Alan Ona Malabanan A stable extracellular calcium concentration and proper compartmentalization of calcium are vitally important to a number of physiologic and cellular functions. A high intracellular calcium level can lead to organellar damage, aggregation of amino and nucleic acids, alteration of the integrity of lipid membranes, and phosphate (i.e., adenosine triphosphate) precipitation.1 Consequently, aberrations in calcium homeostasis may lead to neuromuscular, cardiac, nephrologic, endocrine, coagulatory, and gastrointestinal dysfunction as well as to cellular and organism death. Hypercalcemia, a high level of serum ionized calcium (i.e., ionized calcium >5.3 mg/dL), is a disorder in which the calcium level exceeds the upper limit of the normal range (i.e., total corrected calcium >10.5 mg/dL). Conversely, hypocalcemia is a low level of serum ionized calcium (i.e., ionized calcium <4.4 mg/dL), with a total corrected calcium level below 8.5 mg/dL. Both disorders can be a manifestation of a serious illness, such as malignant disease, or can be detected incidentally by laboratory testing in an asymptomatic patient. The calcium imbalance may have varied causes, may be chronic or acute, and may exhibit variable effects. Hypercalcemia in the outpatient setting, most commonly primary hyperparathyroidism, is common; it has been estimated to be as high as 0.2% in patients older than 60 years,2 although reports have suggested an initial declining incidence of 21.6 per 100,000 person-years3 and a subsequent peak to 101.2 per 100,000 person-years and decline between 1997 and 2010.4 Among inpatients, hypercalcemia is common in those with cancer (occurring in as many as 20%)2 but also in critically ill patients (15%)5 Hypocalcemia is common in hospitalized patients (18%) and particularly those in the intensive care unit (ICU; 85%).6 Ninety-nine percent of the body’s roughly 1 kg of calcium is found in the bone as hydroxyapatite. The remaining 1% is found in the extracellular fluid and soft tissues. Approximately 50% of plasma calcium is in the ionized, biologically active form; 10% is complexed in nonionic form; and 40% is protein bound, predominantly to albumin. Roughly 500 mg of the bone calcium is released from the bone each day in normal bone turnover.7 Alterations in albumin levels may cause changes in total calcium concentrations, without altering ionized calcium levels. The serum calcium concentration can be corrected for alterations in albumin by the following formula: Alterations in acid-base balance may also alter ionized calcium levels. Increased acidemia decreases calcium binding to albumin and increases ionized calcium. Conversely, increased alkalemia increases calcium binding to albumin and decreases ionized calcium. Extracellular calcium homeostasis is regulated primarily by the interaction between the calcium-sensing receptor (CaSR), parathyroid hormone (PTH), and 1,25-dihydroxyvitamin D3 (calcitriol). PTH and calcitriol exert their effects through feedback mechanisms on three major organ systems: the skeleton, the intestinal tract, and the kidneys. A decrease in serum ionized calcium is detected by the CaSR and stimulates the production of PTH. PTH has a direct effect on calcium and phosphorus through increased osteoclast activity, leading to bone resorption. PTH also directly stimulates the kidney tubules to reabsorb calcium and to excrete phosphorus, leading to a rise in serum calcium and a fall in serum phosphorus. PTH also increases renal conversion of 25-hydroxyvitamin D, the major circulating vitamin D metabolite, into calcitriol, the active vitamin D metabolite, which increases intestinal absorption of calcium and phosphate. Increases in serum calcium and 1,25-dihydroxyvitamin D levels inhibit release of PTH, reversing the process. Hypercalcemia can be categorized as either PTH dependent or PTH independent, depending on whether PTH is nonsuppressed or suppressed, respectively. PTH-dependent hypercalcemia is typically a result of primary hyperparathyroidism, and PTH-independent hypercalcemia is typically a result of hypercalcemia of malignancy. Primary hyperparathyroidism results when an autonomous parathyroid cell line develops. There are three malignancy-associated hypercalcemia PTH-independent mechanisms: humoral hypercalcemia of malignancy, local osteolytic hypercalcemia, and calcitriol-induced hypercalcemia. Humoral hypercalcemia of malignancy is the most common (approximately 80%) and is caused by excessive secretion of parathyroid hormone–related protein (PTHrP), which activates the same PTH-1 receptor activated by PTH. This has been associated with squamous carcinomas and breast and renal carcinomas. Local osteolytic hypercalcemia accounts for 20% of malignancy-associated hypercalcemia and has been associated with breast and hematologic cancers with widespread skeletal involvement. Calcitriol-induced hypercalcemia has been associated with lymphomas that increase production of calcitriol. Malignancy-associated hypercalcemia has also been associated with authentic ectopic hyperparathyroidism (i.e., non–PTHrP-related hypercalcemia).8 Milk-alkali syndrome, a combination of hypercalcemia, metabolic alkalosis, and renal insufficiency, develops in response to the simultaneous ingestion of large amounts of calcium and absorbable alkali, such as calcium carbonate. This is the third most common cause of hypercalcemia.9 Disorders involving vitamin D or vitamin A excess or increased vitamin D activation lead to increased bone resorption and intestinal absorption of calcium. Hypercalcemia may also result from common mechanisms but rare causes, such as mammary hyperplasia and pregnancy (increased PTHrP), granulomatous disease from silicone injections (increased calcitriol), and betel nut consumption with oyster shell (milk-alkali syndrome).10 The presence and severity of symptoms in hypercalcemia are influenced by the magnitude of hypercalcemia, rate of rise of calcium, acid-base balance, and presence of hypoalbuminemia. A very high level of calcium may be tolerated chronically, whereas a less elevated but abrupt increase may cause significant symptoms. In general, however, corrected serum calcium levels below 11.5 mg/dL are rarely symptomatic. The symptoms of hypercalcemia of any cause typically involve neuromuscular, cardiac, and gastrointestinal depression. Neurologic changes can range from the subtle, such as an inability to concentrate, increased fatigue, depression, or increased sleep requirement, to the dramatic, with confusion, delirium, stupor, or coma. Cardiovascular manifestations can include bradycardia, hypertension, electrocardiographic abnormalities such as arrhythmias (especially on digitalis), bundle branch or atrioventricular blocks, and shortened QT interval as well as cardiac arrest with severe hypercalcemia. Gastrointestinal symptoms are common and include constipation, anorexia, nausea, and vomiting. Peptic ulcers and pancreatitis are less common and may be more common with type I multiple endocrine neoplasia and primary hyperparathyroidism.11,12 Renal symptoms can range from polyuria and nocturia (from nephrogenic diabetes insipidus), which can lead to volume depletion and worsened hypercalcemia, to nephrolithiasis, nephrocalcinosis, and renal failure. The physical examination findings are often unremarkable. Cardiovascular examination may reveal irregularity of rate and rhythm. Band keratopathy, calcium deposition in the cornea, may be present regardless of the cause of hypercalcemia. The presence of a neck mass (parathyroid carcinoma or medullary thyroid carcinoma in type IIA multiple endocrine neoplasia) or breast mass (breast carcinoma) may suggest the cause of the hypercalcemia. Central nervous system depression is reflected in hyporeflexia, changes in sensorium, muscle weakness, tremor, lethargy, and ataxia. With severe hypercalcemia, stupor and coma may result. There may be flank tenderness if nephrolithiasis is a complication. On occasion, pseudogout (calcium pyrophosphate dehydrate crystal deposition disease) may cause joint swelling or inflammation. The diagnosis of hypercalcemia is usually made by measuring the serum total calcium level along with a serum albumin level, with calculation of the corrected calcium concentration. In patients who are critically ill13 or have chronic kidney disease,14 the ionized calcium level may be useful. Once hypercalcemia has been confirmed, determination of the cause is the next step. Whereas the serum phosphate level (low in primary hyperparathyroidism) and the serum magnesium level (high in familial hypocalciuric hypercalcemia) may be useful, the intact PTH assay, ideally performed fasting, is the best test to determine whether the hypercalcemia is PTH dependent (PTH nonsuppressed) or PTH independent (PTH suppressed). If the hypercalcemia is PTH independent, further testing with serum calcitriol level, serum protein electrophoresis, and PTHrP level would be useful. A high or inappropriately normal serum calcitriol level should lead to an evaluation for tuberculosis, sarcoidosis, or lymphoma as a potential cause. A careful history of nonprescription supplements, such as calcium-containing antacids and cod liver oil, may be helpful in identifying milk-alkali syndrome or vitamin A or D toxicity as a cause of the hypercalcemia.
Hypercalcemia and Hypocalcemia
Definition and Epidemiology
![]() Physician consultation is indicated for patients with serum corrected calcium levels of less than 8.5 mg/dL or more than 10.5 mg/dL.
Physician consultation is indicated for patients with serum corrected calcium levels of less than 8.5 mg/dL or more than 10.5 mg/dL.
Hypercalcemia
Pathophysiology
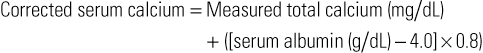
Clinical Presentation
Physical Examination
Diagnostics





