Fig. 61.1
A timeline of advances in cardiac anesthesia and surgery. Anesthesia by Morton enabled modern surgery, allowed the development of myriad new surgical procedures. Conversely, as Fig. 61.1 shows, the development of new surgical procedures, in this case those on the heart, came before and animated the development of people, techniques, and pharmacological approaches to support the work of the surgeon, developments led especially by anesthesiologists. Note that the figure does not include three events of import in the nineteenth century: tracheal intubation, first heart surgery, and positive pressure ventilation, all accomplished by surgeons
Modern cardiac surgery began with operations on intrathoracic blood vessels’rather than on the heart itself. In 1938, at the Children’s Hospital of Boston, surgical resident Robert Gross (1905–1988) successfully ligated a patent ductus arteriosus, a daring unsupervised feat. William Ladd, “the surgeon,” was away on holiday! In 1944, Clarence Crafoord (1899–1983) repaired a coarctation (narrowing) of the aorta in Stockholm [5].
In Baltimore, in 1944 Alfred Blalock (1899–1964) performed the first palliative surgical procedure for cyanotic congenital heart disease (blue baby). The Blalock-Taussig operation involved creating an artificial connection (shunt) between one of the major systemic arteries and the pulmonary artery, enabling greater mixing of the oxygenated and deoxygenated blood in the circulation. In 1946, Merel Harmel (1917-; Fig. 61.2) and Austin Lamont (1905–1969) described the anesthesia for the first 100 Blalock-Taussig Operations, the first cardiac anesthesiology paper [8]. Here follows an abridged contemporary memoire from Harmel (personal communication):

Fig. 61.2
Merel Harmel was the first to administer anesthesia for the successful paliation of Tetrology of Fallot. (Photograph from the private collection of J. Reves)
“About two weeks before the operation, Lamont mentioned that we might be faced with operations upon the heart and/or great vessels. Imagine my surprise, apprehension and excitement, when shortly before the operation was scheduled, he informed me that Dr. Blalock proposed to operate upon a 15 month old cyanotic infant with a tetralogy of Fallot, weighing 4 Kg. Earlier, Lamont had refused to anesthetize the infant for some minor operation because he felt she would not survive. So the prospect of a major experimental procedure involving a thoracotomy would surely hasten the baby’s death. Blalock however was determined to operate. He had discussed the seriousness of the operation with the family, telling them the operation had never been attempted before and the chances were good that the child might die on the operating table. (This is a sterling example of providing informed consent years before the requirement was widely recognized! EL and JGR)
“The day the operation was posted, Lamont offered me the ‘opportunity’ to anesthetize the baby! He would of course be with me in the operating room. He thought that the experience of anesthetizing this infant would be educational (I was just completing my 11th month of training). In 1944, at Hopkins there was no appropriate equipment for conducting endotracheal anesthesia in 4 Kg. infants. We thought to fashion an endotracheal tube from a large bore urethral catheter should intubation be necessary.
“On the 29th of November, Eileen Saxon was brought to the operating room. She was anesthetized with open drop ether, and oxygen was delivered by catheter under the mask. The only monitor was a finger on the carotid pulse. Blalock had Vivian Thomas (an African American laboratory assistant who had devised the procedure in Blalock’s dog lab and who is credited with much of the success of the operation) at his side during the procedure. William Longmire (later the Chairman of Surgery at UCLA) and Denton Cooley (subsequetly a pioneering cardiac surgeon in Houston) assisted Blalock, performing a subclavian to pulmonary artery anastomosis in 1 hour and 45 minutes. Helen Taussig who had proposed the concept of the operation to Blalock was also in the room. When the subclavian and pulmonary arteries were clamped, the pulse became slow and almost imperceptible. Breathing became shallower and cyanosis deepened. The outlook appeared dismal, but she rallied and the operation continued. Toward the end of the operation we thought she might be obstructed and attempted unsuccessfully to intubate her trachea with our makeshift catheter. Positive pressure was applied with our smallest mask in an effort to re-expand her lung. At the end of the procedure her color dramatically improved. The atmosphere was jubilant. Against seemingly insuperable odds, she had miraculously survived and dramatically improved. Eileen recovered and was discharged almost two months later.
“A palpable excitement accompanied the operation, a feeling that something historic was happening. The initial presentation by Blalock and Taussig to the Hopkins Medical and Surgical Association meeting was accompanied by a standing ovation. The “blue baby” turning pink caught the hearts and imagination of parents, surgeons and the general populace. Surgeons from around the world came to see Blalock operate, and patients were referred from home and abroad. By 1946, when our article was published, we had completed over 200 operations, most with an anesthetic technique suggested by Elisabeth Lank (Fig. 61.3), the nurse anesthetist at the Boston Children’s Hospital who had demonstrated the advantages of managing infants with intubation and anesthesia with cyclopropane.”
Fig. 61.3
Elizabeth (Bessie) Lank was a nurse anesthetist of enormous clinical skill. She taught many anesthesiologists who later became leaders in cardiac and pediatric anesthesia. As indicated in the text, she gave the anesthesia for many “firsts” in cardiac surgery. (Courtesy of Mark Rockoff and the archives of Boston Children’s Hospital)
Before 1948, except for repairs of wounds to the heart during World War II, cardiovascular surgeons did not invade the heart or touch its valves. The temptation however, to relieve mitral stenosis (a narrowing of the valve joining the two chambers of the left side of the heart) by dilating the narrowed valve, was great. On January 29, 1948, Horace Smithy of Charleston (Medical University of South Carolina) successfully performed the first mitral commissurotomy using a device he designed [9]. The commissurotomy was performed “closed,” blindly reaching the deformed valve through a small hole made in the heart’s left atrium. Smithy’s affliction with mitral stenosis, from which he died at age 34, surely animated this heroic effort. Smithy had tried to convince Blalock to perform the operation on him, but Blalock refused.
In 1950, Charles Bailey (1910–1993) attacked aortic valve stenosis, performing a closed aortic valvotomy in Philadelphia [10]. With both major left sided heart valves surgically accessible, cardiac surgery had reached major milestones of palliating and/or correcting congenital and valvular heart disease.
Two fears daunted these early cardiac surgeons. One was fear of damage to the heart, either physical damage or damage consequent to impaired blood flow to the heart, injuring the heart muscle so it could not pump effectively. A second parallel fear was concern that they might impair blood flow to the brain and injure that vital organ.
The cardiac surgeon and anesthesiologist extended the time to safely abolish blood flow to the heart and brain by cooling the patient. Cold protects the bloodless brain or heart by decreasing tissue metabolism and oxygen consumption. In the “early days”, i.e., the 1950s,’the anesthetized patient was lowered into a tub of ice water with ice cubes floating in the tub’a tricky business because if the patient became too cold, the heart might stop and not start again. This limited the lowest temperature to about 28°C. Furthermore, with insufficient anesthesia, the patient shivered, opposing cooling. Cooling for cardiac surgery was debated and even transiently abandoned over the past 50 years. Conventional practice now uses moderate cooling (to 32–34°C).
A few procedures required cessation of blood flow to heart and/or brain, and for these a much lower temperature was achieved, sometimes in just the heart by application of cardioplegia (see below) [11]. Sometimes so called deep hypothermic cardiac arrest (DHCA) was required for brain protection [12]. In DHCA the entire body was cooled to <20°C and the circulation halted, while complex congenital anomalies in infants, and (rarely) the aorta in adults, were repaired.
Coronary artery disease, the limitation of arterial blood flow to the heart causing oxygen deprivation, and inadequate function or death of the heart muscle, became recognized as the greatest killer in industrialized countries. It was a topic of surgical attention as early as the 1930s [13]. How could the cardiac surgeon fix this problem?
Two early operations were developed by Vineberg. The first interrupted the two internal mammary arteries, coursing down the back of the sternum or breast bone, on the premise that bilateral ligation (BIMAL) of these vessels “downstream” of the heart would increase “upstream” flow to the coronary arteries. This operation achieved increasing popularity in the mid to late 1950s because it was simple, safe andsome patients experienced decreased severity and/or frequency of angina pectoris. However, animal studies failed to confirm the premise, and many doubted the improvement. Two groups then performed randomized, “double blind” placebo controlled studies in a total of 35 patients [14,15]. The control patients had a sham operation consisting of only a skin incision. These studies each revealed random and equivalent improvements of angina among patients who had undergone the placebo operation and internal mammary ligation, proving the ineffectiveness of the operation and putting an end to its performance. These results prompted anesthesiologist Henry Beecher’s landmark article, arguing that it was unethical to perform new surgical operationswithout first establishing effectiveness [16]. Unfortunately, this call has been largely unheeded.
Contemporary cardiac surgery lacks placebo-controlled, sham operated studies of effectiveness, a reluctance based largely on claims that performing hazardous sham operations is unethical. Whether this is a wise decision, or whether more lives would be preserved and fewer people subjected to hazardous and ineffective operations by conducting sham studies in fully informed cardiac surgical candidates, remains an ethical conundrum that probably will not be confronted.
The second Vineberg operation for relief of angina pectoris actually improved (albeit not immediately) blood flow to the heart in both experimental animals and patients. Devised in the 1940s, and performed in patients in the 1950s and 1960s, the Vineberg operation implanted an internal mammary into the heart muscle [17], and over a period of weeks thereby brought new collateral vessels to the heart. These patients posed a challenge to the anesthetist because the heart lacked adequate blood flow, the muscle was injured by the operation itself and no immediate improvement in flow accompanied the additional stress of surgical trauma. Managing these patients inspired Earl Wynands (Fig. 61.4) to develop principles for anesthetic management of patients with severe coronary artery disease that are valid to this day [18]. Most importantly, Wynands sought to maintain coronary artery perfusion pressure by infusing phenylephrine as needed. He was the first to recognize that patients with unstable angina sustained a higher mortality rate than those with stable angina. Another major contribution was his observation that a resting electrocardiogram was of no use in predicting which patients would experience perioperative problems. Finally, he promoted the life-saving notion of maintaining tracheal intubation and ventilatory support postoperatively.

Fig. 61.4
Earl Wynands who advanced cardiac anesthesiology despite being more or less blind from macular degeneration
The inability of the Vineberg procedure to immediately improve coronary blood flow led, in 1967, to a third operation, one constructing a vascular (initially a vein but later a segment of artery) conduit between the ascending aorta and the diseased coronary artery segment, the coronary artery bypass graft operation or CABG [19]. It achieved almost immediate acceptance, and has been performed (without placebo controlled proof of effectiveness) upon millions of people worldwide.
Now we retreat to the 1950s to describe developments enabling modern cardiac surgery on the valves and vessels of the heart. In 1952, Gross (now having graduated from residency) performed the first true “open heart surgery”, repairing an atrial septal defect (a hole between the right and left sides of the heart). Widely opening the right atrium and attaching a “well” to avoid the lethal entrainment of air into the circulation, he closed the hole in the septum, blindly suturing below the air-blood interface in the well [5].
However, applying the axiom that “necessity demands invention”, widespread conduct of open heart surgery required invention of the heart-lung machine (cardiopulmonary bypass). This device collected venous blood, oxygenated it, removed carbon dioxide, and pumped the nourishing arterialized blood back into the aorta (i.e., back to the body). Thus, while the heart was not pumping and the lungs were not functioning circulation of oxygenated blood to the body was continued., Such a device permitted operations on the heart using direct vision. While a premedical student in 1952, one of the authors, EL, heard with great excitement that this seemingly impossible feat of stopping the heart and repairing it without killing the patient would soon become feasible. Indeed, it became a reality within the year.
On May 6, 1953, John Gibbon Jr (1903–1973) at Jefferson Medical College in Philadelphia, first successfully used a heart-lung machine in a patient in whom he closed an atrial septal defect in an empty heart under direct vision [20]. He and his wife Mary had taken two decades to develop the machine beginning the age of true invasive open heart surgery. He found minimal support for his initial work in the Department of Surgery at the Massachusetts General Hospital (MGH)’his chief, Edward Churchill, suggested that it had little promise. This prompted Gibbon to relocate to Jefferson Medical College in Philadelphia. During the ensuing 20 years, he addressed problems concerning filtration and coagulation of blood, and reestablishing normal coagulation after completion of the surgery. He had to enable oxygen and carbon dioxide gas exchange (as in the alveoli), and devise a pump that could reliably and controllably perfuse the systemic circulation.
Gibbon’s invention, refined and adopted by many, revolutionized cardiac surgery by giving the surgeon a bloodless field and a time limited by how long a hypothermic heart could tolerate no coronary blood flow (about 30 minutes). The reintroduction in 1973, of myocardial protection techniques with a cold hyperkalemic (high potassium concentration) solution (cardioplegia) facilitated the expansion of cardiac surgery [11], giving surgeons well over an hour to perform their work. Because the heart resists longer periods of ischemia than does the brain, Arthur Keats (1923–2007) commented: “The heart is a very tough organ, and when it malfunctions, we simply cannot look to a brief period of ischemia for its failure” [21]. However, the heart is not invincible, and prolonged ischemia during intra-cardiac surgery required a protective strategy. Cardioplegia during intentional interruption of coronary blood flow to the heart was second only to the invention of the heart-lung machine as a factor in the development of cardiac surgery.
Perhaps the last major surgical accomplishment was heart transplantation. Christian Barnard (1922–2001) performed the first cardiac transplant in Capetown, SA in 1967 [5]. The anesthesiologist, J Ozinsky reported: “From the anesthetist’s point of view, it was not anticipated that we would be faced with anything we had not been faced with before, and the anesthesia was conducted with optimism” [22]. Ozinsky and Barnard did not fear their daring venture into the unknown. Perhaps, however, they ventured too soon. Barnard’s patient lived but a short time. Norman Shumway (1923–2006) of Stanford University thought the effort was premature. He believed that success required the development of a multidisciplinary team, one particularly focused on issues of tissue rejection. Indeed, the discovery of the immunosuppressive drug cyclosporine, provided the key to success [21].
Cardiac Anesthesiology
“Anesthesia for patients undergoing cardiac surgery” differs, in our minds, from “cardiac anesthesiology.” Skillful physicians and nurses provide the former; individuals developing the science and art so that others may practice it more safely comprise the latter. Using this definition, we can point to a number of cardiac anesthesiologists. Kenneth Keown’s 1951 description of the anesthetic management and hypothermia for patients undergoing mitral commissurotomy [23] recognized the importance of light anesthesia with minimal cardiac depression, and the benefit of letting the temperature drift down, i.e. of producing mild hypothermia. Artusio’s use in the early 1950s of “ether analgesia” during mitral commisurotomy may be the ultimate in light anesthesia. Patients managed in this fashion responded appropriately to questions and commands during the procedure but experienced no pain or recall [24]. Patrick et al.’s 1957 [25] description of anesthesia for patients undergoing cardiopulmonary bypass, merits special attention, because they used animal experimentation to test hypotheses regarding control of physiologic variables such as pump flow, temperature, and blood gas management during cardiopulmonary bypass. They surmised that these factors were of greater interest than the anesthetic management per se’: “The management of anesthesia for patients undergoing intracardiac surgery by means of a Gibbon type pump-oxygenator differs in no important respect from the anesthesia management of patients for ordinary intrathoracic procedures.” Their work established the principle, that attaining physiologic ends was paramount rather than the means to achieve them.
Monitoring
The first patients undergoing cardiac surgery were anesthetized with minimal monitoring, such as a finger on a temporal or carotid artery! However, as the surgery became more complex, and particularly after cardiopulmonary bypass was employed, monitoring of hemodynamic variables became an important component of cardiac anesthesiology. In 1972, Civetta and Gabel described the use of the pulmonary artery (Swan-Ganz) catheter intra and postoperatively [26], and in 1973 Lappas et al. demonstrated that pulmonary capillary wedge pressure accurately reflected left atrial pressure intraoperatively, which in turn reflected left heart filling and function [27]. Indeed, invasive monitoring and expertise sometimes seemed to define cardiac anesthesiologists, who demonstrated their expertise by their ability to “slip in” invasive lines quickly and deftly, equating this skill, perhaps erroneously, with superior knowledge. The need for arterial pressure measurement and access to arterial blood samples provided sufficient reason for inserting an indwelling arterial catheter. Measurement of central venous pressure seemed similarly beneficial in helping estimate adequacy of blood volume and heart function, although echocardiography is now considered a better reflection. At the MGH, Laver (personal communication) explained the philosophy of hemodynamic measurement versus the clinician’s intuition: “If the measurements contradict your clinical impression, change your clinical impression!”
The introduction of the pulmonary artery catheter in the early 1970s provoked vigorous controversy. Some outstanding cardiac anesthesiologists considered this technology essential for optimal intra- and postoperative management. Others eschewed the routine use of such costly catheters, fearing the complications they could bring (e.g., death), or unconvinced that the additional data facilitated a positive outcome. Reports in 1987 indicated that information supplied from a pulmonary artery catheter added little to the successful management of many cardiac surgical patients [28,29]. Like all tools and drugs, the risks and benefits needed to be balanced. In selected patients, particularly those with impaired pumping function of the heart at any perioperative stage, the use of a pulmonary artery catheter facilitated successful management, but in many others it imposed unnecessary risks and expense [28,29].
Until recently, monitoring in cardiac anesthesia practice lacked visualization of the anatomy of the heart. Anatomy is crucially important because many operations propose to repair or palliate anatomic defects such as incompetent mitral valves or ventricular septal defects, and because visualization of malfunctioning areas of the heart can provide clues to explain impaired circulation. Cardiac anesthesiologists and surgeons had no view of the internal anatomy of the heart as complexity of surgery progressed. Transesophageal echocardiography (TEE) changed that.
The 1980 introduction of M-mode (one dimensional) TEE by Matsumoto [30] was followed in 1982 by Cahalan’s description of 2-dimensional TEE [31], and deBruijn and colleagues’ report of color-flow Doppler TEE in 1987 [32]. Each of these gave different information. M-mode alone was least useful, almost uninterpretable in real time. Two dimensional TEE allowed a rapid meaningful interpretation of structure. Color Doppler TEE enabled visualization of flow allowing anatomic and physiologic interpretations (e.g. seeing turbulent backward flow through an aortic valve means significant aortic valve regurgitation.) Most recently, TEE allows a 3-dimensional view of structures and even better visualization [33]. TEE has permitted cardiac anesthesiologists to literally look into the heart and become intraoperative cardiac diagnosticians, the intraoperative counterpart of the cardiologists providing the preoperative diagnosis guiding the surgeon to the appropriate procedure. Cardiac anesthesiologists can now inform surgeons in real time, of new intraoperative findings, make new diagnoses, confirm proper repair of the cardiac lesion, and specify needs for further immediate surgical management [21].
TEE has transformed the intraoperative experience for patients, anesthesiologists and surgeons alike, particularly in congenital [22] and valvular surgery. Clinical anecdotes demonstrate its value: A surgeon believes a ventricular septal defect in a 6-year-old child has been successfully closed. However, when preparing to terminate cardiopulmonary bypass, high pulmonary artery pressures and blood gas analysis indicate a continuing left to right shunt, and examination by TEE reveals the precise location of a second major but previously unappreciated hole in the ventricular septum. Before the advent of TEE, the second hole would have awaited an emergency postoperative cardiac catheterization, and would have required a second operation to obtain a satisfactory result. Likewise in adults with mitral valve rupture, the anesthesiologist determines which valve leaflet is flailing and whether it is repairable or requires a prosthesis. Once the repair is done, the anesthesiologist assesses valvular integrity. This diagnostic information has reduced re-operations and prevented further heart damage. No other monitoring technology has provided comparable benefit to intra-operative decision making.
Anesthetic Drug Regimen (or Anesthetic Pharmacology)
Before 1969, cyclopropane, ether or halothane dominated anesthesia for cardiac surgical patients. The flammability of the first two led to dominance of the non-explosive halothane. Death during induction of anesthesia, and inability to differentiate cardiac from pulmonary failure after cardiopulmonary bypass and postoperatively, were common. A search for better approaches to anesthesia led to the introduction, at MGH, of high dose morphine. This was the era before coronary revascularization; cardiac surgical patients had primarily valvular or congenital disease. Reminiscing about this introduction, Lowenstein (Fig. 61.5) wrote: “At least three things were necessary to set the stage for a new concept of anesthesia for our most ill patients: an environment that tolerated and even encouraged radically creative solutions; a clinical problem that caused an unacceptably high death rate; and a cast of characters with imagination, vision, courage, and clinical credibility” [34]. It was noted that large doses of intravenous (IV) morphine given postoperatively could relieve pain and, by depressing ventilation, could enable patients to tolerate tracheal intubation and mechanical ventilation. Morphine had recently been demonstrated to not depress heart contraction [35]. Why not deliver large doses of morphinepreoperatively to produce insensibility yet not impose cardiovascular collapse during induction? Continuing this regimen postoperatively would produce the already appreciated salutary benefits.

Fig. 61.5
A photograph of Edward Lowenstein (left) and Jerry Reves, happily at the water’s edge
The paper, published in 1969 in theNew England Journal of Medicine, documented the hemodynamic effects of enormous doses of morphine in patients with valvular heart disease and patients without heart disease, and changed the conduct of cardiac anesthesia [36]. It increased interest in the physiological consequences of drugs used to conduct cardiac anesthesia, for example, documenting a different hemodynamic response to the same drug in patients with and without valvular heart disease. Valvular heart disease leads to heart failure and large blood volumes that predispose to pulmonary edema. Morphine improved the circulation of these patients by causing vasodilation, decreasing systemic vascular resistance and reducing the work required of the heart. On the other hand, patients with uncomplicated coronary artery disease had neither increased blood volume nor cardiac failure, nor had they adapted to the continual release of endogenous catecholamines as had patients with valvular heart disease. Thus, in patients with uncomplicated ischemic heart disease, surgical stimulation during morphine anesthesia was accompanied by increased blood pressures and heart rates, dangerous changes that increased myocardial oxygen demand and consumption.
The increasing number of patients with coronary artery disease prompted a search for better cardiac anesthesia techniques. In retrospect, this was ironic since Wynands’ 1967 article had defined many of the optimal principles, if not specific drugs, for managing such patients [18]. “Recipes” followed, each describing different “ideal” anesthetic regimens. However, two randomized trials in the early 1970s failed to demonstrate an outcome difference with different anesthetic drug regimens [37,38]. A 1978 trial advocated “industrial-sized” doses of fentanyl, a synthetic opioid devoid of the histamine liberating effect of morphine, to provide “stress-free” anesthesia [39].
The originators of high-dose “opioid anesthesia” documented limitations of the technique including patient awareness [40,41]. Patient awareness may occur more commonly in cardiac surgery than in other settings, because less anesthetic may be given to avoid cardiac depression, and because high opioid doses provide unreliable amnesia. As early as 1951’before opioid use as anesthetics’awareness was recognized as a possible outcome: “At the time of the pre-anesthetic examination, and again before anesthesia is induced, an attempt is made to explain the stages of the procedure of which the patient may be aware during operation” [23]. It continues to be a problem. In 2007, Pollard described the incidence of awareness in 87,361 surgical patients [42]. Of the six who remembered events, 4 had undergone cardiac surgery.
The obligatory period of postoperative ventilation was an initial stimulus for using high-dose opioids. While advantageous in many patients, it proved to be unnecessary in others, and may be accompanied by awareness. Subsequent small and then large studies revealed, not surprisingly, that there is no single ideal “cardiac anesthetic” [37,38,43,44]. Harmel and Lamont recognized this in 1946: “…one cannot be dogmatic in regard to choice of anesthetic agent and technique” [4]. Optimal care for any given patient requires matching patient pathophysiology with the pharmacology of anesthetic and other drugs by knowledgeable and skilled clinicians [45].
The Cardiac Anesthesiologist
While choice of anesthetic regimen may matter little, Slogoff and Keats at the Texas Heart Institute found that the choice of anesthesiologist did [46]. In their study of postoperative myocardial infarction as a function of anesthetic choice, in patients operated upon for coronary artery disease, they incidentally found that outcomes of patients anesthetized by one of the nine attending anesthesiologist’s stood out. The frequency of myocardial infarction associated with “Anesthesiologist Number Seven,” significantly exceeded that for the other eight [34]. Patients anesthetized by number seven had more variability of blood pressure and heart rate, the two factors most affecting myocardial supply and demand. Whether this was due to inattention, pharmacologic ignorance, poor choice of drugs or drug doses, or chance, is not known.
Present day cardiac anesthesiologists deliver a mixture of drugs. Moderate doses of an opioid (usually fentanyl or sufentanil) are used to minimize adrenergic responses to noxious stimulation. Muscle relaxants keep patients from moving. Potent inhaled anesthetics augment relaxant effect, protect the heart [47], modify hemodynamic perturbations, and together with benzodiazepines, curtail memory of intraoperative events.
Vexations in the Present Care of the Cardiac Surgical Patient
The level of blood pressure during cardiopulmonary bypass has periodically been a matter of much controversy’the primary concern being whether inadequate brain perfusion during non-pulsatile CPB produced postoperative brain damage. After several decades addressing this issue, Newman et al demonstrated in 1994, that cerebral blood flow is well preserved during cardiopulmonary bypass as long as CO2 levels are normal and perfusion pressure is maintained within the autoregulatory range of 50 to 90 mmHg [48]. The controversy disappeared.
How long should postoperative tracheal intubation continue? In his 1967 study of anesthesia for patients undergoing Vineberg operations, Wynands discovered that postoperative tracheal intubation and elective mechanical ventilation increased survival [18]. In 1968, John Viljoen at the Cleveland Clinic confirmed this result in a larger surgical population, decreasing mortality from 10 to 2% [49]. Such findings strengthened the case for high-dose opioid anesthesia. Immediate removal of tracheal tubes and resumption of spontaneous breathing after cardiac surgery became rare.
But nothing comes for free. Prolonged controlled ventilation through a tracheal tube also carried risks of injury to the trachea, trauma to the lung, and pulmonary infection. Newer shorter-acting opioids and anesthetics decreased the need for prolonged ventilation that the large doses of morphine had imposed. In the 1970s, when aortocoronary bypass graft operations in patients with normal heart function became common, earlier tracheal extubation was advocated to minimize complications. This also decreased intensive care unit (ICU) and hospital stay and costs. But policy then went too far. Many “fit” patients having coronary artery grafts had tracheal extubation in the operating room, as though they had just undergone a simple herniorraphy. If it was possible, it was done…and frequently…for a while. However, urgent postoperative reintubation sometimes followed tracheal extubation in the operating room. Reintubation was required because of respiratory insufficiency, cardiac arrhythmias or dysfunction, or bleeding. Rather than waiting a few hours after establishing satisfactory postoperative conditions, tracheal extubation in the operating room did not improve patient outcomes, nor did it reduce ICU or hospital stay, or cost. The fad passed.
“Off-pump” coronary artery bypass to treat coronary artery disease became a popular deviation from optimum care in the late 1990s. As cardiologists expanded catheter-based methods to treat coronary artery disease, surgeons responded with their own approaches. Coronary artery bypass graft (CABG) surgery was performed without cardiopulmonary bypass (“off-pump CABG”), prompted by the notion that avoiding cardiopulmonary bypass (CPB) would prevent the adverse consequences of CPB on the brain. The lack of CPB required that the heart continue pumping while in unnatural positions and while grafts were placed in difficult-to-reach locations. These positions often compromised venous return and cardiac output, requiring vasopressors to maintain blood pressure and avoid myocardial ischemia. Surgeon Denton Cooley quipped: “We have successfully made cardiac surgery more painful for the surgeon (and anesthesiologist) than the patient.” Studies subsequently established that the revascularization achieved was less effective, and that the brain did not benefit [50]. This fad has also faded.
Influential Individuals and Groups
The authors of this chapter are not professional historians, and have wrestled with the problem of how to recognize contributions made by individuals, versus those made by groups united by their motivation to improve the care and outcome of patients. By its very nature, cardiac anesthesiology must be practiced collaboratively, if only because surgeons, nurses, perfusionists, cardiologists and laboratories are requisites. Nevertheless, we have done our best to differentiate individuals versus groups making particularly important contributions. The historian also tends to provide a chronologic tale. We found this challenging due to the brief period we describe, the fact that many individuals and groups emphasized different activities during this period, and affiliations changed as people worked in different institutions and with different people.
Thus, arbitrarily, we have divided this section into Individuals and Groups who, in our opinion, most altered the development of cardiac anesthesiology (Table 61.1 and Fig. 61.6). The former tend to be those either working alone or with a small group, or who made contributions despite moving to different environments, whereas the latter tended to be gatherings of investigators whose collaborations appeared to synergize each other to make the effort stronger.
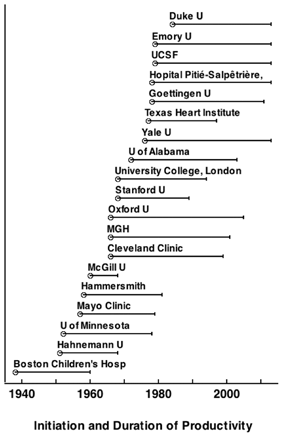
Fig. 61.6
Dozens of institutions and greater numbers of individuals contributed to the advancement of cardiovascular anesthesia and the management of patients with cardiovascular disease. This figure indicates many of those institutions androughly illustrates when the individuals in those institutions began their contributions, and the period over which they had their greatest impact
Table 61.1
Groups and Individuals Influencing the Development of Cardiac Anesthesiology
Initial year & duration of activity | Facultya | School, city |
|---|---|---|
1938–1959 | Lank, Smith | Boston Children’s Hosp, Boston |
1951–1967 | Keown | Hahnemann, Philadelphia |
1952–1977 | Buckley, VanBergen | University of Minnesota, Minneapolis |
1957–1978 | Theye, Patrick, Tarhan, Tinker, Moffitt | Mayo Clinic, Rochester |
1958–1980 | Sykesb | Hammersmith |
1960–1967 | Wynands | McGill University, Montreal |
1966–1998 | Viljoen, Estafanous | Cleveland Clinic, Cleveland |
1966–2000 | Laver, Lowenstein, Bland, Hallowell, Dalton | MGH, Boston |
1966–2004 | Prys-Roberts, Foëx, Sykes | Oxford University |
1968–1988 | Ream, Garman | Stanford University, Stanford |
1968–1993 | Branthwaite | University College, London |
1972–2002 | Lell, Reves, Samuelson, Allarde, Oget | University of Alabama, Birmingham |
1976–present | Barash, Hines | Yale University, New Haven |
1977–1996 | Keats, Slogoff | Texas Heart Institute, Houston |
1978–2010 | Sonntag, Kettler | Göttingen, Germany |
1978–present | Coriat | Hospital Pitié-Salpêtrière, Paris |
1979–present | Cahalan, Mangano, Wallace, Cason

Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|