Fig. 55.1
Dilated esophagus with air-fluid level on chest CT
Blood and sputum cultures from ICU day 3 grew Enterobacter aerogenes, sensitive to aztreonam and piperacillin/tazobactam. A gastric emptying study showed significantly slowed motility. The patient did not tolerate enteral feedings even with post-pyloric administration, and peripheral parenteral nutrition was started on ICU day 11. A CT of the abdomen and pelvis was obtained, which showed colonic distention and retained barium throughout the large and small intestine from the esophagram 3 days prior. The patient became progressively more somnolent, and developed profound hyponatremia. Follow up chest radiology on ICU day 14 was significant for development of bilateral infiltrates. An IgE level was obtained, which was 3280 IU/mL (Fig. 55.2).
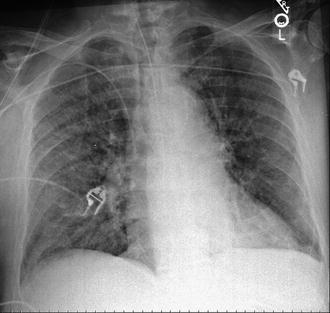

Fig. 55.2
Bilateral infiltrates on chest radiograph
Question
What is the diagnosis?
Answer
Strongyloides hyperinfection with ileus
Strongyloides serologies, stool ova and parasites, and sputum cytology and smear on LPO agar were sent. Sputum cytology showed the following (Fig. 55.3):
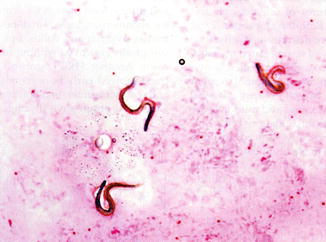

Fig. 55.3
Strongyloides larvae in sputum gram stain
Strongyloides IgG was high at 3.87 IV (>1.0 positive). Larvae were also seen in the fecal exam. The diagnosis of strongyloides hyperinfection was made, and the patient was started on enteral ivermectin therapy. Subsequently, the patient became more obtunded. A decision was made by the family for comfort care, and the patient died shortly thereafter. Autopsy showed diffuse infiltration of larvae in the lungs, wall of the intestines and liver, as well as in the cortex and basal ganglia of the brain (Fig. 55.4). The identification of larvae in the brain on autopsy ultimately defined this case as disseminated strongyloides infection.
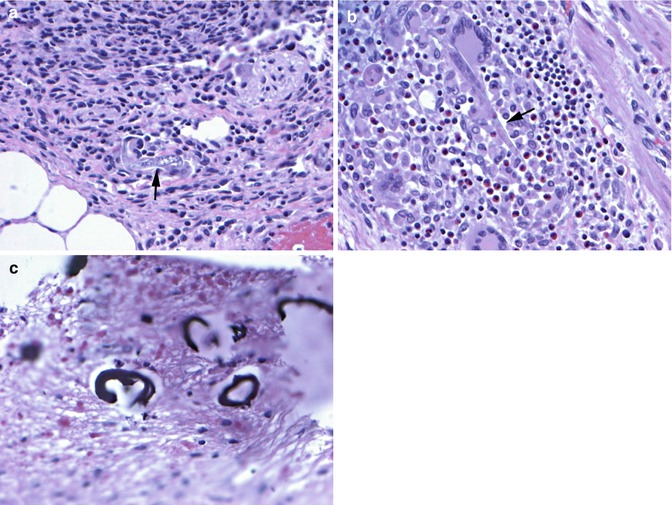

Fig. 55.4
Autopsy findings confirming disseminated strongyloidiasis. (a) Adult worm (arrow) in the lung with foreign body giant cell reaction and marked inflammation. (b) Adult worm form (arrow) with a multinucleated giant cell and background of lymphocytes and eosinophils in wall of the large intestine. (c) Calcified larvae in the basal ganglia
Principles of Management
The most important aspect of management includes high index of suspicion in at risk patient populations and recognition of the clinical signs and symptoms of infection, in order to institute early treatment.
Epidemiology
Strongyloides stercoralis is a parasitic nematode that is endemic in the tropical and subtropical climates of Southeast Asia, sub-Sahara Africa, and parts of Central and South America [1]. In the United States, the highest incidences of infection are in the Southeastern Appalachian states of Kentucky, Tennessee, and Louisiana; farming and mining confer additional risk [2]. With improved sanitation in these areas and increased globalization, new infections are most frequently seen in immigrants or travelers returning from endemic countries. It is especially important to consider those with impaired immune defense – steroid exposure, immunosuppression, solid organ transplant, hematologic malignancies, and infection with HTLV-1 – as possible hosts for disseminated infection and hyperinfection. Screening is now recommended for patients undergoing transplant evaluation [3].
Life Cycle
The nematode is contracted by skin exposure to soils contaminated with larvae. A complex life cycle starts with hematogenous migration of the infective filariform larvae to the alveolar space of the lungs. The larvae are then coughed up and swallowed into the gut, where they mature into adult worms that penetrate and live within the small intestinal mucosa. Here, females produce eggs, which hatch a non-infective, rhabditiform larva that is shed in the stool and can molt into infective filariform larvae or adults in humid soils [4]. The typical life cycle takes 3–4 weeks from time of infection to time of appearance of larvae in the stool.
Alternatively, the non-infective rhabditiform larvae can mature to the infective filariform larvae within the gut in an autoinfective life cycle. These filarial larvae then burrow through the colonic or perianal mucosa, and restart the typical life cycle as above. Autoinfection can occur at low levels with minimal symptoms in some hosts. This autoinfection cycle allows Strongyloides to be uniquely capable of persisting within an infected host for decades without subsequent environmental exposures, especially in cases of asymptomatic chronic infections [4, 5].
Disseminated Infection and Hyperinfection
Decreased cell mediated immunity such as steroid use and HTLV-1 infection increase the propensity for autoinfection [6]. Accelerated autoinfection leads to severe strongyloidiasis, with hyperinfection and disseminated infection. Both entities can carry mortality rates of up to 60–70 % despite appropriate treatment [6]. In hyperinfection syndromes, increased worm burden along the typical life-cycle leads to nausea, vomiting, diarrhea, pulmonary infiltrates and dyspnea, and can eventually lead to ileus, bowel obstruction, and GI bleeding, as well as pulmonary infection and respiratory failure [6]. Pulmonary symptoms are more common in those with prior lung disease, and may even progress to inflammatory pneumonitis, bronchitis, pneumonia, or pulmonary hemorrhage [7]. Disseminated infection is defined as larval presence in organs not part of the typical parasitic life cycle (skin, GI tract, lungs) [6]. Hyperinfection and disseminated infection are often accompanied by bacterial infections involving enteric pathogens. This occurs as the migrating worms transport these bacteria from the gut flora, or through seeding of the blood stream via mucosal ulceration [4].

Full access? Get Clinical Tree







