Fig 60.1
(a, b) Generalized jaundice
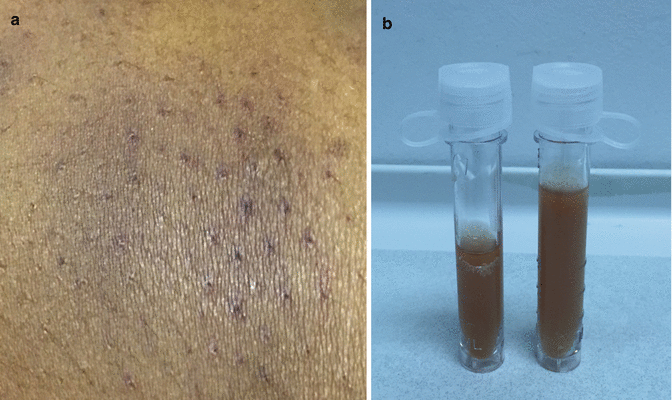
Fig. 60.2
(a, b) Ecchymosis (left) and dark urine demonstrating hemoglobinuria
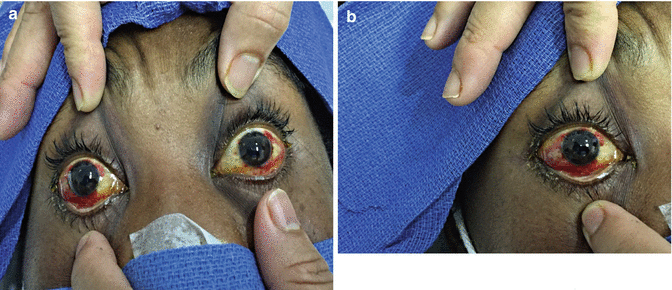
Fig. 60.3
(a, b) Conjunctival suffusion
Question
What is the diagnosis?
Answer
Leptospirosis (Weil’s Disease)
Microscopic agglutination test (MAT) was applied to two serum samples which were collected with a 10 days interval. The first serum sample revealed antibody positivity at 1/200 titer against Leptospira interrogans. After the diagnosis was confirmed he was keep only on Ceftriaxone. By the administration of antibiotic therapy and early supportive care the patient improved clinically and was discharged from the ICU to a step-down unit.
Leptospirosis is a zoonosis that affects the humans and is caused by pathogenic spirochetes of the genus Leptospira. It is a frequent type of zoonosis, especially in tropical regions. Data is insufficient regarding its frequency in non-tropical regions. Half of the cases reported in the United States emanate from Hawaii. Leptospirosis presents with a self-limited to mildly icteric form in nearly 90 % of cases. However in approximately 5–10 % of cases it leads to Weil’s disease, which is characterized by fever, hepatic failure, renal failure and respiratory failure. Weil’s disease is also known as Weil-Vasilyev disease, Swineherd’s disease, rice-field fever, waterborne fever, Nanukayami Fever, cane-cutter fever, swamp fever, mud fever, Stuttgart disease, and Canicola fever.
Principles of Management
Epidemiology
Leptospirosis is usually a result of environmental exposures to infected animal urine or contaminated water or soil. Rarely, it may be contracted after the ingestion of food contaminated with urine or via aerosols [1].
In the United States, the incidence of leptospirosis is relatively low; most cases are reported from the southern and Pacific coastal states. Hawaii consistently reports the most cases of any state, though this may partly be because leptospirosis ceased to be a reportable disease nationally in 1995 but remains reportable in Hawaii. A leptospirosis outbreak was also reported among adventure race participants in Florida [2].
In the tropics, endemic leptospirosis is mainly a disease of poverty (including low education, poor housing, absence of sanitation, and poor income). It is acquired through occupational exposure such as subsistence farming and living in rodent-infested, flood-prone urban slums. Large outbreaks affecting thousands of people and leading to hundreds of deaths are common occurrences. These are often associated with increased rainfall or flooding, such as was the case in epidemics in Guyana and Queensland, Australia [3]. Disease in humans is often sporadic, although outbreaks may occur from common source exposures. See Table 60.1 for risk factors for infection.
Table 60.1
Risk factors for leptospirosis infection
Occupational exposure: | Farmers, ranchers, abattoir workers, trappers, veterinarians, loggers, sewer workers, rice farmers, pet traders, military personnel, laboratory workers |
Recreational activities: | Freshwater swimming, canoeing, kayaking, trail biking |
Household exposure: | Pet dogs, domesticated livestock, rainwater catchment systems, infestation by infected rodents |
Other: | Walking barefoot through surface water, skin lesions, contact with wild rodents, accidental laboratory exposure |
Microbiology
The genus Leptospira contains 20 species; nine are regarded as pathogenic (Leptospira interrogans, L. kirschneri, L. noguchii, L. alexanderi, L. weilii, L. alstonii, L. borgpetersenii, L. santarosai, and L. kmety. Five are of intermediate or unclear pathogenicity and the remaining six are nonpathogenic free-living saprophytic species that do not infect animal hosts.
Leptospira are spiral-shaped, highly motile aerobic spirochetes and are best visualized by dark field microscopy, silver stain, or fluorescent microscopy. They can be distinguished morphologically from other spirochetes by their unique “question mark” hook at the end of the bacterium [6–8].
Pathogenic Leptospira spp can be grown in vitro from clinical specimens including blood, urine, and cerebrospinal fluid (CSF). Special media are required for isolation such as Fletcher’s, Ellinghausen-McCullough-Johnson-Harris (EMJH) or polysorbate 80 media. Therefore, the laboratory needs to be notified if an attempt to isolate leptospires is desired. Growth is usually observed in 1–2 weeks but may take up to 3 months. A method of growing leptospires on solid agar has been developed to facilitate more rapid growth, isolation of single colonies, and simple antimicrobial sensitivity testing [6–8].
Clinical Presentation
Most cases of Leptospirosis are mild and self-limited or subclinical, while some are severe and potentially fatal. The majority of patients describe an abrupt onset of fever, rigors, myalgias, and headache in 75–100 % of patients, following an incubation period of 2–26 days (average 10 days). Nonproductive cough occurs in 25–35 % of cases; nausea, vomiting, and diarrhea occur in approximately 50 % of cases [8, 9].
Less common symptoms include arthralgias, bone pain, sore throat, and abdominal pain. Acalculous cholecystitis and pancreatitis have been described in children. Leptospirosis has been described as a biphasic illness (with an acute bacteremic phase followed by an “immune” phase) but, clinically, the two phases usually merge, particularly in severe disease [8–10].
Conjunctival suffusion is an important but frequently overlooked sign; in one case series, it occurred in 55 % of patients. This is not a common finding in other infectious diseases, and its presence in a patient with a nonspecific febrile illness should raise the possibility of leptospirosis. Muscle tenderness, splenomegaly, lymphadenopathy, pharyngitis, hepatomegaly, muscle rigidity, abnormal respiratory auscultation, or skin rash occur in 7–40 % of patients [9, 10]. Aseptic meningitis is observed in 50–85 % of patients if cerebrospinal fluid (CSF) is examined after 7 days of illness [1].
Severe, potentially fatal illness, “Weil’s disease,” is characterized by jaundice and renal failure and occurs only in a minority of patients. Pulmonary hemorrhage is a potential complication. Mortality rates in hospitalized patients with leptospirosis range from 4 to 52 % [8–10].
Leptospirosis may be complicated by renal failure, uveitis, pulmonary hemorrhage, acute respiratory distress syndrome (ARDS), myocarditis, and rhabdomyolysis. Renal failure is often nonoliguric and associated with hypokalemia. Supportive renal replacement therapy may be required during the acute phase; renal recovery is generally complete. Liver failure generally is reversible and not a cause of death. Vasculitis with necrosis of extremities may be seen in severe cases [7–9].

Full access? Get Clinical Tree







