dysfunction
• Anesthesia dolorosa
CLINICAL PEARLS AND PITFALLS
• Neurolysis is a technique best reserved for individuals in severe pain who are not perceived to be candidates for neuromodulation, surgery, or pharmacological pain management without prohibitive side effects.
• Central neuraxial neurolysis is most commonly used, but techniques for blocking somatic, sympathetic, and visceral pain processes have been reported and should be part of the armamentarium of advanced interventional pain physicians.
• Because of the physical separation of the sensory and motor nerve roots in the spinal canal, intrathecal chemical dorsal rhizotomy is the only neurolytic procedure that allows sensory block without concomitant motor block. For this reason, subarachnoid neurolysis is a unique, effective modality for the management of cancer pain in certain patients.
• If the patients are carefully selected, and the technique is carefully carried out, pain relief can be provided in most cases without an excessive rate of complications.

Full access? Get Clinical Tree






