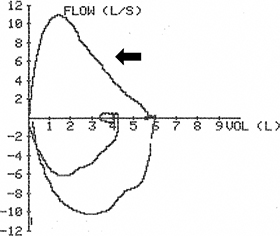
Timothy J. Harkin, John Pawlowski Chronic obstructive pulmonary disease (COPD) occurs frequently as a comorbidity in patients undergoing surgery and interventional procedures and can lead to increased hospital length of stay and increased morbidity and risk for mortality. For the anesthetist, the challenges continue through the intraoperative period and into the postoperative recovery. This chapter will focus on the current bronchoscopic procedures and the anesthetic management for patients who undergo these therapies. chronic obstructive pulmonary disease (COPD); intraoperative period; postoperative recovery; bronchoscopic procedures; endobronchial valve placement; volume reduction procedures; anesthetic management Emphysema is one component of chronic obstructive pulmonary disease (COPD), the third leading cause of death in the United States.1–3 Emphysema has been defined as “a condition of the lung characterized by abnormal permanent enlargement of the airspaces distal to the terminal bronchioles accompanied by destruction of their walls and without obvious fibrosis.”4 Emphysema often manifests with large air-containing bullae that do not contribute to gas exchange. Smoking-related emphysema is most commonly heterogeneously distributed throughout the lung, with upper lobe predominance (Fig. 32.1). Destruction of lung tissue may lead to increased compliance of the remaining lung, with change in compliance directly related to the amount and severity of emphysema present. Whereas other components of COPD, such as chronic bronchitis and bronchospasm usually coexist with emphysema to varying degrees in individual patients and contribute to obstructive dysfunction, the presence of severe emphysema alone is sufficient to cause expiratory airflow obstruction. In normal lungs, the diameter of airways during exhalation progressively decreases in conjunction with the decrease in lung volume. This leads to progressively decreasing flow rates throughout expiration, as exemplified by the characteristic downward slope of the expiratory limb of the flow-volume loop measured by spirometry (Fig. 32.2). The airways are tethered open during exhalation by the integrity of the lung parenchyma. The degree to which the diameter of the airways decreases during exhalation is directly related to this tethering effect, measured as the elastic recoil of the lung. The destruction of lung parenchyma by emphysema decreases elastic recoil (inversely increasing lung compliance), thereby increasing airway obstruction during exhalation by diminishing the tethering effect of the lung on the airways. This effect is exaggerated during forced expiratory maneuvers (Fig. 32.3), causing decreased values for measurements such as the forced expiratory volume in 1 second (FEV1). The obstruction to expiratory airflow may become severe enough to prevent complete exhalation of inspired air. Progressive “air trapping” may eventually lead to chronic hyperinflation of the lungs and increased total lung capacity (TLC). Hyperinflation in turn disrupts the mechanical advantage in the ventilatory design of the thorax, pushing the diaphragm downward toward the abdomen and pushing the ribcage outward. The degree of hyperinflation may be measured in the pulmonary function lab and expressed by the residual volume (RV), which is the amount of air that cannot be exhaled and remains in the lung at the end of a forced exhalation. Measurements in pulmonary function tests (PFTs) are usually expressed in absolute values and compared with predicted values based on the individual’s demographics, including age, sex, race, and height (percent or % predicted). The increase in TLC in a patient with COPD is mostly caused by an increase in RV in patients with very severe emphysema and hyperinflation, the RV may be increased to 150% to 200% of its predicted value or even more (Fig. 32.4). Hyperinflation is a significant contributor to dyspnea and decreased exercise tolerance in such patients, with a commensurate negative impact on quality of life (QOL). Many studies in emphysema treatment measure QOL using the St. George’s Respiratory Questionnaire (SGRQ). The SGRQ is a disease-specific quality of life assessment tool validated in both COPD and asthma. The questionnaire consists of 76 items divided into 3 parts measuring symptoms, activity limitation, and social impact. Higher scores indicate poorer QOL. A decrease of 4 points in the score is considered a significant improvement in QOL. Functional impairments in patients with COPD are also commonly measured with the 6-minute walk test (6MWT), which measures the distance walked during 6 minutes on a standardized course (6-minute walk distance [6MWD]). Current medical management focuses primarily on optimizing bronchodilation and minimizing airway inflammation with a complement of medications including beta-agonists, antimuscarinics, and inhaled corticosteroids, which do not address the loss of elastic recoil from emphysema. Recent estimates place the direct cost of COPD treatment in the USA at about $30 billion per year3; given that more than 3 million individuals in the United States suffer from emphysema, the need for improved treatment options is compelling. The goal of specifically decreasing hyperinflation in emphysema has been recognized as a potential means of improving symptoms and QOL if enough of the natural mechanical advantage of the thorax in ventilation can be restored. This led to exploration of surgical methods targeted to removal of portions of the most severely destroyed lung, as the absence of a significant portion of hyperinflated lung would presumably allow the diaphragm and ribcage to return to a more normal position. In the 1990s, Cooper pioneered the modern technique of lung volume reduction surgery (LVRS),5,6 building upon earlier work by Brantigan in 1959.7 This work led to the landmark National Emphysema Treatment Trial (NETT), which randomized patients with severe emphysema and dyspnea to standard medical therapy or surgical removal of the most severe regions of emphysema. Although the majority of patients experienced either no benefit or increased mortality from surgery, a subgroup of patients, with both upper-lobe predominant emphysema and low exercise capacity at baseline, derived a significant mortality benefit that extended throughout the 5-year follow-up (30% surgery vs. 50% medical therapy, P = .005). Importantly, this subgroup also experienced significant improvements in exercise capacity and health-related QOL, which was related to the decrease in hyperinflation. Unfortunately, the LVRS technique itself was also associated with significant morbidity (e.g., prolonged air leaks, prolonged hospitalizations) and mortality (5.2% vs. 1.5% at 90 days).8 Although NETT was invaluable in proving that selected patients with severe emphysema and dyspnea may benefit from decreasing hyperinflation via removal of a portion of destroyed lung, LVRS remains an uncommonly used technique.9 The incidence of postoperative complications were greater than the potential benefits. LVRS simply has not been a very appealing option to patients with severe emphysema and disabling dyspnea nor to the clinicians managing them. At its core, LVRS by definition is major thoracic surgery on patients with very poor lung function, which inherently imparts significant risk to individual patients and a major burden to the healthcare system in terms of costs and hospital days, while, at best, providing benefit to only a subgroup of patients with emphysema. These concerns naturally led to a new question: can a minimally invasive method be devised to specifically treat hyperinflation in chronically ill patients with very poor lung function and frequent comorbidities that is at least as efficacious as LVRS, with fewer complications, and applicable to a greater proportion of patients? Attempts to answer this question have revolved around bronchoscopic techniques, broadly described as bronchoscopic lung volume reduction (BLVR). A variety of BLVR modalities including methods to focally deflate (one-way valves or airway bypass) or minimize ventilation to areas of air trapping (coils), or functionally “remove” emphysematous lobes by focal fibrosis (tissue engineering with lung sealant or steam) have been explored in recent years. In 2018, the U.S. Food and Drug Administration (FDA) approved two different designs of one-way valves for the treatment of emphysema. The principle of one-way or unidirectional valves in bronchi leading to emphysematous areas of lung is based on a simple concept: valves that permit air to flow from the lung to the airway and thereby to the atmosphere during exhalation but do not allow air to flow from the atmosphere through the bronchus into the lung. This may reduce overall lung hyperinflation by means of regional deflation of the most emphysematous areas of the lung. Currently, there are two available valves with different designs, both made with a nitinol frame. Pulmonx (Redwood City, CA) manufactures the Zephyr valve with a silicone “duckbill” design (Fig. 32.5). Spiration (Redmond, WA) manufactures the Spiration Valve System (SVS), with an umbrella-like structure covered with a plastic polymer (Fig. 32.6). Both valves came under investigation nearly simultaneously soon after the NETT results were known. (Author’s note: Such valves have at times been described as “intrabronchial” or “IBV” usually referring to the Spiration valve, or “endobronchial” or “EBV” often referring to the Pulmonx valve; whereas these terms may be considered synonymous, “endobronchial valve” or “EBV” seems to have become the more commonly used term in discussions of these valves, often meant to apply to both currently available valves.) The one-way nature of the valves also permits lung secretions to move from the lungs to the airways, to minimize pooling of secretions in airways distal to the valves, thereby theoretically reducing the risk of postobstructive pneumonia. Both valves have a similar deployment strategy, using a catheter inserted through a flexible bronchoscope to deliver the valve to a segmental or subsegmental airway that ventilates a targeted area of emphysematous lung. Both valves are currently available in four sizes to enhance full occlusion of targeted airways and use different simple sizing techniques during the procedure. In distinction to LVRS, both valves are easily removed by bronchoscopy should clinical circumstances or patient preference dictate. Selection of appropriate patients for BLVR, as well as which specific airways and areas of lung to treat, has evolved over the past decade based on key studies described later. Early, nonrandomized studies suggested that certain patients with the combination of severe emphysema, dyspnea on exertion, and hyperinflation despite optimal medical therapy would benefit from the insertion of EBV. These studies primarily relied on some type of quantitative analysis of high-resolution chest computed tomography (CT) scans to confirm severity and regional distribution of emphysema and to target specific areas, primarily individual or bilateral upper lobes. In a study of the first 98 patients treated with an early version of the Zephyr valve, FEV1 increased by 11% and 6MWD improved by 23%.10 Similarly, in the Spiration pilot study, patients with severe upper lobe predominant emphysema and hyperinflation treated in both upper lobes experienced significant improvements in QOL, with an 8.2 decrease in SGRQ. This was accompanied by redirection of airflow to less diseased untreated lobes, although FEV1 and 6MWD did not change.11 However, results of early randomized studies were less encouraging or disappointing. In the Endobronchial Valve for Emphysema Palliation Trial (VENT) study, 321 patients with severe heterogeneous emphysema and hyperinflation (FEV1 15%–45%, TLC> 100%, RV > 150% predicted values) were randomized to treatment with the Zephyr valve in the most emphysematous lobe by quantitative CT (QCT) or medical therapy. VENT found small but significant improvements from baseline at 6 months for the primary endpoints FEV1 (increased 4.3%) and 6MWD (increased 2.5%) in the treatment group, whereas both FEV1 and 6MWD decreased in the control group (2.5% and 3.2%, respectively).12 The improvements found in the VENT trial were so modest that the FDA did not grant approval for use of the Zephyr valve based on the results. The IBV Valve Trial was a double-blinded, sham-controlled study of the Spiration valve in upper-lobe predominant severe emphysema with hyperinflation. All patients received bronchoscopy with appropriate anesthesia, during which airways in the upper lobes were measured with a balloon catheter for potential valve insertion. The patients were then immediately randomized 1:1 to treatment and control groups. Patients in the treatment group received valves in both upper lobes, although at least one segment or subsegment in each lobe was intentionally not treated (bilateral-partial lobe occlusion). Patients in the control group had continued bronchoscopy with simulation of valve insertion to provide the sham experience. Similarly to VENT, this study did not show clinically meaningful benefit in the treatment group. Only 5% of patients in the treatment group were considered responders by disease-related QOL measured by the SGRQ. Changes in treated lobe volume was significantly greater than in the control group (–224 mL vs. –17 mL), but the primary endpoint of a 10% or greater increase in nonupper lobe volume (signifying redirection of ventilation to less diseased lung) was not reached.13 Thus the improvements found in the early randomized trials were so slight that they were not likely to provide clinically significant benefit across the treatment group. In sum, the results of these studies seemed confusing. Why did some patients with severe emphysema clearly benefit from EBV treatment, while others with the same degree of hyperinflation and similar distribution of emphysema did not? The answer was suggested by subgroup analysis in the VENT study. Individuals whose QCT identified greater heterogeneity of emphysema had greater improvements in FEV1 and 6MWT (Fig. 32.7), and those whose QCT found complete fissures separating targeted from nontargeted lobes had greater improvements in FEV1. Specifically, at 6 months postprocedure, patients in the treatment group with complete fissures had improvement in FEV1 of 16.2% versus 2.0% in patients with incomplete fissures (P < .001). The authors noted: “Greater heterogeneity of emphysema between lobes and intact interlobar fissures appear to identify patients with a greater likelihood of clinically important functional and physiologic responses to endobronchial-valve therapy.”12 The significance in this finding is related to the role of intact fissures in preventing air from passing directly from one lobe to another within the lung parenchyma as opposed to ventilation exclusively provided by the bronchi. The knowledge of collateral ventilation (CV) between adjacent lobes of the lung in certain people was not new, but the amount of CV across presumed fissural boundaries, its frequency, and clinical significance had not fully been appreciated previously.14 Based on this finding, the Pulmonx Chartis System was designed to confirm isolation of lobar ventilation and lack of CV between adjacent lobes.15 This system uses a balloon-tipped catheter passed through a flexible bronchoscope to occlude a lobar bronchus, and measures air flow and airway pressure from the lobe into the catheter. As the lobe cannot be ventilated from proximal airways while occluded by the balloon, air flow will gradually diminish if CV from an adjacent lobe is not present (Fig. 32.8). Using Chartis to identify presence or absence of CV in 80 patients immediately before insertion of Pulmonx EBV, Herth et al. found evidence of CV in 29 patients. The 51 patients without CV had significantly greater improvements in FEV1 and greater reductions in volume of the treated lobe as measured by QCT than patients with CV.16 Selection of patients for BLVR with valves who are likely to have little or no CV based on presence of complete fissures on QCT analysis and/or evaluation with Chartis proved critical in subsequent trials showing efficacy of EBV treatment. For instance, additional support for the importance of assessment of CV in targeted lobes was provided by the Bronchoscopic Lung Volume Reduction for patients with Heterogeneous emphysema and Intact Fissures (BeLieVeR HIFi) study, (BLVR) in which entry criteria included FEV1 under 50%, TLC greater than 100%, and RV over 150% in patients with heterogeneous emphysema and more than 90% intact fissures on chest CT. Davey et al.16b reported on this single center double-blind sham-controlled study in which 50 patients had a target lobe identified by CT destruction scores and were randomized 1:1 before bronchoscopy. During bronchoscopy, CV in the target lobe was assessed by Chartis, and then valves were inserted in the target lobe in the treatment group irrespective of Chartis findings, whereas the control group had sham valve placement. Among the treated group in whom absence of CV was confirmed by Chartis, 47% had more than 15% improvement in FEV1, 58% had greater than 0.35 L improvement in RV, and 58% had significant improvement in QOL measured by SGRQ. In contrast to most other valve trials, which exclusively included patients with heterogeneous emphysema, patients with homogeneous emphysema and severe hyperinflation (FEV1 < 45%, TLC > 100%, and RV > 200%) were studied in the Informing the Pathway of COPD Treatment (IMPACT) trial. Homogeneous emphysema was defined as less than 15% difference in emphysema destruction score between target and ipsilateral lobes. Lack of CV in primary or secondary target lobes was confirmed by Chartis, after which 93 subjects were randomized 1:1 to immediate treatment with the Zephyr valve (n = 43) or standard of care (n = 50). Improvement in FEV1 of at least 15% was achieved in 35% of the treated subjects, with significant improvement in QOL measured by SGRQ in 57%. COPD exacerbation requiring hospitalization occurred in 16% of treated patients versus 12% in control group (not significant), and 25.6% of treated patients experienced pneumothorax (vs. none of controls).17 The Zephyr Endobronchial Valve Treatment in Heterogeneous Emphysema (LIBERATE) study examined use of the Zephyr EBV in patients with severe emphysema and hyperinflation (FEV1 15%–45%, TLC > 100%, RV> 175% predicted values). Exclusion criteria included severe carbon dioxide retention (partial pressure of carbon dioxide [PCO2] > 50 mm Hg breathing room air) and pulmonary hypertension (systolic pulmonary artery pressure [SPAP] > 45 mm Hg). Subjects were randomized to either EBV or standard of care (SoC) immediately after lack of CV was confirmed by Chartis, whereas subjects found to have CV with Chartis exited the study. The single target lobe treated with EBV was selected based on greater than 50% emphysema destruction score and heterogeneous emphysema defined by degree of difference in destruction between target and ipsilateral lobes as measured by a QCT analysis (Fig. 32.9). A median of four valves were placed per subject. At 12 months, 48% of EBV patients achieved the primary outcome of 15% or more improvement in FEV1 compared with 17% of SoC patients. All secondary endpoints improved with statistical significance in favor of EBV. The difference of FEV1 from baseline at 12 months was 0.106 L compared with SoC. 6MWD improved 39 m, and QOL measured by SGRQ also significantly improved, by 7 points. The most important and common adverse event was pneumothorax, which occurred in 27% of EBV subjects, with none in the SoC group. Importantly, 76% of pneumothoraces occurred within 3 days of bronchoscopy.18 The Evaluation of the Spiration Valve System for Emphysema to Improve Lung Function (EMPROVE) study examined use of the SVS in subjects with severe emphysema and hyperinflation (FEV1 < 45%, TLC > 100%, RV > 150% predicted values). Exclusion criteria included severe carbon dioxide retention (PCO2 > 50 mm Hg breathing room air) and severe pulmonary hypertension. At a preprocedural visit, 174 subjects were randomized 2:1 to either treatment with SVS or the control group. The single target lobe treated with SVS was selected based on 40% or more emphysema destruction and 10% or more difference in emphysema severity between target and ipsilateral lobes as measured by a QCT analysis. The primary outcome was the mean change in FEV1 at 6 months, which found a 0.1 L difference from baseline in the treatment group, with a between-group difference of 0.1 L compared with controls. A mean of about four valves were placed per subject. At 6 months, 37% of SVS patients achieved the secondary outcome of 15% or more improvement in FEV1 compared with 10% of control patients. All secondary endpoints improved with statistical significance in favor of SVS, including SGRQ, which improved by 9.5 points. Mean reduction in target lobe volume (TLV) was nearly 1 L; 75% of the treatment group experienced a clinically meaningful reduction of TLV of least 350 mL, and 40% experienced complete atelectasis of the treated lobe. The 6MWD improved by only 6.9 m in the treatment group. Serious adverse events occurred in 31% of the treatment group compared with 11.9% in controls. A total of 32 pneumothoraces occurred in 30 treated patients (27%), 66% of which occurred within 3 days of bronchoscopy.19 Building on nearly 2 decades of investigation of BLVR, the LIBERATE and EMPROVE studies achieved the goal of finding a minimally invasive means to treat hyperinflation that is safe and effective in a significant number of patients. The recognition of the essential contribution of intact fissures in the selection of patients most likely to benefit from valve treatment was the key factor that led to successful trials of the two valves. Based on the results of LIBERATE and EMPROVE, in 2018, the FDA granted approval to both the Pulmonx Zephyr valve and the Spiration SVS for the treatment of severe emphysema. As previously discussed, pneumothorax is the most common serious complication following EBV treatment and may manifest as either an early (<3 days, up to 76%) or late complication up to 12 months after valve insertion (three pneumothoraces during 6–12-month follow-up in EMPROVE). Serious pneumothoraces may require removal of one or more valves to resolve, and both LIBERATE and EMPROVE followed pneumothorax management protocols that were defined in the respective study design. A recent metaanalysis of five randomized controlled trials (not including LIBERATE or EMPROVE) with 703 patients found a relative risk of 8.16 for pneumothorax after EBV treatment.20 In general, the lobe with the most lung destruction should be targeted. This may not be possible if CV is present in that lobe, and if there is good candidate lobe for treatment on the contralateral side that may be considered for treatment. However, if the target lobe is not the most diseased, and the contralateral lung had more than 60% lung destruction, the LIBERATE trial suggests that valve treatment has increased risk of pneumothorax, which may be fatal.18 Death related to pneumothorax occurred in three patients in LIBERATE (two on day 3 and one on day 13), and one in EMPROVE (on day 95). Given the significant risk of pneumothorax within 3 days of valve insertion, patients must be observed in the hospital for a minimum of 3 days following the procedure. All staff involved in the care of the patient must be alerted to the manifestations of pneumothorax, and all relevant equipment to treat pneumothorax urgently should be kept at the bedside. Patients also must be educated regarding the symptoms of pneumothorax and instructed to seek medical care immediately if these symptoms develop.
Endoscopic Treatment of Chronic Obstructive Pulmonary Disease
Abstract
Keywords
One Way Valves in the Treatment of Emphysema and Persistent Air Leaks
Emphysema



Surgical Treatment of Emphysema and its Offspring (see Chapter 29)
A Brief History of Valves in the Treatment of Emphysema




The Definitive Valve Trials: LIBERATE and EMPROVE

Pneumothorax Following Valve Insertion

Full access? Get Clinical Tree


Endoscopic Treatment of Chronic Obstructive Pulmonary Disease






