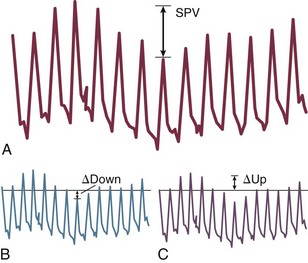
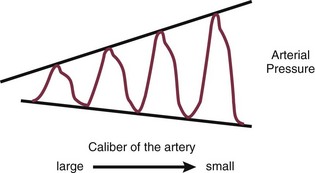
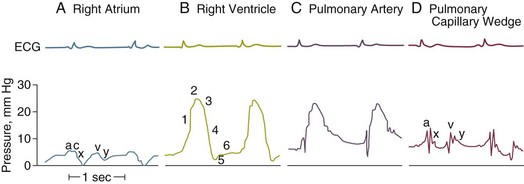
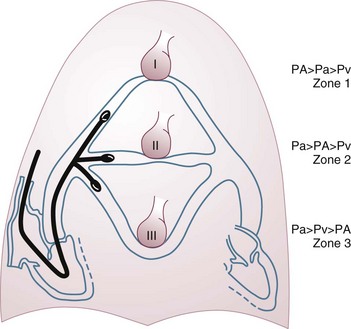
4 The optimal range of arterial pressure depends on individual patient characteristics, on underlying diseases, and also on treatment. Hence, it is impossible to give an optimal range of arterial pressure that is applicable in all patients. When arterial pressure needs to be evaluated accurately, oscillometric measurements become unreliable,1 and insertion of an arterial catheter is indicated. 1. Hypotensive states associated with (a risk of) altered tissue perfusion. Hypotension that is resistant to fluid administration requires the administration of vasopressor agents, and invasive measurement of arterial pressure is then necessary to titrate this form of therapy. Norepinephrine is the vasopressor agent most commonly used in this setting. A mean arterial pressure (MAP) of 65 to 70 mm Hg is usually targeted, but this level must be adapted to the individual patient and the clinical scenario; in particular, elderly patients with atherosclerotic disease may require higher levels than younger individuals with normal arteries. 2. Intravenous vasodilator therapy. Vasodilating therapy (e.g., nitrates and hydralazine) is a mainstay in the management of heart failure, because it can increase cardiac output. Close monitoring of arterial pressure is essential to avoid excessive hypotension. 3. Severely hypertensive states. Extreme hypertension may result in organ impairment, especially of the brain and the heart. Sodium nitroprusside or calcium entry blockers usually are used to lower arterial pressure, and careful, accurate monitoring is essential to titrate the antihypertensive therapy. 4. Induction of hypertension. Hypertension is sometimes induced in patients with neurologic diseases. Severe cerebral edema with intracranial hypertension, in particular, requires vasopressor support to maintain cerebral perfusion pressure (the gradient between the MAP and the intracranial pressure); likewise, hypertension may be used to treat or prevent the development of vasospasm secondary to subarachnoid hemorrhage, as part of the so-called triple-H therapy (hypertension, hypervolemia, hemodilution). Norepinephrine usually is used for this purpose. Variations in arterial pressure during positive-pressure ventilation have been used as a measure of fluid responsiveness. The transient increases in intrathoracic pressure influence venous return in patients who are likely to respond to fluid administration. This fluctuation in ventricular filling will translate into fluctuations in arterial pressure a few beats later. Accordingly, the greater the degree of systolic arterial pressure, or pulse pressure, variation during the respiratory cycle, the greater will be the increase in cardiac output in response to fluid administration (Fig. 4.1). However, this observation is valid primarily in patients without spontaneous respiratory movements and without significant arrhythmias, and only when a sufficient tidal volume is applied.2,3 For placement of arterial catheters, usually the radial artery is used. The femoral artery can be easily cannulated and gives a better signal, but presence of a femoral catheter interferes more with patient mobility and warrants concern about infection.4 Use of other sites, such as the brachial or the axillary artery or even the dorsalis pedis artery,5 can be considered. An important point to keep in mind is that the pulse pressure increases from the core to the periphery. In other words, the systolic pressure is overestimated in smaller arteries (Fig. 4.2). Hence, it may be better to rely more on mean values than on systolic or diastolic pressures. Clinically, CVP can be assessed by evaluation of the degree of jugular distention6 or liver enlargement. A single CVP measurement is not very useful and is not a good indicator of a positive response to fluids; an increase in CVP without a concurrent increase in cardiac output is not only useless but also harmful, because it will lead to increased edema formation. Measuring the central venous oxygen saturation (ScvO2) is a surrogate for measurement of the true mixed venous oxygen saturation ( The central venous catheter generally is introduced via the internal jugular vein; the subclavian vein also can be used, although the risk of pneumothorax may be somewhat higher with this route. Peripherally inserted central catheters can also be placed, via the cephalic vein, basilic vein, or brachial vein. Introduction of a femoral catheter through the abdominal inferior vena cava to the right atrium can also yield reliable CVP measurements.8 The use of femoral catheters, however, is associated with a greater risk of infections and thrombophlebitis.9 Catheter-related infections constitute the major long-term complication. Adherence to basic hygiene guidelines can decrease the incidence of catheter-related sepsis. Triple-lumen catheters may be associated with a higher incidence of catheter-related infection,10 primarily as a result of increased catheter manipulation. The use of antimicrobial-coated catheters may decrease the risk of infections,11 but fears remain about the risks of development of resistant organisms.12 Routine replacement of catheters after 3 to 7 days is not recommended.13 PACs allow collection of data on right atrial, pulmonary artery, and pulmonary artery occlusive pressures (Fig. 4.3); flow (cardiac output); and oxygenation ( As indicated earlier, the RAP is identical to the CVP in the vast majority of cases. A first question is whether the PAOP reflects the pressure in the pulmonary veins and not the alveolar pressure. The tip of the catheter should be in a West zone III position, where a continuous column of blood exists between the catheter tip and the left atrium (Fig. 4.4). These considerations are less important with fluid optimization and with today’s lower positive end-expiratory pressures (PEEPs).
Arterial, Central Venous, and Pulmonary Artery Catheters
Arterial Catheters
What Do They Offer?
Arterial Pressure Measurement
Fluid Responsiveness
Access
Central Venous Catheters
What Do They Offer?
Measurement of Central Venous Pressure
Access to Blood in Superior Vena Cava
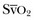 ). Although absolute values of ScvO2 are not identical to single
). Although absolute values of ScvO2 are not identical to single 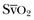 values, trends in ScvO2 over time follow the same pattern as trends in
values, trends in ScvO2 over time follow the same pattern as trends in 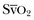 , making ScvO2 a useful measure in patients who do not require an arterial catheter.7 ScvO2 can be obtained either intermittently (by withdrawal of blood samples) or continuously (with the use of a catheter equipped with fiberoptic fibers).
, making ScvO2 a useful measure in patients who do not require an arterial catheter.7 ScvO2 can be obtained either intermittently (by withdrawal of blood samples) or continuously (with the use of a catheter equipped with fiberoptic fibers).
Access
Complications
Pulmonary Artery Catheters
What Do They Offer?
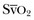 ).
).
Pressures
Right Atrial Pressure
Pulmonary Artery Occlusion Pressure

Full access? Get Clinical Tree