Chapter 1 History of Pediatric Critical Care
In his book Retrospectroscope: Insights into Medical Discovery,1 Dr. Julius H. Comroe Jr, wrote about the courage to fail. He concluded there is no single definition of courage but that it comes in different sizes, each with its own definition. There are four sizes.
Examples of Courage, Size 1 awards go to James Carroll, William Dean, and Jesse Lazear (a former house officer of W. Osler),2 who all volunteered to be bitten by yellow fever-infected mosquitoes to prove the mosquitoes were the human-to-human vector of the disease. They proved it and Lazear died. Also Werner Forssmann, who in 1929 introduced a catheter into his own right atrium in order to improve diagnosis for treatment of certain disorders, not knowing whether the tip would cause ventricular fibrillation.2,3 He received little acclaim for this breakthrough until 1956, when he won the Nobel Prize.
Courage, Size 4: “Size 4 medals go to patients who, informed by specialists they have advanced disease and statistically have only weeks, months, or years to live, elect to undergo a previously untested operation or other form of therapy…It might benefit them or lead to earlier death.” Examples are the first man (1925) to have bilateral sympathectomy for very high arterial blood pressure; Dr. James Gilmore (1933), the first patient to have one whole lung removed surgically at a single operation; the family of the first patient to receive insulin; the first patient to receive penicillin; and the family of the first “blue baby” operated on by Blalock.
Definitions
Definition of a Pediatric Intensive Care Unit
In the 1983, Guidelines for Pediatric Intensive Care Units (PICUs)4 (updated 19935 and 20046) the committee defined a PICU as “…a hospital unit which provides treatment to children with a wide variety of illnesses of life-threatening nature including children with highly unstable conditions and those requiring sophisticated medical and surgical treatment.” Randolph et al.7 have expanded this definition, stating, “A PICU is a separate physical facility or unit specifically designated for the treatment of pediatric patients who, because of shock, trauma, or other life-threatening conditions, require intensive, comprehensive observations and care.”
Definition of Pediatric Intensivist
Randolph et al.7 define a pediatric intensivist (in the United States) as “…any one of the following: (a) a pediatrician with subspecialty training in PCCM and subspecialty certification from the American Board of Pediatrics (ABP); (b) a pediatric anesthesiologist with special competency in critical care with subspecialty certification from the American Board of Anesthesiology; (c) a pediatric surgeon with special competency in critical care with subspecialty certification from the American Board of Surgery; or (d) a physician (as above) eligible for subspecialty certification by their respective board.” Similar requirements for training exist or are in development elsewhere in the world.
History of Critical Care
Resuscitation and Ventilation
The key to understanding the present practice of intensive care for children lies in knowing the history of scientific study of cardiorespiratory anatomy and physiology and of the discovery of techniques to support ill patients. Although one could think our current practice suddenly emerged with the late twentieth-century technical discoveries, Downes and Todres have skillfully reminded us3,8 that accomplishments in the development of resuscitation and ventilation that we take for granted today date back to the Bible, and numerous events and contributions led to our current practice. In a biblical story,9 Elisha resurrected a young boy who was dead when, “ …he climbed onto the bed and stretched himself on top of the child, putting his mouth to his mouth, his eyes to his eyes, and his hands to his hands, and as he lowered himself onto him the child’s flesh grew warm….Then the child sneezed and opened his eyes.” In 117 CE, Antyllus performed tracheotomies for patients with upper airway obstruction.10 Paracelsus, a sixteenth-century Swiss alchemist and physician, first provided artificial ventilation to both animals and dead humans using a bellows,10 and Andreas Vesalius, a Flemish professor of anatomy, in De Humani Corporis Fabrica reported ventilating open-chest dogs and pigs using a fireplace bellows in 1543.11–13
The French obstetrician Desault, in 1801, described how to successfully resuscitate apneic or limp newborns by digital orotracheal intubation with a lacquered fabric tube and then blowing into the tube.3 In 1832, Dr. John Dalziel in Scotland developed a bellows-operated intermittent negative-pressure device to assist ventilation,14 In 1864, Alfred F. Jones, of Lexington, Kentucky, built a body-enclosing tank ventilator, and in the 1880s, Alexander Graham Bell developed a “vacuum jacket” driven by hand-operated bellows.14 In 1876, Woillez, in Paris, built what was probably the first workable iron lung, which was strikingly similar to the respirator introduced by Emerson in 1931.14 Braun developed an infant resuscitator, as described by Doe in 1889, which was used successfully in 50 consecutive patients. A respirator developed by Steuart in Cape Town, South Africa, in 1918 apparently successfully treated a series of polio patients, but he did not report it.14
In 1888, Joseph O’Dwyer, a physician working at the New York Foundling Hospital who was concerned about the severe death rate in croup and laryngeal diphtheria, instituted the manual method of blind laryngeal intubation. Despite severe criticism from associates and the other practitioners, he persisted in the use of this technique. He assembled a series of sized tubes for the palliation of adult and pediatric laryngeal stenosis and, with George Fell, devised a method of ventilation with a foot-operated bellows connected by rubber tubing to the endotracheal tube (Figure 1-1).12 O’Dwyer may deserve a Courage, Size 4 award for his work.

Figure 1–1 The Fell-O’Dwyer Respiratory Apparatus.
(Reproduced with permission, Blackwell Scientific Publications, Oxford.)
In 1898, Rudolph Matas of New Orleans adapted the Fell-O’Dwyer technique to perform chest wall surgery and, in the early 1900s, George Morris Dorrance of Philadelphia used the technique to perform resuscitations.12 In 1910, at the Trendelenburg Clinic in Leipzig, two thoracic surgeons, A. Lawen and R. Sievers, developed a preset, electrically powered piston-cylinder ventilator with a draw-over humidifier. It was used with a tracheotomy tube during and after surgery and for a variety of diseases.3 Over a long career, Chevalier Jackson (1858-1955), a surgeon at Temple University in Philadelphia, developed the techniques for laryngoscopy, bronchoscopy, and tracheotomy.3
In 1958, Peter Safar published work in which he showed the longstanding resuscitation technique of chest-pressure arm-lift was virtually worthless and, in effect, went back to Elisha and proved jaw thrust and mouth-to-mouth resuscitation superior.15 Soon after, W.B. Kouwenhoven and James Jude at Johns Hopkins published work on the effectiveness of closed-chest cardiac massage.16 Beck and his team, in 1946, had demonstrated open-chest electrical defibrillation, and, in 1952, Zoll and his team proved the efficacy of external defibrillation and, in 1956, the effectiveness of external cardiac pacing.17
Anesthesia
In 1842, Crawford W. Long, a University of Pennsylvania Medical School graduate practicing medicine in rural Georgia, observed that bruises encountered by participants during “ether frolics” caused no pain when they occurred during the “exhilatory” effects induced by inhalation of vapor. This also occurred when nitrous oxide was inhaled. Both of these agents, at the time, were inhaled for their hallucinatory effects in the United States. Long utilized this serendipitous observation to provide ether to James Venable and incise a cyst from his neck, without pain. This was 4 years before Morton’s demonstration of the use of ether at Massachusetts General Hospital in 1846. In 1849, Long reported his experience with his third patient, an 8-year-old boy who had a diseased toe, which was amputated without pain in 1842.18
The widely publicized public demonstration of the use of ether by the dentist William T.G. Morton took place at the Massachusetts General Hospital on October 16, 1846. Dr. John Collins Warren removed a mandibular tumor, without the patient experiencing pain. This great success was quickly picked up and used by John Snow in London and later by Friedrich Trendelenburg in Leipzig, who first used anesthesia via an endotracheal tube in 1869.19
Anatomy and Physiology
What seems simple and obvious today took a great deal of time, effort, and insight to understand. Downes3 has provided a thorough review of this topic, and we briefly note here some of the contributions that advanced medicine and enabled the development of cardiorespiratory support and, eventually, intensive care. Andreas Vesalius (1514-1564), the Flemish anatomist, corrected many previous mistakes in anatomy and provided positive-pressure ventilation via a tracheotomy tube to asphyxiated fetal lambs. Michael Servetus of Spain (1511-1553) correctly described the pumping action of the heart’s ventricles and the circulation of the blood from the right heart through the lungs to the left heart. He was burned at the stake for his views and thus deserves a Courage, Size 1 award. Matteo Realdo Columbo (1515?-1559) described the pulmonary circulation and the concept that the lungs added a spirituous element to the blood by the admixture of air. William Harvey (1578-1657), with his genius and perseverance, published De Motu Cordis (On the Motion of the Heart)20 in 1678. Since he did not yet have the microscope available, he could not see the capillaries and thus could not include the mechanism for transfer of blood from the arterial to the venous system of the pulmonary circulation. Capillaries were described by Marcello Malpighi (1628-1694, Italian) in De Pulmonibus (On the Lungs) in 1661. Thomas Willis (1611-1675) and, eventually, William Cullan (1710-1790) led the way to understanding the role of the nervous system as the site for consciousness and the regulation of vital phenomena. Richard Cower (1631-1641) proved it was the passage of blood through the lungs, ventilation of the lungs, and gas exchange with blood that vivified the blood and turned it red. Stephen Hales (1677-1761) measured blood pressure with a brass tube connected to a 9-foot glass tube in a horse. Joseph Black (1728-1799) identified carbon dioxide as a gas expired from human lungs. Karl Wilhelm Scheele (1742-1785) isolated oxygen, as did Joseph Priestley (1733-1804), who named it dephlogisticated air and determined its vital role in supporting combustion. Antoine Laurent Lavoisier (1743-1794) identified oxygen as the vital element taken up by the lungs that maintains life and gave it its name, but its essential role in physiology and biochemistry was clarified much later. Joseph Lister (1817-1916), one of the founders of modern histology, reasoned that bacteria were the source of pus in rotten organic material and used carbolic acid in surgical fields to eliminate bacteria. This technique improved patient outcomes for wounds and after surgery. Along with the discovery of antibiotics, antiseptic technique was an important step in patient care. Nonetheless, imperfect antiseptic technique, sepsis, inflammation, and the consequences of multiorgan failure are still a major portion of what pediatric intensivists deal with today. Felix Hoppe-Seyler (1825-1895) described the transportation of oxygen in blood by hemoglobin. Robert Koch (1843-1910) developed his postulates in 1882. William Konrad von Röntgen (1845-1923) discovered x-rays. Scipione Riva-Rocci (1863-1937), in 1846, measured blood pressure using the sphygmomanometer, and Nikolai Korotkoff, in 1905, introduced his auscultation method.3 In the present day, cardiac catheterization, echocardiography, computerized tomography, and magnetic resonance imaging have enabled clinicians to delve into anatomy and physiology in the living patient with relative ease.
History of Pediatric Critical Care
Pediatric Anesthesiology
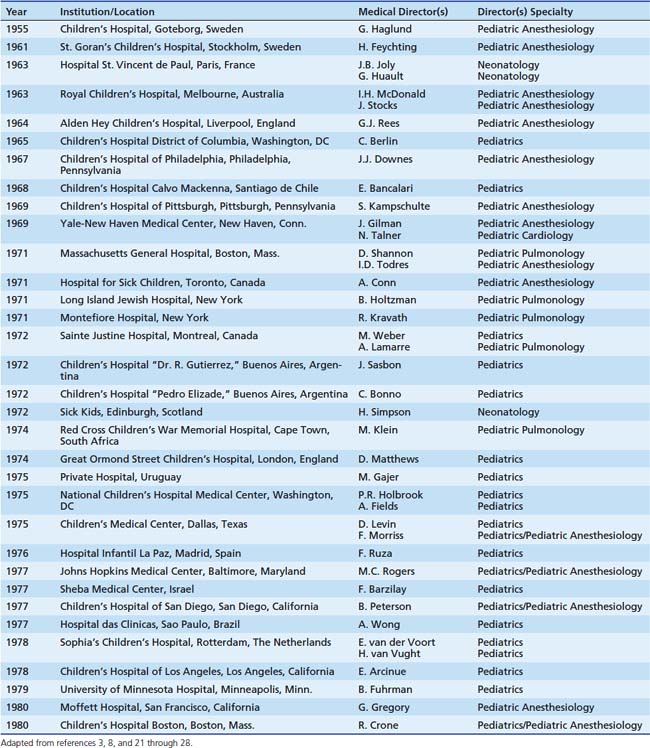
The development of PCCM rests on the efforts of pediatric anesthesiologists, as well as pediatric general and cardiac surgeons, and neonatologists. In fact, most of the original PICUs were founded by pediatric anesthesiologists (Table 1-1).3,8,21–28 Much depends on the definition of a PICU, very much a moving target in the early days, with units eventually evolving from separate areas within recovery rooms and adult units to separate freestanding PICUs. In addition to those noted in Table 1-1, there were probably others which are not as well documented.
Pediatric General and Cardiac Surgery
The pioneering efforts of Dr. William E. Ladd (1880-1967) at Boston Children’s Hospital (BCH) in developing many of the techniques to operate on noncardiac congenital malformations and Dr. Robert Gross, also at BCH, to operate on congenital cardiac lesions (7-year-old Lorraine, coarctation of the aorta, August 23, 1938) were instrumental in developing their surgical fields and demonstrating the need for good postoperative care. Dr. C. Everett Koop trained there for 6 months and then returned to Children’s Hospital of Philadelphia (CHOP) where he, with the help of nursing staff, developed the first neonatal surgical intensive care unit in 1956. This was staffed by Dr. Leonard Bachman (anesthesiology) and his colleagues. Dr. Bachman’s young associate, John J. Downes, subsequently set up the PICU in the hospital in 1967. Dr. C. Crawfoord in Sweden repaired a coarctation of the aorta in 1945, and Drs. Alfred Blalock (surgeon) and Helen Taussig (cardiologist) with Mr. Vivien Thomas (laboratory assistant) at Johns Hopkins created the subclavian-to-pulmonary artery shunt for tetralogy of Fallot, also in 1945. Dr. John Gibbon at Jefferson Medical College Hospital in Philadelphia performed the first successful open-heart surgery (for atrial septal defect) using cardiopulmonary bypass in 1953.3 As the surgical procedures became more invasive, the need for improved postoperative support of all organ systems advanced rapidly. Although some surgeons believed the success or failure of treatment was solely determined in the operating room, others credited improved survival to better postoperative care.
Neonatology
Pediatric critical care owes a great debt to fellow neonatal pediatricians.3,8,29 In the 1880s and 1890s special care nurseries were developed in Paris, and in 1914, the first premature infant center in the United States was opened at Michael Reese Hospital in Chicago by Dr. Julius Hess (1876-1955). In Canada, Dr. Alfred Hart performed exchange transfusions in 1928, and in 1932, Drs. Louis Diamond, Kenneth Blackfan, and James Batey at BCH described the pathophysiology of hemolytic anemia and jaundice of erythroblastosis fetalis; in 1948, the same team performed exchange transfusions using a feeding tube inserted in the umbilical vein. In the 1950s and 1960s, Dr. Geoffrey Dawes at the Nuffield Institute for Medical Research at Oxford University began work, using fetal and newborn lambs, to describe the circulation of mammalian neonates. This work was continued, and the fetal transitional circulation further elucidated, by Dr. Abraham Rudolph and colleagues at the Cardiovascular Research Institute (CVRI) of the University of California, San Francisco (UCSF).
In 1959, Drs. Peter Smythe (pediatrician) and Arthur Bull (anesthesiologist) had the first real success in long-term mechanical ventilation of neonates, treating infants with neonatal tetanus for 4 to 14 days using tracheotomy and a modified Radcliff adult ventilator.30 Up until that time, infants were not given ventilatory support for more than a few hours using manual ventilation. There were no pediatric ventilators, humidifiers, or blood gas analysis. Dr. Smythe had to overcome these obstacles by innovation. On July 13, 1957, he began intermittent positive-pressure ventilation on a baby with neonatal tetanus at Groote Schuur Hospital, with the assistance of anesthesiologist Bull. This was truly a landmark event in the evolution of PCCM. There are three interesting points to be made about their work. First, although considered a success story in that it was the first time infants survived long-term positive-pressure mechanical ventilation, the first 7 of 9 patients died. Eventually their survival rate reached 80% to 90%. Surely Smythe and Bull deserve Perseverance, Size 1 awards. Second, they commented that, “No praise can be too high for the nursing staff, who were all student nurses and without any special training.” And third, Dr. David Todres, a medical student at the time, administered intramuscular curare to these patients. Dr. Smythe moved to Red Cross Children’s Hospital when it opened in 1958, and established a 6-bed neonatal tetanus unit.
In 1963–1964 in Toronto, Drs. Paul Swyer, Maria Delivoria-Papadopoulos and Henry Levison were the first to successfully treat premature infants with RDS with positive-pressure mechanical ventilation and supportive care.31 They emphasized the importance of a full-time team, including dedicated nurses and therapists as well as physicians. In 1968, Dr. George Gregory and colleagues demonstrated greatly improved survival with the addition of continuous positive airway pressure (CPAP) and positive end-expiratory pressure (PEEP) to the mechanical ventilation regimen.32 However, as always, progress in treating a disorder leads to unforeseen complications and new disorders, and successful treatment of RDS led to survivors with chronic lung disease, retinopathy of prematurity, and hypoxic brain injury. When Morriss and Levin were first working in Dallas they complained that the beds were taken up by chronic patients. One of the pediatricians commented, “Before you started doing this, we didn’t have chronic patients.” This was an early observation still relevant today: Pediatric intensive care allows successful treatment of disorders previously considered hopeless, but may also result in a population of children with long-term problems that also require study and clinical attention.
Pediatric Cardiology
As previously indicated, the vision of Dr. Taussig in devising a method to treat “blue babies,” in cooperation with pediatric cardiac surgeons, led to infants and children who survived surgery and then needed postoperative care. This sequence has been well documented by Dr. Jacqueline Noonan.33 She notes that, “Much success of the surgery can be attributed to a group of pediatric intensivists, pediatric intensive care units, improved ventilatory support, and trained respiratory therapists.” Advances in technology, especially for imaging, have allowed clinicians to “see” into living patients with astounding accuracy. Increased understanding of anatomy and physiology has led to improved surgical care for children with very complex problems. Perhaps ironically, some recent developments in cardiac catheterization and interventional radiology have enabled clinicians to treat many lesions without surgery, improving outcomes without the need for open-heart surgery and potentially difficult postoperative intensive care. The burgeoning growth of techniques, both interventional and surgical, has resulted in many centers creating specific cardiac intensive care units often run by pediatric cardiac intensivists, although not without some controversy in the world of PCCM.
Poliomyelitis
The interwoven history of resuscitation and ventilation, anesthesia, anatomy and physiology, pediatric anesthesiology, pediatric general and cardiac surgery, neonatology, and pediatric cardiology all come together in an astounding story of the treatment of paralytic polio and respiratory failure (“bulbar polio”). The confluence of great scientific and clinical minds and the organizational efforts of physicians, nurses, and technicians addressing the needs of polio patients rapidly led to the creation of PICUs. In 1929, Philip Drinker, an engineer, Dr. Louis Shaw, and Dr. Charles F. McKhann published their experience with a mechanical ventilator which was an electrically powered negative-pressure body tank, eventually termed the “iron lung” by a now unknown journalist (Figure 1-2).34 On October 12, 1928, an 8-year-old girl with polio and difficulty breathing was admitted to BCH. On October 13, her respiration was failing and she was placed in the respirator at low pressure. She improved and was taken off the device, but on October 14, she was comatose and cyanotic and was placed back in the respirator at high pressures. She regained consciousness and a little later asked for ice cream. “Most of the people who witnessed the scene were in tears.”14 Even though this patient died on October 19, with necropsy findings of poliomyelitis and bronchopneumonia, the device subsequently saved the lives of a student nurse at Bellevue Hospital in New York and a Harvard College student at Peter Bent Brigham Hospital.

Figure 1–2 The Drinker negative-pressure mechanical ventilator.
(Reproduced with permission, Blackwell Scientific Publications, Oxford.)
As dramatic as this was, it seems to be overshadowed by the remarkable polio epidemics in Los Angeles in the early 1950s and in Copenhagen in 1952.14 Writing in 1953,35 H.C.A. Lassen, Chief Epidemiologist of the Department of Communicable Disease, Blegdam Hospital, Copenhagen, describes treating 2772 patients for polio between July 24 and December 3, 1952. Of these, 866 patients had paralysis and 316 of these were in respiratory failure. Of the 316, 250 eventually underwent tracheotomy. Previously, starting in 1948, such patients underwent tracheotomy and suctioning for secretions without ventilatory support, but all died. Of the 15 patients treated with a mechanical respirator without tracheostomy, five patients, one adult and four children, survived. During the first month of the 1952 epidemic, of the 31 patients with respiratory paralysis, 27 died, for a mortality rate of 85% to 90%. Thereafter they consulted Dr. Bjorn Ibsen, an anesthesiologist, who suggested tracheotomy, rubber-cuff tubes, and manual positive-pressure ventilation (“iron lungs” were not commonly available in Europe at the time) using a rubber bag. From August 28 to September 3, 1958, they were admitting 50 patients a day, 12 of whom had respiratory failure and were admitted to a special unit for respiratory care. In this unit they had as many as 70 cases at once in respiratory failure. There were 200 patients admitted to the unit who underwent tracheotomy, manual positive-pressure ventilation with 50% oxygen, and suctioning. They employed 200 extra nursing auxiliaries (students and aides), 200 medical students at a time each working 8-hour shifts to provide manual ventilation (1000 in all), and 27 technicians per day to care for the patients.35–37 The mortality decreased from 90% to 40%. Ibsen adds that the first patient was a 12-year-old girl with paralysis of all four extremities and atelectasis of the left lung, who was gasping for air and drowning in her own secretions. She had a temperature of 40.7° C and was cyanotic and sweating. The tracheotomy was done under local anesthetic and a cuffed endotracheal tube was inserted. During the procedure she became unconscious. They connected her to the ventilator but could not ventilate her. He then gave 100 mg of pentothal IV and she collapsed, her own respirations stopped, and he could then provide manual ventilation. She then developed signs of carbon dioxide retention even with full oxygenation (rise in blood pressure, skin clammy, and sweating), and she again started her own respirations with gagging and bucking. Secretions began to pour out of her mouth and nose. This was relieved in a few moments with increased ventilation but then her blood pressure dropped and she appeared to be in shock. He gave a blood transfusion and her condition improved, with her skin becoming warm, dry, and pink, “Which always makes an anesthesiologist happy.” A chest radiograph showed atelectasis of the left lung and she was placed on a mechanical positive-pressure ventilator, after which all the signs of underventilation recurred, along with cyanosis. She was given supplemental oxygen and her color improved, but she still showed signs of carbon dioxide retention. Manual ventilation was started and she improved.36
He concluded that tracheotomy with local anesthetic without an endotracheal tube in place was too difficult. The patients were anxious, vomited, aspirated, and had airway spasms. Few survived, so they started doing the tracheotomies earlier with endotracheal intubation and anesthesia and had great success.36,37 Another change in strategy was that patients from outlying areas were being sent in ambulances without sufficient attendants and airway care and arrived moribund. They started to send teams in ambulances out to the pick up the patients in the countryside, with marked improvement (“retrieval teams”). This was the beginning of an important aspect of PCCM that many believe still has great potential for improving care in the future and which remains far from fully implemented. They also started passing stomach tubes for nutrition and the rubber-cuffed tubes were replaced with a silver cannula. Even with all the improvements he concludes, “Naturally we ran into a lot of complications.”37
Although these units tended to disband after the summer-fall polio season, they led to the creation of full-time units, the first of which was described by Dr. Goran Haglund in 1955, at the Children’s Hospital of Goteberg, Sweden.23 He called the unit a Pediatric Emergency Ward. The patient who inspired Dr. Haglund to organize the unit was a 4-year-old boy who was operated on in 1951 for a ruptured appendix. Postoperatively, he lapsed into a coma and the surgeon declared they had done all they could and he would die of “bacteriotoxic coma.” The anesthesiologist offered to help and the boy was intubated, given manual positive-pressure respiration with generous oxygen, tracheostomized, and given a large blood transfusion. After about 8 hours, the bowels started to move, and 4 hours later he was out of coma. After 20 hours, he had spontaneous respiration and had been successfully treated for respiratory insufficiency and shock. The unit had 7 acute care beds, 6 full-time nurses and 15 nursing assistants, with 24-hour coverage. In the first 5 years, the team treated 1183 infants and children, with a mortality rate of 13.6%. Haglund goes on to state, “But what we did was something else. It was the application of the basic physiology to clinical practice. Our main purpose was not to heal any disease, it was to forestall the death of the patient. The idea was—and is—to gain time, time so that the special medical and/or surgical therapy can have desired effects.”23 (Morriss and Levin38 took this approach in organizing the first edition of their textbook in 1979.) He was also careful to point out that, “There are few jobs more exacting, demanding, and taxing than emergency nursing. Our nurses and nurse assistants are tremendous. They must be!”23
Nursing
As has been shown, the dissemination of the knowledge and skills that the anesthesiologists had developed in the operating room to postoperative recovery rooms, surgical and medical wards, and eventually to geographically defined units, permitted improved treatment of patients with a variety of disorders, only some of which required surgical intervention. Among the diseases treated were polio in the 1920s to 1950s, tetanus in the 1950s and 1960s, and Reye syndrome in the 1970s and 1980s.3 These epidemics, along with developments in neonatology, pediatric general and cardiac surgery, and pediatric cardiology created a demand for greater services for more unstable patients. The events paralleled those in the world of adult critical care, with early intensive care units opened in 1923 at Johns Hopkins in Baltimore, a three-bed unit for postoperative neurosurgical patients directed by Dr. W.D. Dandy,10,39,40 in 1953 at North Carolina Memorial Hospital in Chapel Hill, North Carolina, in 1954 at Chestnut Hill Hospital, Philadelphia, in 1955 at the Hospital of the University of Pennsylvania in Philadelphia,41 and 1958 at Baltimore City Hospital (Dr. Peter Safar) and Toronto General Hospital (Dr. Barrie Fairley).3
Although many sources emphasize the role of advanced technology in the creation of adult, neonatal, and pediatric ICUs,3,24 it is interesting to consider the important role of nursing in this evolving process. Porter,42 as well as others, reminds us of the vital role of nursing in triage and organization of care for patients by degree of illness. Long before the organizational efforts just described, Florence Nightingale (1820-1920) organized the military hospital at Scutari in 1854, during the Crimean War, to provide more care to the most severely injured soldiers by grouping them together. Although the care consisted mostly of better hygiene and nutrition, the mortality rate dropped from 40% to 2%.43 These efforts were continued in the United States by Dorothea Dix (1802-1887) and Clara Barton (1821-1912), the “Angel of the Battlefield,” during the American Civil War, and when Barton brought the Red Cross to America in 1882. It was Nightingale who provided the definition of nursing as “helping the patient to live.”42,43 Fairman and Kagan41 conducted an interesting study looking at the creation and evolution of an adult intensive care unit by researching the historical records and interviewing the people involved at the Hospital of the University of Pennsylvania from 1950 to 1965. They emphasize that there really was no new equipment, only the migration of existing equipment from the operating room to the wards. In fact, some nurses remembered that they did not really have much in the way of equipment at all, even monitors. Certain social factors and the need for nurses were much more influential in forming geographically defined units away from the operating room or recovery room. Most patients at that time were operated on for gallbladder disease, appendectomies, and tonsillectomies. Poor patients were admitted to the ward postoperatively and wealthier patients were admitted to private or semi-private rooms and hired, at their own expense, private-duty nurses to care for them. This resulted in a two-tier system, with poorer patients having little postoperative care. Then a shortage of private-duty nurses occurred; many private-duty nurses refused to work nights, weekends, and holidays, and nurses with less training worked “off-shifts.” Surgeons and families complained they could not get care, and of course the poorer patients did not receive adequate care at all. The hospital, to save money, (the average cost per patient per day for recovery room care in 1960, at Baltimore City Hospital, was $50 to $80)44 demanded more from existing hospital nurses, tried to hire more private-duty nurses at family expense, and failing that, shifted some semiprivate patients to the ward. This resulted in complaints of noise (from patients) on the ward and understaffing to the point of safety concerns. One of the patients became disconnected from a ventilator and died unnoticed.
There was a move by nurses for better training, improved safety, and better staffing, as well as for more specialized rooms to organize the care of the sickest surgical and medical patients in architecturally distinct areas at no extra cost to the patients. This resulted in the creation of the Fifth Special Unit, closure of obsolete wards, and a more egalitarian admission policy to the special unit. There developed a shared sense of adventure between nurses and physicians in the ICUs, which seemed like experimental laboratories. Similar development was mirrored in PICUs, and the camaraderie and spirit were evident. The ICU nurse in adult, neonatal, and pediatric units rose to the top of the ladder in the hospital hierarchy. As one graduating Dartmouth Medical School student said in his class address, “When I started on clinical rotations I needed to learn how to function in the hospital. In order to do this I needed to understand the hierarchy in the institution. It quickly became apparent to me that the ICU nurse was at the very top of the pecking order.”45
Several other references to the central importance of nursing in creating and enabling intensive care to develop have been cited.3,8,23,30,31,42 As Fairman and Kagan41 conclude, “…powerful social contextual forces, such as workforce and economics, architectural changes, and an increasingly complex hospital population—rather than new technology—supported the development of critical care.”

Full access? Get Clinical Tree