Fig. 12.1
Structure of chlorhexidine (1:6-di[4-chlorophenyldiguanido]-hexane). The two biguanide groups (green arrows) are protonated at physiological pH (as shown in the yellow box) so that the molecule is positively charged
Chlorhexidine binds strongly to proteins in the skin and mucosa, thus resulting in a persisting topical antimicrobial effect with limited systemic absorption. Particularly, chlorhexidine in mouthwash solutions binds to oral mucosal surfaces via electrostatic forces, inhibits dental plaque formation, and exerts a bacteriostatic action that persists for several hours.
The antimicrobial activity of chlorhexidine depends on its concentration. At bacteriostatic concentrations, once adsorbed onto phosphate-containing proteins in the bacterial cell wall, it penetrates and disrupts the bacterial cytoplasmic membrane, resulting in leakage of cytoplasmic components. At higher concentrations, it exerts a bactericidal action by forming irreversible precipitates with intracellular adenosine triphosphate (ATP) and nucleic acids after entering the cytoplasm via the damaged cytoplasmic membrane (Fig. 12.2).
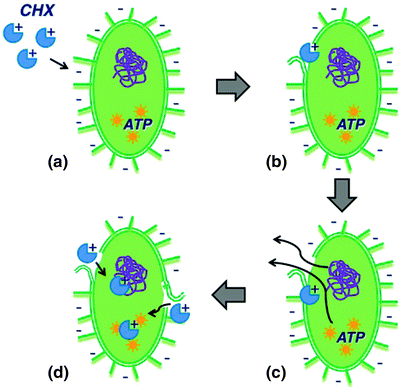

Fig. 12.2
Mechanism of action of chlorhexidine. Biguanide groups bind strongly to exposed anionic sites on the cell membrane and cell wall (a), thereby producing loss of membrane integrity (b) and leakage of ions and other cytoplasmic components (c). At higher concentrations, once entered the cytoplasm through the damaged membrane, chlorhexidine forms irreversible precipitates with intracellular ATP and nucleic acids (d) and cell lysis occurs (not shown). CHX Chlorhexidine; ATP Adenosine triphosphate
Chlorhexidine is most active against Gram-positive bacteria due to its increased affinity for the cell wall of Gram-positive organisms. However, it has also activity against Gram-negative bacteria, anaerobes, fungi, and some enveloped viruses. Moreover, prolonged exposure increases the bactericidal effect for most bacteria [14, 19].
Among the four possible mechanisms that lead to nosocomial pneumonia (aspiration of oropharyngeal organisms, either directly from the oropharynx or, secondarily, by reflux from the stomach into the oropharynx; inhalation of aerosol containing bacteria; hematogenous spread from distant body sites; bacterial translocation from the gastrointestinal tract), the major route of pulmonary infection in endemic VAP is aspiration of oropharyngeal secretions colonized by nosocomial organisms [5–7, 12], especially S. aureus—the most common pathogen involved in VAP, with over one half being methicillin resistant (MRSA)—or enteric Gram-negative bacilli [7].
While in healthy patients the oropharyngeal flora is mainly composed of Streptococcus viridans, Haemophilus species, and anaerobes, colonization of the oropharynx by aerobic Gram-negative bacilli and S. aureus is an almost universal occurrence in critically ill patients receiving mechanical ventilation. In fact, bacterial adherence to the orotracheal mucosa is fostered by reduced mucosal immunoglobulin A and increased protease production, exposed and denuded mucous membranes, elevated airway pH, and increased number of airway receptors for bacteria.
Chlorhexidine as a 0.12 % oral rinse applied for 30 seconds is effective in reducing dramatically aerobic and anaerobic bacterial counts and, as mentioned previously, its action is long lasting due to its adsorption onto oropharyngeal soft tissues and subsequent release over time [12]. Due to the activity on oral microbial flora and its role in the pathogenesis of VAP, chlorhexidine is one of the few measures for VAP prevention showing an impact on patients’ outcomes, together with noninvasive positive pressure ventilation, sedation and weaning protocols, selective digestive decontamination, and endotracheal tube with drainage of subglottic secretions, with a survival benefit limited to selective oral/digestive decontamination and daily spontaneous breathing trial associated with sedative interruption [6].
12.4 Therapeutic Use
Oral chlorhexidine appears to be effective in reducing VAP incidence in several studies, but the regimens employed range from 0.12 % chlorhexidine twice daily, as in the investigation by DeRiso et al. [5], to 2 % chlorhexidine four times a day [1, 5, 8–10, 12, 13]. While there seems to be no difference between once or twice daily administration [16], the recent meta-analysis by Labeau et al. [8] reports a more pronounced favorable effect in reducing VAP incidence in subgroup analyses for 2 % chlorhexidine.
Adverse effects of chlorhexidine are rare [19]. Those documented in the dental literature include mucosal irritation, staining of the teeth, which is easily reversible with professional cleaning and, occasionally, a transient abnormality of taste that dissipates with prolonged use and is totally reversible with discontinuation [5, 16]. Although oral ingestion of chlorhexidine is usually well tolerated due to negligible systemic absorption, adverse effects such as pulmonary edema, acute respiratory distress syndrome, and liver toxicity can occur when large doses are ingested [19].
Hypersensitivity (anaphylactic or anaphylactoid) reactions to chlorhexidine may occur, but their exact incidence is difficult to estimate [19, 20]. However, more severe reactions seem to be related to chlorhexidine-coated central venous catheter use and none of the aforementioned investigations about oral chlorhexidine for VAP prevention in ICU patients reports safety issues, with regard to both adverse effects or hypersensitivity reactions.

Full access? Get Clinical Tree




