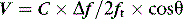Chapter 23 Echocardiography in intensive care
Echocardiography refers to a group of interrelated ultrasound applications used to examine the heart and great vessels. The reflected and processed sonic waves are displayed on a monitor and the images can be stored on videotape or disk. Echocardiography includes: (1) two-dimensional (2-D) anatomical imaging; (2) M-mode echocardiography, usually obtained with 2-D guidance; and (3) Doppler techniques. Echocardiography is a safe non-invasive technique which is integral to clinical cardiology. More recently, echocardiography has evolved into a powerful diagnostic and management tool in critically ill patients, especially in cardiovascular emergencies of uncertain cause.1,2
Cardiologists, although highly skilled in echocardiography, may not appreciate the complex pathophysiology of critically ill patients in intensive care, and they have time commitments elsewhere. Echocardiography is operator-dependent; optimal image acquisition requires both technical knowledge of the machine’s capabilities as well as a degree of manual dexterity. As a general rule, echocardiography is not an efficient technique for monitoring haemodynamic indices over the medium term (hours–days) in the intensive care unit (ICU). Echocardiography can help determine whether and when continuous haemodynamic monitoring should be commenced.3 Repetitive trend measurements, such as stroke volume, cardiac output and pulmonary artery wedge pressure, are more suited to the pulmonary artery catheter. Moreover, intensive care staff are skilled and experienced in its use. Echocardiography technicians are not present in ICU, especially out of hours; intensivists must be able to act as their own sonographers as well as interpreting and reporting on the echographic images obtained. It is important that intensivists acquire all requisite skills so that echocardiography becomes more widely available.
PRINCIPLES AND TECHNICAL CONSIDERATIONS4
2-D ECHOCARDIOGRAPHY
2-D echocardiography is the cornerstone of cardiac ultrasound as Doppler and M-mode are usually performed with reference to the 2-D image. Each 2-D image is defined by the position of the transducer (acoustic window) and image plane which is determined by the axis of the heart and not the spine (Table 23.1).
Table 23.1 Some standard views in transthoracic and transoesophageal echocardiography
| Acoustic window | Image plane |
|---|---|
| Transthoracic | |
| Parasternal | Long axis |
| Short axis | |
| Apical | Four-chamber |
| Two-chamber | |
| Long axis | |
| Subcostal | Multiple |
| Transoesophageal* | |
| Transgastric | e.g. Short axis |
| Long axis | |
| Deep transgastric | e.g. Long axis/five-chamber |
| Lower transoesophageal | e.g. Four-chamber |
| Two-chamber | |
| Long axis | |
| Upper transoesophageal | e.g. Short axis of aortic valve |
| Long axis of aortic valve | |
| Right ventricular inflow–outflow | |
M-MODE ECHOCARDIOGRAPHY
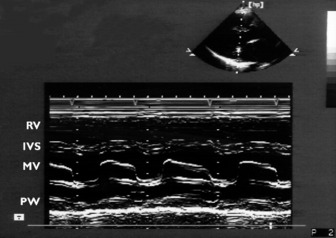
Rapidly repeated transmit and receive cycles allow production of images with good resolution. M-mode echocardiography complements 2-D echocardiography. The beam is not scanned but stays in a fixed position, allowing the display of structures along the beam as a function of time. The rapid sampling rate makes identification of thin moving structures, such as valve leaflets, relatively easy. The cursor is usually directed through a 2-D image, giving a one-dimensional slice or ‘icepick’ view. Distance (depth) is on the vertical and time is on the horizontal axis (Figure 23.1).
DOPPLER ECHOCARDIOGRAPHY4,5
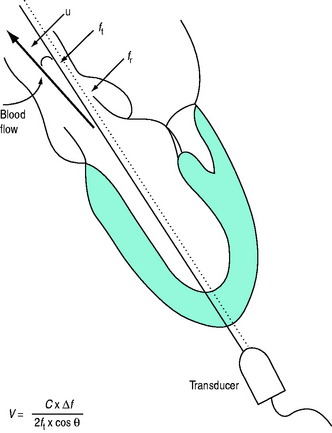
where V = velocity of blood flow, C = speed of sound in soft tissue (1540 m/s), Δf = Doppler (frequency) shift:difference in frequency between received (fr) and transmitted (ft) ultrasound and θ = angle between ultrasound beam and direction of blood flow (Figure 23.2). Echocardiographic machines contain computers that automatically calculate the Doppler shift which is then entered into the Doppler equation. Blood flow velocity (m/s) is calculated and displayed on the monitor.
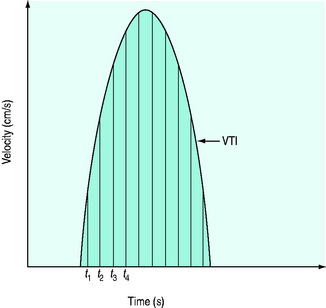
It is very important that the ultrasound beam is parallel or nearly parallel with the direction of blood flow. Ifθ equals 0 then cosign θ equals 1; however, if θ > 20° then the velocity of blood flow will be significantly underestimated. Doppler data are processed and a spectral display plots instantaneous blood velocities over time. Blood flow velocity can be expressed as peak velocity or mean velocity throughout a cardiac cycle (velocity–time integral: VTI) (Figure 23.3).
CONTINUOUS-WAVE DOPPLER
The transducer consists of two crystals: one continuously transmits and the other continuously receives ultrasound waves. It can measure high-velocity blood flow, such as occurs in aortic stenosis. Unlike PW Doppler, CWDoppler measures all the frequency shifts along the beam path and it is not possible to estimate the velocity at a specific site (range ambiguity).
Blood flow velocities measured by either PW or CW Doppler can be converted to pressure gradients using the simplified Bernoulli equation, ΔP = 4V2 (where ΔP is the instantaneous pressure gradient and V is the instantaneous velocity), to provide estimates of pressure.
THREE-DIMENSIONAL
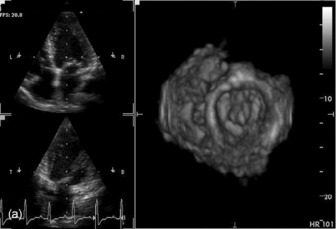
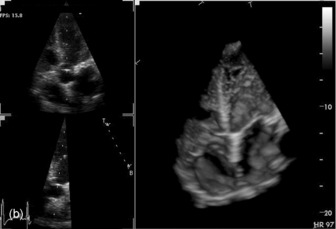
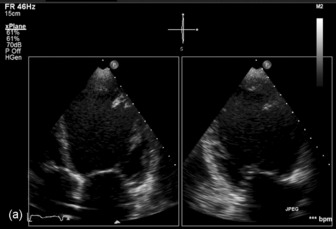
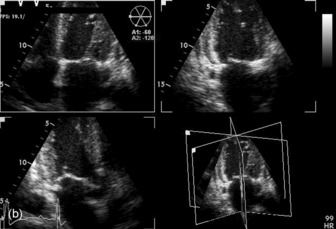
Three-dimensional (3-D) echocardiography is an evolving technique. Current equipment provides ‘real-time’ 3-D images (Figure 23.4a). The acquisition of a complete volume data set typically requires four sequential cardiac cycles. This 3-D data set must then be viewed and analysed. The full 3-D volume can then be ‘cropped’ so that the interior is exposed (Figure 23.4b). The 3-D volume set can be ‘opened’ in any imaging plane, providing both long- and short-axis views of cardiac structures. The 3-D volumetric probe can also be used for simultaneous acquisition of true real-time biplane (Figure 23.5a) and triplane (Figure 23.5b) images.
TRANSTHORACIC (TTE) AND TRANSOESOPHAGEAL ECHOCARDIOGRAPHY
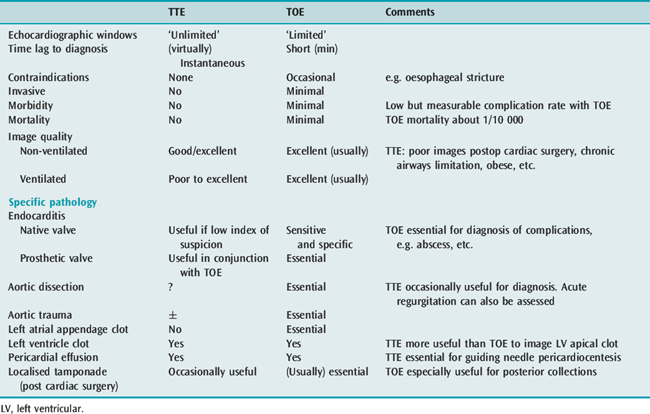
TTE and TOE are complementary ultrasound techniques, each with its own strengths and weakness (Table 23.2). For example, a vegetation imaged on the mitral valve of a septic patient will usually require a TOE to evaluate complications of endocarditis.
Table 23.2 Transthoracic echocardiography (TTE) and transoesophageal echocardiography (TOE): advantages and disadvantages
TRANSOESOPHAGEAL ECHOCARDIOGRAPHY6,7
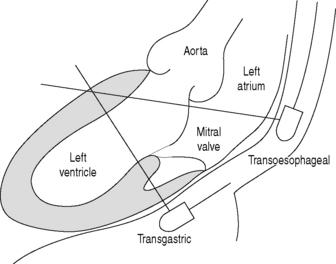
The transducer is mounted at the tip of a flexible gastroscope-like probe which can be manoeuvred to various positions in the oesophagus and stomach close to the heart (Figure 23.6).
The modern multiplane probes allow rotation of the ultrasound scanning plane from 0 to 180°, which provides a mirror-image orientation. Multiple tomographic imaging planes can be obtained without the necessity for further probe manipulation. Compared to TTE, TOE provides additional information in 32–100% of intensive care examinations and unexpected new diagnoses in 38–59%, leading to significant changes in treatment.8 Approximately 20% of patients with unexplained hypotension require surgery on the basis of new TOE findings.9,10
INDICATIONS FOR ECHOCARDIOGRAPHY11,12
The indications for echocardiography broadly fall into three categories:
Some common syndromes in which echocardiography is especially useful are listed in Table 23.3.
Table 23.3 Indications for echocardiography in the intensive care unit according to clinical syndrome
| Clinical syndrome | Findings | Comments |
|---|---|---|
| Hypotension | ||
| Acute myocardial infarct | ||
| No murmur | LV RWMA(s) | Usually severe ↓ LV systolic function |
| RV RWMA | RV ↓ > LV ↓ | |
| Hypovolaemia | ‘Empty’ LV cavity, systolic function largely preserved | |
| Acute MR | Often no murmur with normal LA size | |
| New murmur | Papillary muscle rupture (partial or complete) | ‘Good’ LV function with severe MR |
| VSD | RWMA. High-velocity left-to-right systolic jet | |
| Rarely | Cardiac rupture/tamponade | Acute pericardial tamponade, usually fatal |
| LV pseudoaneurysm | Containment of rupture | |
| Cardiothoracic surgery | Cardiac tamponade | Often localised. There may be no ‘echo-free space’ due to clot compressing the heart. Cardiac filling pressures may be normal |
| (New) RWMA | May be due to graft occlusion, air embolism | |
| Hypovolaemia | ‘Empty’ LV with RWMA | |
| Global LV ↓ | Stunning or long-standing cardiomyopathy | |
| Valvular dysfunction | Often long-standing but may be worsened by surgery, e.g. ischaemia to a papillary muscle of mitral valve | |
| Dynamic LVOT obstruction | LVOT pressure gradient, SAM, MR | Inotropes/IABP/hypovolaemia worsen LVOT obstruction |
| Trauma | Hypovolaemia | ‘Empty’ LV with vigorous contraction |
| Cardiac contusion | RWMA (RV > LV) | |
| Valvular injury | Most common aortic valve (AR) or mitral valve (MR), occasionally tricuspid valve (TR) | |
| VSD/ASD | Occasionally | |
| Cardiac tamponade | More common in penetrating chest injuries | |
| Ruptured thoracic aorta | Widened mediastinum (90%) at isthmus of aorta. TOE required | |
| Sepsis | Often normal LV systolic function with ‘empty’ LV | Possible global/regional LV depression |
| Infective endocarditis – vegetations/abscess/regurgitation | TOE more sensitive than TTE for imaging vegetations/abscess/fistula | |
| ‘Isolated’ hypotension (‘hypotension ?cause’) | Global LV ↓ or RWMA | Cardiomyopathy or stunning |
| Valvular dysfunction | Usually chronic, occasionally acute (e.g. ruptured papillary muscle of mitral valve) | |
| Dynamic LVOT obstruction | ||
| Acute cor pulmonale | Pulmonary embolism – usually dilated right heart with depressed systolic function, sometimes with clot in a proximal pulmonary artery | |
| Sepsis (source?) | Vegetations, regurgitation ± abscess | Infective endocarditis until proven otherwise |
| Normal TOE examination | Infective endocarditis unlikely. If clinically indicated serial TOE | |
| Systemic emboli (source?) | LA/LAA clot | Usually enlarged LA and atrial fibrillation TOE usually required for diagnosis |
| LV clot | Usually associated with RWMA or global LV depression. TTE for apical clot | |
| Aortic atherosclerotic | TOE essential for diagnosis plaques | |
| Vegetations – aortic or abscess | Septic? | |
| Clot – prosthetic aortic or mitral valve | Associated prosthetic valve dysfunction | |
| Patent foramen ovale with paradoxical embolism | RA pressure > LA pressure (e.g. IPPV with high PEEP). TOE (bubble contrast) usually necessary for diagnosis | |
| Tumour (e.g. LA myxoma) | Uncommon | |
| Pulmonary oedema (cause?) | ↓ LV systolic/diastolic function | Isolated LV diastolic dysfunction not uncommon |
| Valvular dysfunction (MR, MS, AR, AS) | If flail leaflet/ruptured papillary muscle suspected then TOE indicated | |
| Intracardiac shunt | ||
| Normal | Suggests non-cardiac cause (e.g. ARDS) | |
| Dyspnoea/hypoxia without pulmonary oedema (dyspnoea cause?) | Pulmonary embolism (dilated right heart chambers ± clot in pulmonary artery) | Infers moderate to large clot burden i.e. submassive/massive PE |
| Cardiac tamponade | Usually other clinical signs of tamponade present | |
| Miscellaneous e.g. chronic cor pulmonale, RVH, constrictive pericarditis, diastolic dysfunction, intracardiac shunt, RV volume overload | ||
| Chest pain of uncertain aetiology (chest pain cause?) | RWMA | Infers presence of coronary artery disease |
| Dissecting aortic aneurysm (intimal flap, true/false lumen) | TOE more sensitive than TTE | |
| PE (dilated right heart chambers ± clot in pulmonary artery) | Moderate to large embolus | |
| Pericarditis | Effusion often too small to diagnose with echocardiography | |
| Aortic stenosis | Clinical signs of stenosis may be absent | |
AR, aortic regurgitation; ARDS, acute respiratory distress syndrome; AS, aortic stenosis; ASD, atrial septal defect; IABP, intra-aortic balloon pump; IPPV, intermittent positive-pressure ventilation; LA, left atrium; LAA, left atrial appendage; LV, left ventricle; LVOT, left ventricular outflow tract; MR, mitral regurgitation; MS, mitral stenosis; PE, pulmonary embolism. PEEP, positive end-expiratory pressure; RA, right atrium; RV, right ventricle; RVH, right ventricular haemorrhage; RWMA, regional wall motion abnormality; SAM, systolic anterior motion; TOE, transoesophageal echocardiography; TR, tricuspid regurgitation; VSD, ventricular septal defect.
As a general rule, unless there is gross haemodynamic instability or some other overriding reason, the echocardiographic examination should be structured around a basic set of 2-D views. This applies to both TTE and TOE. The focus of the TOE examination should be on the most important specific clinical question. The second priority should be other potential pathology in the differential diagnosis. A printed report should be performed after each echocardiographic examination and should be stored and available for future reference.13
VALVULAR HEART DISEASE14,15
Echocardiography is the ‘gold standard’ for evaluating valvular heart disease. 2-D echocardiography provides excellent imaging of all cardiac valves. Various Doppler techniques allow accurate haemodynamic evaluation of the valves.
VALVULAR STENOSIS
Aortic stenosis
Assessment of severity
Visual assessment of aortic valve cusp thickness, calcification, mobility and degree of LV hypertrophy will often indicate the physiological significance of the aortic stenosis. Doppler echocardiographyis essential for assessment of severity: (1) peak aortic velocity; (2) mean transvalvular pressure gradient; (3) aortic valve area; and (4) ratio of LVOT velocity/aortic valve velocity (V1/V2).

Full access? Get Clinical Tree