PREVENTIVE MEASURES
Aspiration of pathogenic organisms from the oropharynx is believed to be the inciting event in most cases of VAP (10). The pathogens that most often colonize the oropharynx in ICU patients are Gram-negative aerobic bacilli (see Figure 5.5 on page 89), which explains the predominance of these pathogens in VAP. The instances of VAP that appear within 4 days of intubation are most likely caused by pathogens that have been dragged into the airways during the intubation procedure.
Oral Decontamination
The realization that VAP begins with pathogenic colonization of the oropharynx prompted the introduction of measures to decontaminate the oropharynx as a preventive measure for VAP. The methods of oral decontamination (i.e., with antiseptic agents and nonabsorbable antibiotics) are described in Chapter 5 (see pages 88–90), and the benefits of oral decontamination in reducing tracheal colonization and VAP are shown in Figure 5.6 (page 90). Oral decontamination (usually with the antiseptic agent chlorhexidine) is now a standard measure in all ventilator-dependent patients.
Routine Airway Care
(Note: This topic is described in more detail on pages 540–542 in the last chapter.) Tracheal tubes serve as a nidus for pathogenic colonization and biofilm formation, and these biofilms protect the colonized pathogens and allow them to proliferate (see Figure 28.4 on page 541). Endotracheal suctioning can disrupt the biofilms and dislodge microbes colonizing the inner surface of tracheal tubes, which introduces pathogens into the lower airways. For this reason, endotracheal suctioning is not recommended as a routine procedure (11), but only when necessary to clear secretions from the airways.
Clearance of Subglottic Secretions
Contrary to popular belief, inflation of the cuff on tracheal tubes to create a seal does not prevent aspiration of mouth secretions into the lower airways. Aspiration of saliva and liquid tube feedings has been documented in over 50% of ventilator-dependent patients with tracheostomies; in over three-fourths of the cases, this aspiration is clinically silent (12).
Concern about aspiration around inflated cuffs prompted the introduction (in 1992) of specialized endotracheal tubes equipped with a suction port just above the cuff (e.g., Mallinckrodt TaperGuard Evac Tube, Covidien, Boulder, CO). The suction port is connected to a source of continuous suction (usually not exceeding -20 cm H2O) to clear the secretions that accumulate in the subglottic region, as illustrated in Figure 29.1.
Clinical studies have shown a significant reduction in the incidence of VAP when subglottic secretions are cleared using specialized endotracheal tubes (13). As a result, suction clearance of subglottic secretions is recommended as a preventive measure for VAP (3). Despite this recommendation, aspiration of subglottic secretions has yet to gain widespread acceptance.
CLINICAL FEATURES
Diagnostic Accuracy
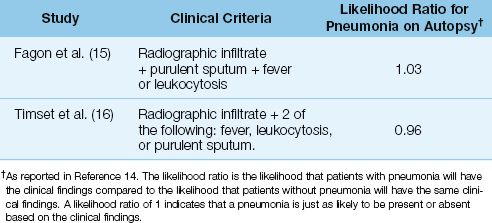
The traditional clinical criteria for the diagnosis of VAP include: (a) fever or hypothermia, (b) leukocytosis or leukopenia, (c), an increase in volume of respiratory secretions or a change in character of the secretions, and (d) a new or progressive infiltrate on the chest x-ray (4). Unfortu-nately, in patients who are suspected of having VAP based on these clinical criteria, the incidence of pneumonia on postmortem exam is only 30% to 40% (14). The diagnostic accuracy for VAP based on traditional clinical criteria is demonstrated in Table 29.2. This table shows the results of two studies that used autopsy evidence of pneumonia to evaluate the premortem diagnosis of VAP based on clinical findings (15,16). In both studies, the clinical criteria for identifying VAP were just as likely to occur in the presence or absence of pneumonia. These studies demonstrate that the diagnosis of VAP is not possible using clinical criteria alone.
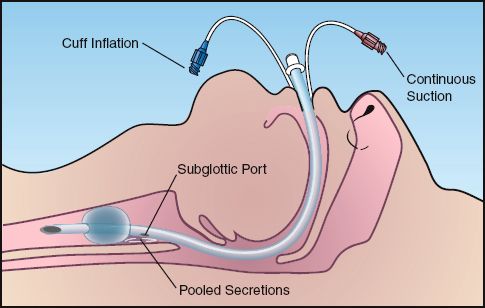
FIGURE 29.1 Endotracheal tube with a suction port placed just above the cuff to clear secretions that accumulate in the subglottic region.
Table 29.2 Predictive Value of Clinical Criteria in the Diagnosis of Ventilator-Associated Pneumonia
Specificity of Chest Radiography
The limited diagnostic accuracy in VAP is primarily due to the nonspecific nature of pulmonary infiltrates. Pneumonia accounts for only one-third of all pulmonary infiltrates in ICU patients (17,18), which means that conditions other than pneumonia are the most frequent cause of pulmonary infiltrates in ICU patients. The noninfectious causes of pulmonary infiltrates in the ICU include pulmonary edema, acute respiratory distress syndrome, and atelectasis.
An example of how a decrease in lung volume can produce spurious changes in a portable chest x-ray is illustrated in Figure 29.2. Both images in this figure were obtained within minutes of each other in the same patient. The image on the left, which was taken during expiration in the supine position (a common body position in ICU patients) shows a marked reduction in lung volume accompanied by a crowding of lung markings at the base of the right lung (dotted triangle). In a patient with fever, this radiographic change could be mistaken for a basilar pneumonia.
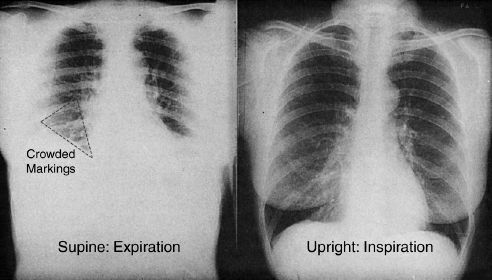
FIGURE 29.2 The effect of a decrease in lung volume on the appearance of a portable chest x-ray. Both images were obtained within minutes of each other in the same patient. The dotted triangle outlines an area of crowded lung markings that could be mistaken for a basilar pneumonia in a febrile patient. Image on the left digitally enhanced.
ARDS: The most common noninfectious cause of pulmonary infiltrates in ICU patients is the acute respiratory distress syndrome (ARDS) (18). This condition, which is described in Chapter 23, is an inflammatory disorder of the lungs that produces bilateral infiltrates on chest x-ray (see Figure 23.2 on page 451). Because it is often accompanied by fever, ARDS can be difficult to distinguish from a multilobar pneumonia.
Sensitivity of Chest Radiography
The other limitation of chest radiography is a limited sensitivity for the detection of pulmonary infiltrates. This is demonstrated in Figure 29.3, where the portable chest x-ray of a patient with cough and fever shows no apparent infiltrates, while the CT image of the same patient shows a fine pattern of infiltration in the posterior region of both lungs. Since CT scans are not obtained routinely in patients with suspected pneumonia and an unrevealing chest x-ray, it is not clear how often the example in Figure 29.3 actually occurs. However, this example clearly demonstrates that the diagnosis of pneumonia cannot be ruled out with a portable chest x-ray.
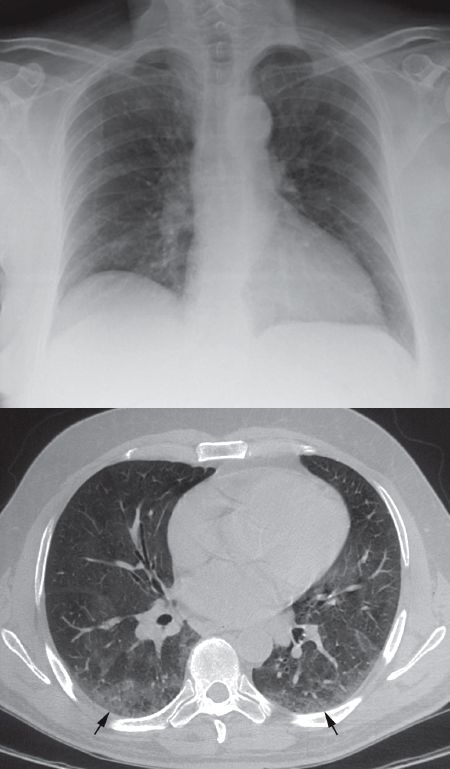
FIGURE 29.3 Demonstration of the limited sensitivity of portable chest radiography in the detection of pulmonary infiltrates. A portable chest x-ray of a patient with fever shows no apparent pulmonary infiltrates, while the CT image from the same patient reveals infiltrates in the posterior region of both lungs (indicated by the arrows).
New Clinical Criteria?
The National Healthcare Safety Network (NHSN) has recently published an algorithm for the diagnosis of VAP that does not include findings on a chest x-ray (1). This algorithm is shown in Figure 29.4. The clinical suspicion of VAP in this algorithm is triggered by a deterioration in arterial oxygenation (ventilator-associated complication) combined with a change in body temperature or leukocyte count in the blood (infection-related ventilator-associated complication). Although this algorithm has not been validated, it does recognize the limitations of chest radiography in the diagnosis of VAP.
MICROBIOLOGICAL EVALUATION
The diagnosis of VAP rests heavily on identifying a responsible patho-gen, but there is little agreement on the optimal method of either collecting or culturing respiratory secretions. Blood cultures have limited value in the diagnosis of VAP because organisms isolated from blood in cases of suspected VAP are often from extrapulmonary sites of origin (14). The NHSN algorithm in Figure 29.4 relies on quantitative cultures of respiratory secretions or lung biopsies for the diagnosis of VAP, but quantitative cultures are not a standard practice in the diagnostic evaluation of suspected VAP. The following is a brief review of the methods used to collect and culture respiratory secretions in the diagnostic approach to VAP.
Tracheal Aspirate
The traditional approach to suspected VAP involves aspiration of respiratory secretions through an endotracheal or tracheostomy tube, and these specimens can be contaminated with mouth secretions that are aspirated into the upper airway. A screening method for identifying contaminated and uncontaminated specimens is described next. (This method should be performed routinely in the microbiology laboratory.)
Microscopic Analysis
The cells identified in Figure 29.5 can help to determine if secretions collected by aspiration through tracheal tubes are contaminated by mouth secretions, and if there is evidence of infection. Each type of cell can be identified and interpreted as follows.
1. The squamous epithelial cells that line the oral cavity are large and flattened with abundant cytoplasm and a small nucleus. The presence of more than 10 squamous epithelial cells per low-power field (×100) indicates that the specimen is contaminated with mouth secretions, and is not an appropriate specimen for culture (1).

FIGURE 29.4 National Health Safety Network algorithm for the diagnosis of ventilator-associated pneumonia. From Reference 1.
2. Lung macrophages are large, oval-shaped cells with a granular cytoplasm and a small, eccentric nucleus. The size of the nucleus in a macrophage is roughly the same size as a neutrophil. Although macrophages can inhabit the airways (20), the predominant home of the macrophage is the distal airspaces. Therefore, the presence of macrophages, regardless of the number, is evidence that the specimen is from the lower respiratory tract.
3. The presence of neutrophils in respiratory secretions is not evidence of infection because neutrophils can make up 20% of the cells recovered from a routine mouthwash (20). The neutrophils should be present in abundance to indicate infection. More than 25 neutrophils per low-power field (x100) can be used as evidence of infection (21).
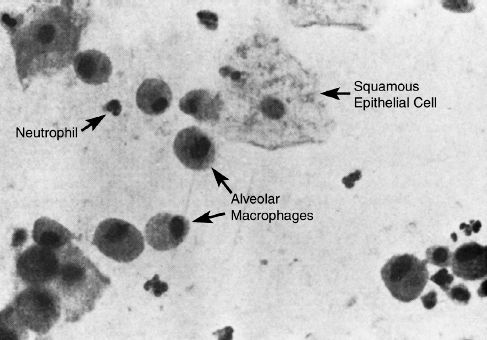
FIGURE 29.5 Microscopic appearance (magnification 400) of bronchial brushings from a ventilator-dependent patient. The paucity of squamous epithelial cells and the presence of alveolar macrophages is evidence that the specimen is from the distal airways (and thus would be an appropriate specimen for culture in a case of suspected VAP).
Qualitative Cultures
The standard practice is to perform qualitative cultures on endotracheal aspirates (where the growth of organisms is reported, but there is no assessment of growth density). These cultures have a high sensitivity (usually >90%) but a very low specificity (15–40%) for the diagnosis of VAP (22). This means that for qualitative cultures of tracheal aspirates, a negative culture can be used to exclude the diagnosis of VAP, but a positive culture cannot be used to confirm the presence of VAP. The poor predictive value of positive cultures is due to contamination of tracheal aspirates with secretions from the mouth and upper airways.
Quantitative Cultures
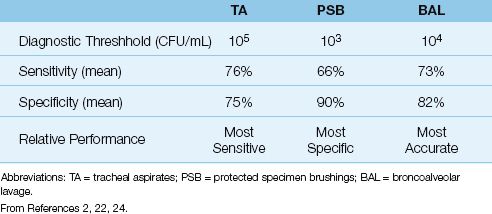
For quantitative cultures of tracheal aspirates (where growth density on the culture plate is reported), the threshold growth for the diagnosis of VAP is 105 colony-forming units per mL (CFU/mL). This threshold has a sensitivity and specificity of 76% and 75%, respectively, for the diagnosis of VAP (see Table 29.3) (2,22). Comparing these results to the sensitivity and specificity of qualitative cultures (i.e., sensitivity >90% and specificity ≤40%) shows that, for cultures of tracheal aspirates, quantitative cultures are less sensitive but more specific than qualitative cultures for the diagnosis of VAP.
Table 29.3 Quantitative Cultures for the Diagnosis of Pneumonia in Ventilator-Dependent Patients
Bronchoalveolar Lavage
Bronchoalveolar lavage (BAL) is performed by wedging the bronchoscope in a distal airway and performing a lavage with sterile isotonic saline. A minimum lavage volume of 120 mL is recommended for adequate sampling of the lavaged lung segment (23), and this is achieved by performing a series of 6 lavages using 20 mL for each lavage. The same syringe is used to introduce the fluid and aspirate the lavage specimen (only 25% or less of the volume instilled will be returned via aspiration). The first lavage is usually discarded, and the remainder of the lavage fluid is pooled and sent to the microbiology lab for microscopic analysis and quantitative culture.
Quantitative Cultures
The threshold for a positive BAL culture is 104 CFU/mL (1). The reported sensitivity and specificity of BAL cultures are shown in Table 29.3 (2,24). BAL does not have the highest sensitivity or specificity when compared to the other diagnostic methods in Table 29.3, but when sensitivity and specificity are considered together, BAL cultures have the highest overall accuracy for the diagnosis of pneumonia.
Intracellular Organisms
Inspection of BAL specimens for intracellular organisms can help in guiding initial antibiotic therapy until culture results are available. When intracellular organisms are present in more than 3% of the cells in the lavage fluid, the likelihood of pneumonia is over 90% (25). This is not done on a routine Gram’s stain, but requires special processing and staining, and will require a specific request of the microbiology lab.
BAL Without Bronchoscopy
BAL can also be performed without the aid of bronchoscopy using a sheathed catheter like the one illustrated in Figure 29.6. This catheter (COMBICATH,, KOL Bio-Medical, Chantilly, VA) is inserted through a tracheal tube and advanced “blindly” until it wedges in a distal airway. An absorbable polyethylene plug at the tip of the catheter prevents contamination while the catheter is advanced. Once wedged, an inner cannula is advanced for the BAL, which is performed with 20 mL of sterile saline. Only 1 mL of BAL aspirate is required for culture and microscopic analysis.
Nonbronchoscopic BAL (also called mini-BAL because of the lower lavage volume) is a safe procedure that can be performed by respiratory therapists (26). Despite the uncertainty regarding the location of the catheter tip in relation to the region of suspected infection, the yield from quantitative cultures with mini-BAL is equivalent to the yield with bronchoscopic BAL (2,27).
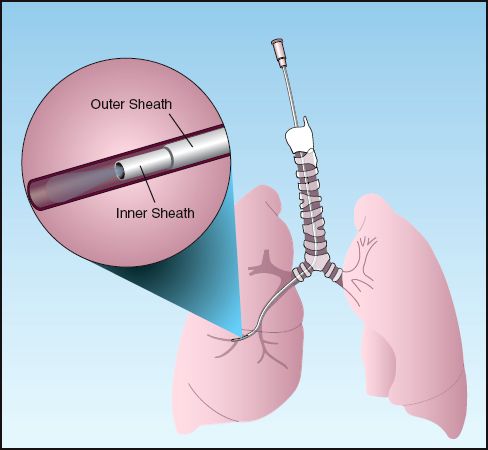
FIGURE 29.6 Protected catheter for performing bronchoalveolar lavage without the aid of bronchoscopy. See text for explanation.
Protected Specimen Brush
Aspiration of secretions through a bronchoscope produces false-positive cultures because of contamination as the bronchoscope is advanced through the tracheal tubes and upper respiratory tract (23). To eliminate this problem, a specialized brush called a protected specimen brush (PSB) was developed to collect uncontaminated secretions from the distal airways during bronchoscopy. The catheter design for the PSB is similar to the catheter design in Figure 29.6, and the brush is advanced from the inner cannula to collect samples from the distal airways.
Quantitative Cultures
The threshold for the diagnosis of VAP with PSB cultures is 103 CFU/mL (1,2), and the yield from PSB cultures is shown in Table 29.3 (2). A positive culture result has a low sensitivity (66%) but a high specificity (90%) for the diagnosis of VAP. Therefore, a negative PSB culture does not exclude VAP, but a positive PSB culture is considered evidence of VAP.
Which Method Is Preferred?
Despite the recommendations in clinical practice guidelines, there is little agreement about which microbiological method to use for the diagnosis of VAP, and qualitative cultures of tracheal aspirates continues to be popular. The following recommendations deserve mention.
1. The diagnostic yield from all culture methods is adversely affected by ongoing antibiotic therapy (2). Therefore, when possible, cultures should be obtained before antibiotics are started.
2. Most studies show that the mortality in VAP is not influenced by the microbiological method used to identify infection (2,28), and this observation is used to support the continued use of tracheal aspirates and qualitative cultures in the evaluation of VAP. However, there may be little mortality directly attributable to VAP, as mentioned earlier, so this observation may not be relevant to the selection of an appropriate microbiological method for the diagnosis of VAP.
3. If tracheal aspirates are used for the evaluation of suspected VAP, the specimens should be screened by microscopic examination and discarded if there is evidence of contamination with mouth secretions.
4. Quantitative cultures of tracheal aspirates are preferred to qualitative cultures because they have a higher specificity, and thus are more likely to detect VAP and identify the responsible pathogen(s). Management based on qualitative cultures of tracheal aspirates will result in the excessive use of antibiotics.
5. Non-bronchoscopic BAL provides a safe and relatively effective method of detecting VAP and identifying the responsible pathogen(s).
PARAPNEUMONIC EFFUSIONS
Pleural effusions are present in up to 50% of bacterial pneumonias (29), and these parapneumonic effusions typically do not require an intervention unless the conditions described next are indentified.
Indications for Thoracentesis
The typical indications for evaluation of a parapneumonic effusion in-clude the following:
1. The effusion is large or increasing in size.
2. There is an air-fluid level in the effusion or a hydropneumothorax (indicating a bronchopleural fistula and possible empyema).
3. The patient develops severe sepsis or septic shock.
4. The patient is not responding to antimicrobial therapy.
Evaluation of the pleural fluid should focus on appropriate stains and cultures in addition to a cell count, glucose level, and pH. Tests to identify an exudate or transudate are not necessary as these will not help to identify an infection.
Indications for Drainage
The indications for immediate drainage of a parapneumonic effusion include the following:
1. Evidence of a bronchopleural air leak (i.e., hydropneumothorax).
2. Grossly purulent pleural aspirate.
3. Pleural fluid pH <7.0
4. Pleural fluid glucose <40 mg/dL (<2.4 ∝mol/L).
Complicated parapneumonic effusions (i.e., a positive pleural fluid culture without evidence of an empyema) do not necessarily require drainage unless the patient’s condition does not improve or deteriorates on antimicrobial therapy.
ANTIMICROBIAL THERAPY
Antimicrobial therapy for pneumonia accounts for half of all antibiotic use in the ICU, and 60% of this antibiotic use is for suspected pneumonias that are not confirmed by bacteriologic studies (30). The aggressive use of antibiotics in cases of suspected VAP is fueled by studies showing that the mortality rate in VAP is increased by delays in initiating appropriate antibiotic therapy (31). However, there is a paucity of strong evidence directly linking VAP with fatal outcomes (4,6), and several studies show no direct relationship between VAP and mortality rate (7–9). (See the last section of this chapter for a comment on this situation.)
Empiric Antibiotic Therapy
Empiric antimicrobial therapy for VAP should include coverage for Gram-negative aerobic bacilli and Staphylococcus aureus (i.e., the principal pathogens listed in Table 29.1), unless the results of a sputum Gram stain or BAL specimen indicates one prominent type of organism. Accepted regimens include pipericillin/tazobactam or a carbepenem (imipenem or meropenem) or antipseudomonal cephalosporin (ceftazidime or cefepime) plus vancomycin or linezolid (2). (See Chapter 52 for recommended dosing regimens for these antibiotics).
There is a tendency to continue empiric antibiotics despite negative culture results if patients are improving, but this is not justified unless there is some other evidence of a treatable infection (e.g., a sputum Gram stain showing a dense population of organisms) (32).
Treatment of Documented Pneumonia
Antibiotic therapy of documented VAP will be dictated by the responsible pathogen(s) and by the antibiotic susceptibilities of pathogenic organisms in your hospital (i.e., the antibiogram).
Duration of Antibiotic Therapy
The traditional duration of antibiotic therapy for VAP has been 14 to 21 days (2). However, there is evidence that 8 days of antibiotic therapy for VAP is as effective as 15 days of therapy (33), and the popular opinion now is that one week of antibiotic therapy is adequate for most patients with VAP.
A FINAL WORD
The paucity of evidence directly linking ventilator-associated pneumonias with a mortality rate (4,6–9) suggests one or more of the following scenarios:
1. We’re really good at treating ventilator-associated pneumonias.
2. Ventilator-associated pneumonias are not life-threatening infections.
3. Ventilator-associated pneumonias are overdiagnosed, and many cases represent colonization or tracheobronchitis.
The first scenario seems unlikely, and the second scenario is the accepted choice, but it is very likely that the third scenario is the best answer, based on the results of the postmortem studies in Table 29.2.
REFERENCES
Clinical Practice Guidelines
1. Centers for Disease Control, National Healthcare Safety Network. Device-associated Module: Ventilator-Associated Event Protocol. January 2013. Available on the National Healthcare Safety Network website (www.cdc.gov/nhsn).
2. American Thoracic Society and Infectious Disease Society of America. Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia. Am J Respir Crit Care Med 2005; 171:388–416.
3. Muscedere J, Dodek P, Keenan S, et al. for the VAP Guidelines Committee and the Canadian Critical Care Trials Group. Comprehensive evidence-based clinical practice guidelines for ventilator-associated pneumonia: Prevention. J Crit Care 2008; 23:126–137.
General Information
4. Kollef MH. Ventilator-associated complications, including infection-related complications: The way forward. Crit Care Clin 2013; 29:33–50.
5. Vincent J-L, Rello J, Marshall J, et al. International study of the prevalence and outcomes of infection in intensive care units. JAMA 2009; 302:2323–2329.
6. Nguile-Makao M, Zahar JR, Francais A, et al. Attributable mortality of ventilator-associated pneumonia: respective impact of main characteristics at ICU admission and VAP onset using conditional logistic regression and multi-state models. Intensive Care Med 2010; 36:781–789.
7. Rello J, Quintana E, Ausina A, et al. Incidence, etiology, and outcome of nosocomial pneumonia in mechanically ventilated patients. Chest 1991; 100:439–444.
8. Papazian L, Bregeon F, Thirion X, et al. Effect of ventilator-associated pneumonia on mortality and morbidity. Am J Respir Crit Care 1996; 154:91–97.
9. Bregeon F, Cias V, Carret V, et al. Is ventilator-associated pneumonia an independent risk factor for death? Anesthesiology 2001; 94:554–560.
Preventive Measures
10. Estes RJ, Meduri GU. The pathogenesis of ventilator-associated pneumonia: I. Mechanisms of bacterial transcolonization and airway inoculation. Intensive Care Med 1995; 21:365–383.
11. AARC Clinical Practice Guideline. Endotracheal suctioning of mechanically ventilated patients with artificial airways 2010. Respir Care 2010; 55:758–764.
12. Elpern EH, Scott MG, Petro L, Ries MH. Pulmonary aspiration in mechanically ventilated patients with tracheostomies. Chest 1994; 105:563–566.
13. Muscedere J, Rewa O, Mckechnie K, et al. Subglottic secretion drainage for the prevention of ventilator-associated pneumonia: a systematic review and meta-analysis. Crit Care Med 2011; 39:1985–1991.
Clinical Features
14. Wunderink RG. Clinical criteria in the diagnosis of ventilator-associated pneumonia. Chest 2000; 117:191S–194S.
15. Fagon JY, Chastre J, Hance AJ, et al. Detection of nosocomial lung infection in ventilated patients: use of a protected specimen brush and quantitative culture techniques in 147 patients. Am Rev Respir Dis 1988; 138:110–116.
16. Timsit JF. Misset B, Goldstein FW, et al. Reappraisal of distal diagnostic testing in the diagnosis of ICU-acquired pneumonia. Chest 1995; 108:1632–1639.
17. Louthan FB, Meduri GU. Differential diagnosis of fever and pulmonary densities in mechanically ventilated patients. Semin Resp Infect 1996; 11:77–95.
18. Singh N, Falestiny MN, Rogers P, et al. Pulmonary infiltrates in the surgical ICU. Chest 1998; 114:1129–1136.
Microbiological Evaluation
19. Luna CM, Videla A, Mattera J, et al. Blood cultures have limited value in predicting severity of illness and as a diagnostic tool in ventilator-associated pneumonia. Chest 1999; 116:1075–1084.
20. Rankin JA, Marcy T, Rochester CL, et al. Human airway macrophages. Am Rev Respir Dis 1992; 145:928–933.
21. Wong LK, Barry AL, Horgan S. Comparison of six different criteria for judging the acceptability of sputum specimens. J Clin Microbiol 1982; 16:627–631.
22. Cook D, Mandell L. Endotracheal aspiration in the diagnosis of ventilator-associated pneumonia. Chest 2000; 117:195S–197S.
23. Meduri GU, Chastre J. The standardization of bronchoscopic techniques for ventilator-associated pneumonia. Chest 1992; 102:557S–564S.
24. Torres A, El-Ebiary M. Bronchoscopic BAL in the diagnosis of ventilator-associated pneumonia. Chest 2000; 117:198S–202S.
25. Veber B, Souweine B, Gachot B, et al. Comparison of direct examination of three types of bronchoscopy specimens used to diagnose nosocomial pneumonia. Crit Care Med 2000; 28:962–968.
26. Kollef MH, Bock KR, Richards RD, Hearns ML. The safety and diagnostic accuracy of minibronchoalveolar lavage in patients with suspected ventilator-associated pneumonia. Ann Intern Med 1995; 122:743–748.
27. Campbell CD, Jr. Blinded invasive diagnostic procedures in ventilator-associated pneumonia. Chest 2000; 117:207S–211S.
28. Shorr AF, Sherner JH, Jackson WL, Kollef MH. Invasive approaches to the diagnosis of ventilator-associated pneumonia: a meta-analysis. Crit Care Med 2005; 33:46–53.
29. Light RW, Meyer RD, Sahn SA, et al. Parapneumonic effusions and empyema. Clin Chest Med 1985; 6:55–62.
Antimicrobial Therapy
30. Bergmanns DCJJ, Bonten MJM, Gaillard CA, et al. Indications for antibiotic use in ICU patients: a one-year prospective surveillance. J Antimicrob Chemother 1997; 111:676–685.
31. Iregui M, Ward S, Sherman G, et al. Clinical importance of delays in the initiation of appropriate antibiotic treatment for ventilator-associated pneumonia. Chest 2002; 122:262–268.
32. Singh N, Rogers P, Atwood CW, et al. Short-course empiric antibiotic therapy for patients with pulmonary infiltrates in the intensive care unit. Am J Respir Crit Care Med 2000; 162:505–511.
33. Chastre J, Wolff M, Fagon J-Y, et al. Comparison of 8 vs. 15 days of antibiotic therapy for ventilator-associated pneumonia in adults. JAMA 2003; 290:2588–2598.

Full access? Get Clinical Tree








